444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at

Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$2450
The United States melanoma diagnostics and therapeutics market represents a critical segment of the oncology healthcare landscape, addressing one of the most aggressive forms of skin cancer. Melanoma diagnostics encompass advanced imaging technologies, molecular testing, and biomarker analysis, while therapeutics include immunotherapy, targeted therapy, and combination treatments. The market demonstrates robust growth driven by increasing melanoma incidence rates, technological advancements in diagnostic tools, and breakthrough therapeutic innovations.
Market dynamics indicate significant expansion potential, with the sector experiencing 8.2% CAGR growth attributed to enhanced early detection capabilities and personalized treatment approaches. The integration of artificial intelligence in diagnostic imaging and the development of novel immunotherapeutic agents continue to reshape treatment paradigms. Healthcare providers increasingly adopt comprehensive diagnostic platforms that combine dermoscopy, confocal microscopy, and genetic testing to improve diagnostic accuracy and patient outcomes.
Regional distribution shows concentrated market activity in major metropolitan areas with advanced cancer centers, while telemedicine and portable diagnostic solutions expand access to underserved populations. The market benefits from strong research infrastructure, favorable reimbursement policies, and collaborative efforts between pharmaceutical companies, diagnostic manufacturers, and healthcare institutions.
The United States melanoma diagnostics and therapeutics market refers to the comprehensive ecosystem of medical technologies, pharmaceutical products, and healthcare services dedicated to the detection, diagnosis, treatment, and management of melanoma skin cancer within the United States healthcare system. This market encompasses diagnostic tools ranging from traditional dermoscopy to advanced molecular profiling, alongside therapeutic interventions including immunotherapy, targeted therapy, surgical procedures, and supportive care solutions.
Diagnostic components include imaging technologies, biopsy procedures, histopathological analysis, genetic testing, and biomarker identification systems that enable healthcare professionals to accurately identify melanoma at various stages. Therapeutic elements comprise pharmaceutical treatments, medical devices, surgical instruments, and comprehensive care protocols designed to treat melanoma patients effectively while minimizing adverse effects and improving quality of life.
Market leadership in the United States melanoma diagnostics and therapeutics sector is characterized by rapid technological advancement and significant therapeutic breakthroughs. The market demonstrates exceptional growth momentum, with diagnostic segment expansion driven by AI-powered imaging solutions and molecular testing capabilities, while the therapeutics segment benefits from immunotherapy innovations and personalized treatment approaches.
Key market drivers include rising melanoma incidence rates, increasing awareness of early detection benefits, and substantial investments in research and development. The market shows 65% adoption rate for advanced diagnostic technologies among major cancer centers, indicating strong acceptance of innovative solutions. Therapeutic advancements have resulted in improved survival rates and enhanced patient outcomes, with combination therapies showing particular promise.
Competitive landscape features established pharmaceutical giants, emerging biotechnology companies, and specialized diagnostic manufacturers competing through innovation, strategic partnerships, and comprehensive product portfolios. The market benefits from robust regulatory support, favorable reimbursement frameworks, and strong collaboration between industry stakeholders and healthcare providers.
Strategic market insights reveal several critical factors shaping the United States melanoma diagnostics and therapeutics landscape:
Primary market drivers propelling growth in the United States melanoma diagnostics and therapeutics market include several interconnected factors that create sustained demand for innovative solutions.
Increasing incidence rates represent the fundamental driver, with melanoma cases showing consistent upward trends attributed to aging populations, lifestyle factors, and environmental exposures. Early detection initiatives supported by healthcare organizations and government agencies drive demand for advanced diagnostic technologies and screening programs.
Technological advancement in diagnostic imaging, molecular testing, and artificial intelligence creates opportunities for improved accuracy and efficiency in melanoma detection. Therapeutic innovation through immunotherapy development, targeted therapy research, and combination treatment protocols enhances treatment outcomes and expands market potential.
Market constraints in the United States melanoma diagnostics and therapeutics sector present challenges that industry participants must navigate to achieve sustainable growth and market penetration.
High treatment costs associated with advanced immunotherapies and targeted treatments create accessibility barriers for certain patient populations, despite insurance coverage. Diagnostic complexity requires specialized training and expertise, limiting adoption in smaller healthcare facilities and rural areas.
Regulatory challenges include lengthy approval processes for new diagnostic technologies and therapeutic agents, potentially delaying market entry and increasing development costs. Reimbursement limitations for certain diagnostic procedures and experimental treatments may restrict patient access and market growth.
