444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The Tygacil market is an integral component of the pharmaceutical industry, focused on providing effective treatment options for bacterial infections. Tygacil, a broad-spectrum antibiotic, plays a crucial role in combating multidrug-resistant pathogens and addressing unmet medical needs in infectious diseases. With its unique mechanism of action and proven clinical efficacy, Tygacil has become a cornerstone therapy in the management of complicated intra-abdominal infections (cIAI) and complicated skin and skin structure infections (cSSSI).
Meaning
Tygacil, also known by its generic name tigecycline, is a glycylcycline antibiotic that inhibits bacterial protein synthesis by binding to the 30S ribosomal subunit, thereby exerting bacteriostatic activity against a broad spectrum of Gram-positive and Gram-negative pathogens, including multidrug-resistant strains such as methicillin-resistant Staphylococcus aureus (MRSA), extended-spectrum β-lactamase (ESBL)-producing Enterobacteriaceae, and Acinetobacter baumannii.
Executive Summary
The Tygacil market is driven by the increasing incidence of antibiotic-resistant infections, rising demand for novel antimicrobial agents, and the expanding global burden of infectious diseases. Tygacil’s unique pharmacological profile, favorable safety profile, and broad-spectrum activity position it as a valuable therapeutic option for healthcare providers managing complex bacterial infections. However, challenges such as antimicrobial resistance, regulatory constraints, and competition from generic alternatives necessitate strategic initiatives to sustain market growth and enhance patient access to Tygacil therapy.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The Tygacil market operates within a dynamic landscape shaped by factors such as antimicrobial resistance trends, regulatory developments, clinical practice guidelines, healthcare economics, and patient preferences. Market dynamics influence prescribing patterns, product positioning, pricing strategies, and market access initiatives for Tygacil and competing antibiotics in the infectious disease therapeutics market.
Regional Analysis
Regional variations in antimicrobial resistance patterns, healthcare infrastructure, regulatory frameworks, and market dynamics impact the adoption and utilization of Tygacil across different geographic regions. Key markets for Tygacil include North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa, each presenting unique opportunities and challenges for market stakeholders.
Competitive Landscape
Leading Companies in Tygacil Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
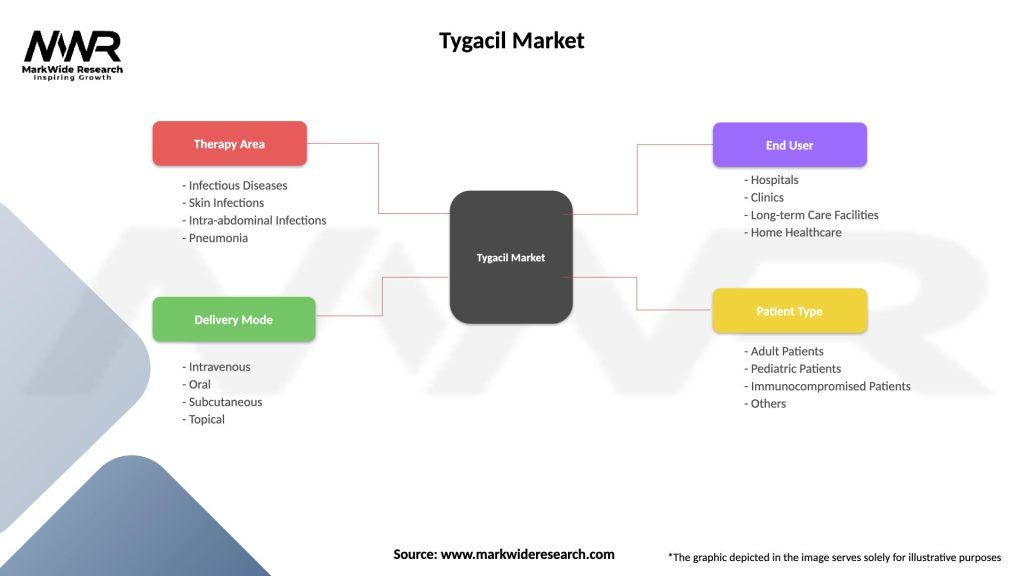
Segmentation
The Tygacil market can be segmented based on factors such as therapeutic indication, patient population, geographic region, and distribution channel, providing insights into market trends, treatment preferences, and growth opportunities across different segments.
