444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The recombinant vaccines market is experiencing significant growth due to the increasing prevalence of infectious diseases and the rising demand for advanced vaccination strategies. Recombinant vaccines are produced by inserting the genetic material of a pathogen into a host organism, such as bacteria or yeast, to stimulate an immune response without causing the disease. These vaccines offer several advantages over traditional vaccines, including improved safety and efficacy.
Meaning
Recombinant vaccines are a type of genetically engineered vaccines that utilize recombinant DNA technology. This technology involves the insertion of specific genes from a pathogen into a host organism’s DNA. The host organism then produces the antigens of the pathogen, triggering an immune response when administered as a vaccine. Recombinant vaccines have revolutionized the field of vaccinology by enabling the development of safer and more effective vaccines.
Executive Summary
The global recombinant vaccines market is witnessing robust growth, driven by the increasing focus on preventive healthcare and the need for innovative vaccination approaches. With the advancements in genetic engineering and biotechnology, recombinant vaccines have emerged as a promising solution to combat infectious diseases. This report provides an in-depth analysis of the market, including key trends, drivers, restraints, opportunities, and regional insights.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
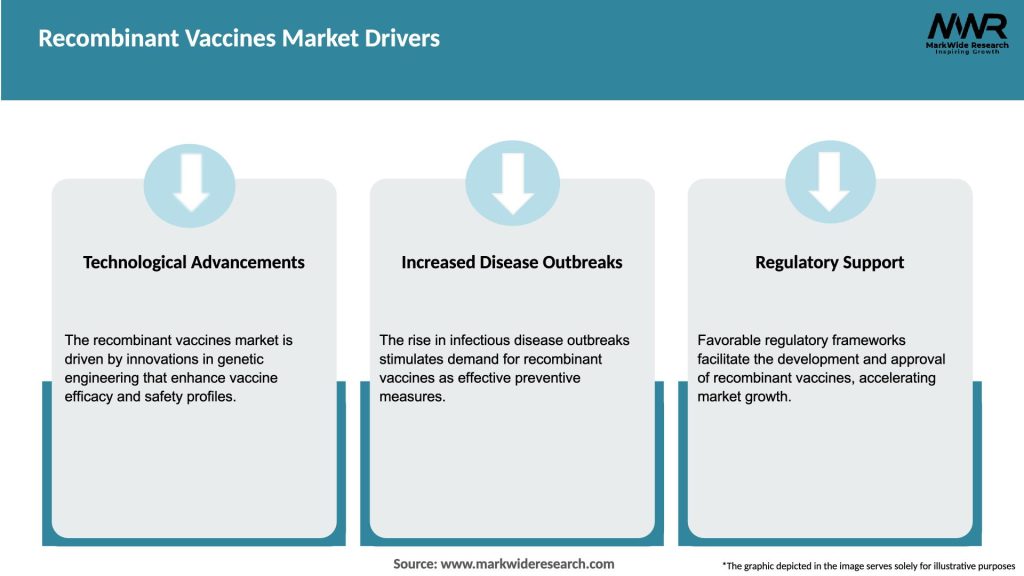
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The recombinant vaccines market is characterized by intense competition and rapid technological advancements. Market players are focusing on research and development activities to launch new and improved vaccines. Strategic collaborations and partnerships with academic institutions and government bodies are also common strategies to enhance product portfolios and geographical reach.