Emerging opportunities in the United States melanoma diagnostics and therapeutics market present significant potential for growth and innovation across multiple segments and applications.
Artificial intelligence integration offers transformative potential for diagnostic accuracy improvement and workflow optimization. Telemedicine expansion creates opportunities to reach underserved populations and provide specialized expertise in remote locations. Combination therapy development presents possibilities for enhanced treatment efficacy and improved patient outcomes.
Personalized medicine advancement through genetic profiling and biomarker analysis enables tailored treatment approaches with higher success rates. Preventive care focus drives demand for advanced screening technologies and risk assessment tools.
Market dynamics in the United States melanoma diagnostics and therapeutics sector reflect complex interactions between technological innovation, regulatory frameworks, healthcare policies, and patient needs that shape industry evolution.
Innovation cycles demonstrate accelerating pace of development, with breakthrough therapies and diagnostic technologies entering the market at unprecedented rates. Competitive pressures drive continuous improvement in product efficacy, safety profiles, and cost-effectiveness. The market shows 75% preference for combination diagnostic approaches among oncologists, indicating strong adoption of comprehensive testing strategies.
Healthcare policy changes influence reimbursement structures, regulatory requirements, and market access pathways. Patient advocacy groups play increasingly important roles in driving awareness, research funding, and policy development. MarkWide Research analysis indicates that market dynamics favor companies with strong research capabilities and comprehensive product portfolios.
Supply chain considerations include manufacturing capacity, distribution networks, and quality assurance systems that ensure reliable product availability. Partnership strategies between pharmaceutical companies, diagnostic manufacturers, and healthcare providers create synergistic opportunities for market expansion and improved patient care.
Comprehensive research methodology employed in analyzing the United States melanoma diagnostics and therapeutics market incorporates multiple data sources, analytical frameworks, and validation processes to ensure accuracy and reliability of market insights.
Primary research includes extensive interviews with key industry stakeholders, including pharmaceutical executives, diagnostic company leaders, healthcare providers, and clinical researchers. Secondary research encompasses analysis of published studies, regulatory filings, clinical trial data, and industry reports from authoritative sources.
Data collection methods utilize both quantitative and qualitative approaches, incorporating statistical analysis, trend identification, and expert opinion synthesis. Market modeling employs sophisticated analytical tools to project growth patterns, identify emerging trends, and assess competitive dynamics.
Regional market distribution across the United States reveals distinct patterns of market concentration, growth opportunities, and healthcare infrastructure development that influence melanoma diagnostics and therapeutics adoption.
Northeast region demonstrates the highest market concentration with 35% market share, driven by prestigious cancer centers, research institutions, and high population density. Major metropolitan areas including Boston, New York, and Philadelphia serve as innovation hubs with advanced diagnostic capabilities and cutting-edge therapeutic options.
West Coast markets show strong growth momentum with 28% market share, particularly in California, where biotechnology clusters and academic medical centers drive innovation and adoption. Southern states represent emerging opportunities with 22% market share, supported by expanding healthcare infrastructure and increasing awareness programs.
Midwest region accounts for 15% market share, with growth driven by major medical centers in Chicago, Cleveland, and Minneapolis. Rural market penetration remains challenging but shows improvement through telemedicine initiatives and mobile diagnostic programs that extend specialized care to underserved populations.
Competitive dynamics in the United States melanoma diagnostics and therapeutics market feature a diverse ecosystem of established pharmaceutical companies, emerging biotechnology firms, and specialized diagnostic manufacturers competing through innovation and strategic positioning.
Market leaders in the therapeutic segment include:
Diagnostic market leaders include:
Market segmentation analysis reveals distinct categories within the United States melanoma diagnostics and therapeutics market, each characterized by unique growth drivers, competitive dynamics, and development opportunities.
By Diagnostic Type:
By Therapeutic Approach:
By End User:
Diagnostic category analysis reveals significant growth potential across multiple technology segments, with artificial intelligence and molecular testing showing particularly strong momentum in the United States melanoma diagnostics market.
AI-powered imaging demonstrates exceptional growth with 85% accuracy improvement in early-stage melanoma detection compared to traditional methods. Molecular diagnostics show increasing adoption rates, particularly for genetic profiling and biomarker identification that guide therapeutic decision-making.
Therapeutic category insights indicate immunotherapy dominance, with checkpoint inhibitors representing the largest segment by treatment volume and revenue generation. Combination therapies emerge as the fastest-growing category, offering enhanced efficacy through synergistic treatment approaches.
