444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The vials leak testing market plays a critical role in ensuring the safety and integrity of pharmaceutical and biotechnology products packaged in vials. Leak testing is a crucial step in the manufacturing process, preventing contamination and maintaining product efficacy. With stringent regulatory requirements and a focus on quality assurance, the vials leak testing market is integral to the pharmaceutical and biopharmaceutical industries.
Meaning
Vials leak testing refers to the process of detecting and evaluating leaks or defects in vials used for packaging pharmaceutical and biotechnology products. These leaks can compromise the sterility and integrity of the product, leading to contamination and potential harm to patients. Vials leak testing involves various methods, including visual inspection, vacuum decay, and helium leak detection, ensuring that vials meet the required quality standards.
Executive Summary
The vials leak testing market is a niche but essential segment within the broader pharmaceutical and biotechnology industries. As the demand for sterile and high-quality pharmaceutical products continues to rise, vials leak testing becomes increasingly vital. Stakeholders in the market need to navigate regulatory landscapes, adopt advanced testing technologies, and emphasize quality control to meet industry standards and ensure patient safety.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The vials leak testing market operates within dynamic dynamics shaped by factors such as regulatory changes, technological advancements, industry collaborations, and emerging trends in pharmaceutical manufacturing. Adapting to these dynamics is crucial for industry participants to maintain competitiveness and uphold product quality.
Regional Analysis
Regional variations in pharmaceutical manufacturing practices, regulatory frameworks, and industry standards influence the adoption of vials leak testing. Key regions include North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa. Understanding regional nuances is essential for companies seeking to navigate diverse markets.
Competitive Landscape
Leading companies in the Vials Leak Testing Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
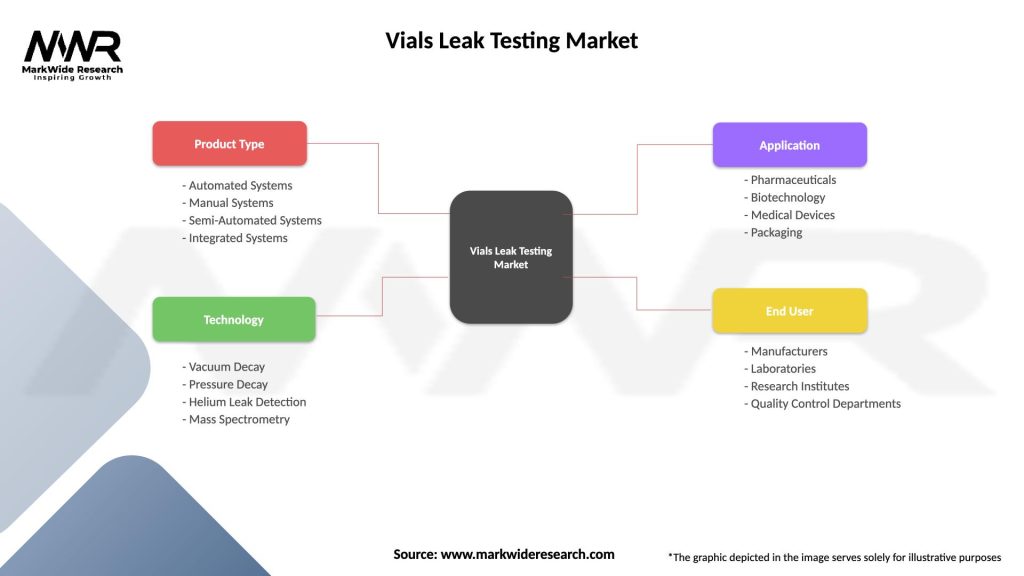
Segmentation
The vials leak testing market can be segmented based on factors such as the type of testing methods employed, the scale of manufacturing operations, and the types of pharmaceutical products being tested.