Regional Analysis
Competitive Landscape
Leading Companies in the Recombinant Vaccines Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
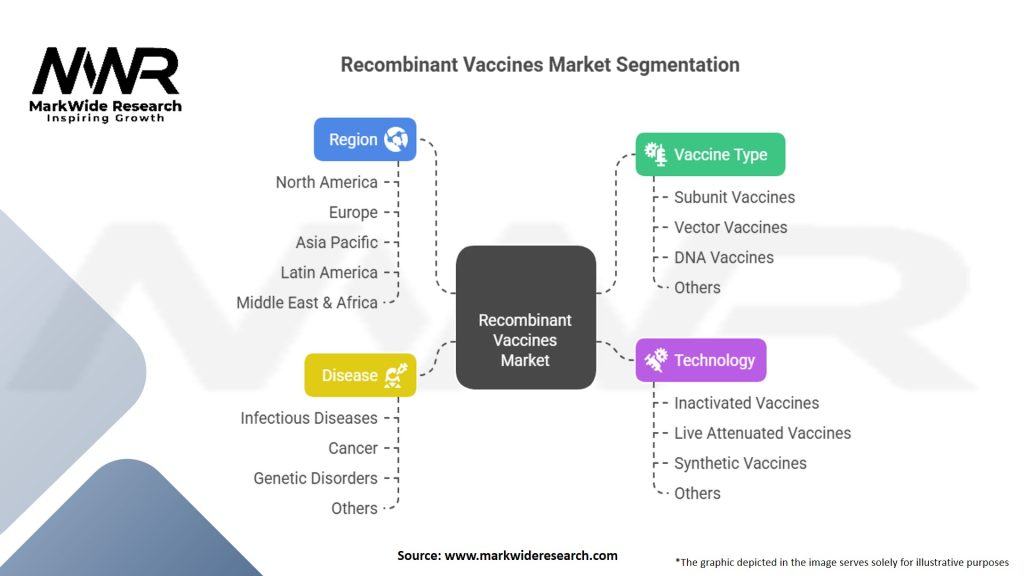
Segmentation
The recombinant vaccines market can be segmented based on technology, disease indication, and end-user.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Market Key Trends
Covid-19 Impact
The COVID-19 pandemic has had a significant impact on the recombinant vaccines market. The urgent need for an effective vaccine against SARS-CoV-2 has accelerated vaccine research and development efforts worldwide. Several recombinant COVID-19 vaccines have been authorized for emergency use, showcasing the potential of recombinant vaccine technology in responding to emerging infectious diseases. The pandemic has also increased public awareness about the importance of vaccination and boosted the overall demand for vaccines.
Key Industry Developments
Analyst Suggestions
Future Outlook
The future of the recombinant vaccines market looks promising, with substantial growth opportunities on the horizon. The increasing prevalence of infectious diseases, technological advancements in genetic engineering, and expanding immunization programs will drive market expansion. Personalized vaccines, nanotechnology in vaccine delivery, and combination vaccines are expected to be key areas of focus for future research and development. However, overcoming challenges such as high manufacturing costs, regulatory complexities, and vaccine hesitancy will be crucial for market success.
Conclusion
The recombinant vaccines market is experiencing significant growth and presents a promising solution to prevent the spread of infectious diseases. These vaccines, produced using genetic engineering techniques, offer improved safety, efficacy, and customization. The market is driven by factors such as the rising prevalence of infectious diseases, government initiatives, technological advancements, and growing awareness about vaccination benefits. However, challenges such as high manufacturing costs and regulatory requirements need to be addressed. The market’s future looks promising, with opportunities in emerging markets, technological innovations, and expanding vaccination programs. Strategic investments in research and development, collaborations, and addressing vaccine hesitancy will be key to unlocking the full potential of the recombinant vaccines market.
What is Recombinant Vaccines?
Recombinant vaccines are a type of vaccine created by inserting genetic material from a pathogen into a host cell, which then produces antigens that stimulate an immune response. These vaccines are used to prevent various infectious diseases by training the immune system to recognize and combat specific pathogens.
What are the key players in the Recombinant Vaccines Market?
Key players in the Recombinant Vaccines Market include companies such as Merck & Co., Sanofi Pasteur, GlaxoSmithKline, and Pfizer, among others. These companies are involved in the research, development, and production of recombinant vaccines for various diseases.
What are the growth factors driving the Recombinant Vaccines Market?
The growth of the Recombinant Vaccines Market is driven by factors such as the increasing prevalence of infectious diseases, advancements in genetic engineering technologies, and the rising demand for effective vaccination programs. Additionally, the focus on preventive healthcare is contributing to market expansion.
What challenges does the Recombinant Vaccines Market face?
The Recombinant Vaccines Market faces challenges such as high development costs, regulatory hurdles, and public skepticism regarding vaccine safety and efficacy. These factors can hinder the timely introduction of new vaccines into the market.
What opportunities exist in the Recombinant Vaccines Market?
Opportunities in the Recombinant Vaccines Market include the potential for developing vaccines for emerging infectious diseases and the expansion of vaccination programs in developing countries. Additionally, collaborations between public and private sectors can enhance research and development efforts.
What trends are shaping the Recombinant Vaccines Market?
Trends shaping the Recombinant Vaccines Market include the increasing use of mRNA technology, the development of combination vaccines, and a growing emphasis on personalized medicine. These trends are expected to enhance vaccine efficacy and broaden their applications.
Recombinant Vaccines Market
| Segmentation | Details |
|---|---|
| Vaccine Type | Subunit Vaccines, Vector Vaccines, DNA Vaccines, Others |
| Technology | Inactivated Vaccines, Live Attenuated Vaccines, Synthetic Vaccines, Others |
| Disease | Infectious Diseases, Cancer, Genetic Disorders, Others |
| Region | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Recombinant Vaccines Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at