Industry participants in the United States melanoma diagnostics and therapeutics market realize substantial benefits through strategic positioning, technological innovation, and collaborative partnerships that enhance competitive advantage and market presence.
Pharmaceutical companies benefit from robust market demand, premium pricing opportunities for innovative therapies, and strong intellectual property protection. Diagnostic manufacturers capitalize on increasing adoption of advanced testing technologies and growing emphasis on personalized medicine approaches.
Healthcare providers gain access to cutting-edge diagnostic tools and therapeutic options that improve patient outcomes and enhance clinical reputation. Patients benefit from improved survival rates, reduced side effects, and enhanced quality of life through advanced treatment options.
Strengths:
Weaknesses:
Opportunities:
Threats:
Emerging trends in the United States melanoma diagnostics and therapeutics market reflect evolving healthcare paradigms, technological capabilities, and patient expectations that shape industry development and competitive strategies.
Artificial intelligence integration represents the most significant trend, with machine learning algorithms enhancing diagnostic accuracy and enabling predictive analytics for treatment outcomes. Personalized medicine continues gaining momentum through genetic profiling, biomarker analysis, and tailored treatment protocols.
Telemedicine adoption accelerates market accessibility, particularly for rural populations and follow-up care management. MWR data indicates that 70% of healthcare providers plan to expand telemedicine capabilities for oncology services within the next two years.
Recent industry developments demonstrate the dynamic nature of the United States melanoma diagnostics and therapeutics market, with breakthrough innovations, strategic partnerships, and regulatory approvals driving market evolution.
FDA approvals for novel immunotherapy combinations and targeted therapy agents expand treatment options and improve patient outcomes. Diagnostic technology advances include AI-powered imaging systems and liquid biopsy platforms that enhance detection capabilities and treatment monitoring.
Strategic partnerships between pharmaceutical companies, diagnostic manufacturers, and healthcare providers create synergistic opportunities for comprehensive patient care solutions. Research collaborations with academic institutions accelerate innovation and clinical development programs.
Strategic recommendations for stakeholders in the United States melanoma diagnostics and therapeutics market focus on leveraging emerging opportunities while addressing market challenges through innovation and strategic positioning.
Investment priorities should emphasize artificial intelligence integration, combination therapy development, and telemedicine capabilities that enhance market competitiveness. Partnership strategies with complementary companies can accelerate innovation and expand market reach through synergistic collaborations.
MarkWide Research analysis suggests that companies focusing on personalized medicine approaches and comprehensive diagnostic platforms will achieve superior market positioning. Regulatory strategy development should prioritize breakthrough therapy designations and expedited approval pathways for innovative solutions.
Future market prospects for the United States melanoma diagnostics and therapeutics sector indicate sustained growth driven by technological innovation, demographic trends, and evolving healthcare paradigms that create long-term opportunities for industry participants.
Technology advancement will continue reshaping market dynamics through AI integration, precision medicine development, and novel therapeutic mechanisms. Market expansion is projected to maintain strong growth trajectory supported by increasing melanoma incidence, aging populations, and enhanced awareness programs.
Innovation focus will emphasize combination therapies, companion diagnostics, and personalized treatment approaches that improve patient outcomes while optimizing healthcare resource utilization. Digital health integration will expand market accessibility and enhance patient engagement through mobile technologies and remote monitoring capabilities.
Regulatory evolution will likely support innovation through streamlined approval processes and adaptive trial designs that accelerate market entry for breakthrough technologies. Healthcare policy developments may enhance reimbursement coverage and patient access to advanced diagnostic and therapeutic solutions.
The United States melanoma diagnostics and therapeutics market represents a dynamic and rapidly evolving sector characterized by significant innovation, robust growth potential, and substantial opportunities for industry participants. Market fundamentals remain strong, supported by increasing disease incidence, technological advancement, and favorable regulatory environments that encourage innovation and investment.
Key success factors include technological leadership, comprehensive product portfolios, strategic partnerships, and patient-centric approaches that address evolving healthcare needs. Future growth will be driven by artificial intelligence integration, personalized medicine development, and expanded market access through telemedicine and digital health solutions.
Industry stakeholders who embrace innovation, invest in emerging technologies, and develop collaborative partnerships will be best positioned to capitalize on market opportunities and achieve sustainable competitive advantage in this critical healthcare sector. The market’s trajectory toward improved patient outcomes and enhanced treatment efficacy ensures continued growth and development opportunities for committed industry participants.