Type of Testing Methods:
Scale of Manufacturing Operations:
Types of Pharmaceutical Products:
Category-wise Insights:
Key Benefits for Industry Participants and Stakeholders:
SWOT Analysis:
Understanding these factors through a SWOT analysis allows companies to leverage strengths, address weaknesses, capitalize on opportunities, and mitigate potential threats in the vials leak testing market.
Market Key Trends:
Covid-19 Impact:
The COVID-19 pandemic has underscored the importance of robust quality control measures in pharmaceutical manufacturing. The need for vials leak testing remains crucial, especially in the production of vaccines and other essential pharmaceuticals. The pandemic has accelerated the adoption of advanced testing technologies to meet the increased demand for reliable and efficient leak testing processes.
Key Industry Developments:
Analyst Suggestions:
Future Outlook:
The future outlook for the vials leak testing market is positive, driven by the increasing emphasis on pharmaceutical quality control, technological advancements, and the rise of biopharmaceutical innovations. As pharmaceutical manufacturing becomes more sophisticated and global, vials leak testing will remain a critical step in ensuring the safety and efficacy of pharmaceutical products. The integration of cutting-edge technologies, collaboration for standardization, and a focus on sustainability will shape the market’s trajectory.
Conclusion:
In conclusion, the vials leak testing market occupies a vital role in pharmaceutical and biopharmaceutical manufacturing, ensuring the safety, integrity, and quality of products packaged in vials. With patient safety at the forefront, pharmaceutical companies are increasingly investing in advanced testing technologies to meet stringent regulatory requirements and maintain industry credibility. While facing challenges such as high initial costs and technical complexities, the market presents opportunities for innovation, collaboration, and sustainability initiatives. The future of the vials leak testing market is closely tied to advancements in Industry 4.0, cross-industry collaboration, and a commitment to quality assurance in pharmaceutical manufacturing.
What is Vials Leak Testing?
Vials leak testing refers to the process of evaluating the integrity of vials to ensure they are sealed properly and do not allow the escape of contents or the entry of contaminants. This is crucial in industries such as pharmaceuticals and biotechnology, where product safety and efficacy are paramount.
What are the key players in the Vials Leak Testing Market?
Key players in the Vials Leak Testing Market include companies like HPT, Inc., DÜRR NDT GmbH, and Seidenader Maschinenbau GmbH, which specialize in leak testing technologies and solutions for vials and other containers. These companies focus on providing innovative testing methods to enhance product reliability, among others.
What are the main drivers of the Vials Leak Testing Market?
The main drivers of the Vials Leak Testing Market include the increasing demand for high-quality pharmaceutical products, stringent regulatory requirements for packaging integrity, and advancements in leak testing technologies. These factors contribute to the growing emphasis on ensuring product safety and compliance in the industry.
What challenges does the Vials Leak Testing Market face?
Challenges in the Vials Leak Testing Market include the high costs associated with advanced testing equipment and the need for skilled personnel to operate these systems. Additionally, variations in vial materials and designs can complicate the testing process, impacting efficiency.
What opportunities exist in the Vials Leak Testing Market?
Opportunities in the Vials Leak Testing Market include the development of automated testing solutions and the integration of artificial intelligence for improved accuracy and efficiency. As the pharmaceutical industry continues to grow, there is a rising need for reliable leak testing methods to ensure product integrity.
What trends are shaping the Vials Leak Testing Market?
Trends in the Vials Leak Testing Market include the increasing adoption of non-destructive testing methods and the use of advanced technologies such as machine learning for data analysis. Additionally, there is a growing focus on sustainability, with companies seeking eco-friendly testing solutions.
Vials Leak Testing Market
| Segmentation Details | Description |
|---|---|
| Product Type | Automated Systems, Manual Systems, Semi-Automated Systems, Integrated Systems |
| Technology | Vacuum Decay, Pressure Decay, Helium Leak Detection, Mass Spectrometry |
| Application | Pharmaceuticals, Biotechnology, Medical Devices, Packaging |
| End User | Manufacturers, Laboratories, Research Institutes, Quality Control Departments |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading companies in the Vials Leak Testing Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at