Category-wise Insights
Tygacil falls within the category of glycylcycline antibiotics, a subclass of tetracyclines, characterized by a broader spectrum of activity, enhanced potency against resistant pathogens, and favorable pharmacokinetic properties compared to traditional tetracyclines. As a novel antibiotic class, glycylcyclines offer therapeutic advantages in the treatment of complicated infections and multidrug-resistant organisms, driving market demand for Tygacil and related agents.
Key Benefits for Industry Participants and Stakeholders
The Tygacil market offers several benefits for industry participants and stakeholders, including pharmaceutical companies, healthcare providers, patients, and regulatory agencies:
SWOT Analysis
A SWOT analysis of the Tygacil market provides insights into its strengths, weaknesses, opportunities, and threats:
Market Key Trends
Key trends shaping the Tygacil market include:
Covid-19 Impact
The COVID-19 pandemic has underscored the importance of effective antibiotics in managing secondary bacterial infections and sepsis associated with severe COVID-19 cases, highlighting the continued relevance of antibiotics like Tygacil in infectious disease management amidst public health crises. While the pandemic disrupted healthcare systems, clinical trials, and supply chains, it also accelerated digital health adoption, telemedicine utilization, and infectious disease research, shaping the future of antibiotic development and antimicrobial stewardship in the post-pandemic era.
Key Industry Developments
Key industry developments in the Tygacil market include:
Analyst Suggestions
Analyst suggestions for industry stakeholders in the Tygacil market include:
Future Outlook
The Tygacil market is poised for continued growth and innovation, driven by factors such as antimicrobial resistance, infectious disease burden, regulatory support, and market dynamics. Market opportunities include therapeutic expansion, pipeline development, and strategic partnerships, shaping the future of antibiotics and infectious disease management worldwide.
Conclusion
In conclusion, the Tygacil market represents a dynamic and evolving sector within the pharmaceutical industry, offering valuable therapeutic options for complicated bacterial infections and multidrug-resistant pathogens. Despite challenges such as antimicrobial resistance, regulatory constraints, and generic competition, Tygacil’s clinical efficacy, safety profile, and broad-spectrum activity position it as a cornerstone therapy in infectious disease management, contributing to improved patient outcomes, public health preparedness, and antimicrobial stewardship efforts globally. By embracing therapeutic innovation, market differentiation, and strategic collaboration, stakeholders can navigate market complexities, address unmet medical needs, and drive positive outcomes in infectious diseases and antibiotic therapy.
What is Tygacil?
Tygacil is an antibiotic medication used to treat various bacterial infections, particularly those caused by resistant strains. It is often utilized in cases of complicated skin and soft tissue infections, as well as intra-abdominal infections.
What are the key companies in the Tygacil Market?
Key companies in the Tygacil Market include Wyeth Pharmaceuticals, Pfizer, and Astellas Pharma, among others. These companies are involved in the development and distribution of Tygacil and similar antibiotic therapies.
What are the growth factors driving the Tygacil Market?
The Tygacil Market is driven by the increasing prevalence of antibiotic-resistant infections and the rising demand for effective treatment options. Additionally, the growing awareness of healthcare-associated infections contributes to market growth.
What challenges does the Tygacil Market face?
The Tygacil Market faces challenges such as the potential for adverse side effects and the emergence of new resistant bacterial strains. Furthermore, competition from alternative antibiotics can impact market share.
What opportunities exist in the Tygacil Market?
Opportunities in the Tygacil Market include the development of new formulations and combination therapies that enhance efficacy. Additionally, expanding into emerging markets presents growth potential for Tygacil.
What trends are shaping the Tygacil Market?
Trends in the Tygacil Market include a focus on personalized medicine and the use of advanced diagnostic tools to identify resistant infections. There is also an increasing emphasis on stewardship programs to optimize antibiotic use.
Tygacil Market
| Segmentation Details | Description |
|---|---|
| Therapy Area | Infectious Diseases, Skin Infections, Intra-abdominal Infections, Pneumonia |
| Delivery Mode | Intravenous, Oral, Subcutaneous, Topical |
| End User | Hospitals, Clinics, Long-term Care Facilities, Home Healthcare |
| Patient Type | Adult Patients, Pediatric Patients, Immunocompromised Patients, Others |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in Tygacil Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at