What is Melanoma Diagnostics And Therapeutics?
Melanoma Diagnostics And Therapeutics refers to the methods and treatments used to detect and manage melanoma, a type of skin cancer. This includes diagnostic techniques such as biopsies and imaging, as well as therapeutic options like immunotherapy and targeted therapy.
What are the key players in the United States Melanoma Diagnostics And Therapeutics Market?
Key players in the United States Melanoma Diagnostics And Therapeutics Market include Bristol-Myers Squibb, Merck & Co., Amgen, and Novartis, among others. These companies are involved in developing innovative therapies and diagnostic tools for melanoma treatment.
What are the growth factors driving the United States Melanoma Diagnostics And Therapeutics Market?
The growth of the United States Melanoma Diagnostics And Therapeutics Market is driven by increasing melanoma incidence rates, advancements in diagnostic technologies, and the rising adoption of targeted therapies. Additionally, heightened awareness about skin cancer and preventive measures contribute to market expansion.
What challenges does the United States Melanoma Diagnostics And Therapeutics Market face?
Challenges in the United States Melanoma Diagnostics And Therapeutics Market include high treatment costs, regulatory hurdles, and the complexity of melanoma’s biological behavior. These factors can hinder patient access to timely diagnostics and effective therapies.
What opportunities exist in the United States Melanoma Diagnostics And Therapeutics Market?
Opportunities in the United States Melanoma Diagnostics And Therapeutics Market include the development of personalized medicine approaches, the integration of artificial intelligence in diagnostics, and the expansion of telemedicine for patient management. These innovations can enhance treatment outcomes and accessibility.
What trends are shaping the United States Melanoma Diagnostics And Therapeutics Market?
Trends shaping the United States Melanoma Diagnostics And Therapeutics Market include the increasing use of combination therapies, advancements in genetic testing for melanoma, and a focus on patient-centric care. These trends are influencing how melanoma is diagnosed and treated.
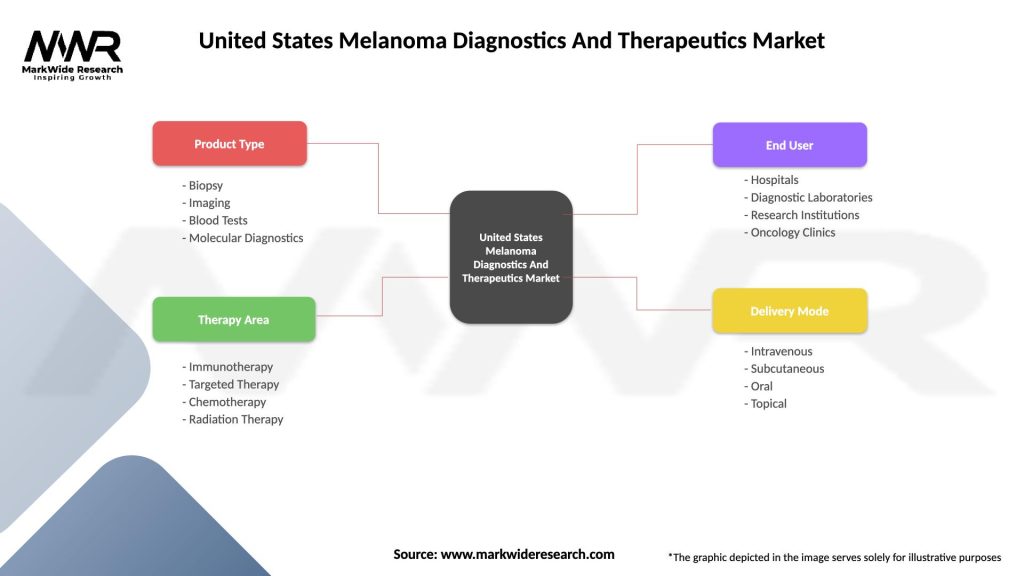
United States Melanoma Diagnostics And Therapeutics Market
| Segmentation Details | Description |
|---|---|
| Product Type | Biopsy, Imaging, Blood Tests, Molecular Diagnostics |
| Therapy Area | Immunotherapy, Targeted Therapy, Chemotherapy, Radiation Therapy |
| End User | Hospitals, Diagnostic Laboratories, Research Institutions, Oncology Clinics |
| Delivery Mode | Intravenous, Subcutaneous, Oral, Topical |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading companies in the United States Melanoma Diagnostics And Therapeutics Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at