444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview: The Rare Hemophilia Factors Market is a crucial segment within the broader healthcare industry, addressing the specific needs of individuals with rare forms of hemophilia. Hemophilia is a genetic disorder characterized by the deficiency or absence of certain blood clotting factors, leading to prolonged bleeding and difficulty in clot formation. While hemophilia A and B are more common, the rare hemophilia factors market caters to patients with less prevalent types, such as hemophilia C.
Meaning: Rare hemophilia factors refer to specific blood clotting factors that are deficient or dysfunctional in individuals with less common types of hemophilia. These factors, including Factor V, Factor VII, Factor X, and Factor XIII, play a crucial role in the coagulation cascade. Understanding the unique nature of rare hemophilia factors is essential for healthcare providers, researchers, and pharmaceutical companies involved in developing targeted therapies for affected individuals.
Executive Summary: The Rare Hemophilia Factors Market presents both challenges and opportunities within the healthcare landscape. While these conditions affect a smaller patient population compared to hemophilia A and B, addressing the specific needs of individuals with rare hemophilia factors requires specialized medical interventions and therapeutic approaches. The market has witnessed advancements in treatment options, with a focus on personalized medicine and precision therapies to improve patient outcomes.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights:
Market Drivers:
Market Restraints:
Market Opportunities:
Market Dynamics: The dynamics of the Rare Hemophilia Factors Market are influenced by various factors, including scientific advancements, regulatory landscapes, patient advocacy efforts, and global market trends. The interplay of these dynamics shapes the market’s trajectory and the development of new therapeutic options for affected individuals.
Regional Analysis: The prevalence of rare hemophilia factors may vary regionally, influenced by genetic factors, healthcare infrastructure, and access to diagnostic capabilities. A regional analysis provides insights into the distribution of affected individuals and the corresponding healthcare needs. Let’s explore key regions:
Competitive Landscape:
Leading Companies in Rare Hemophilia Factors Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
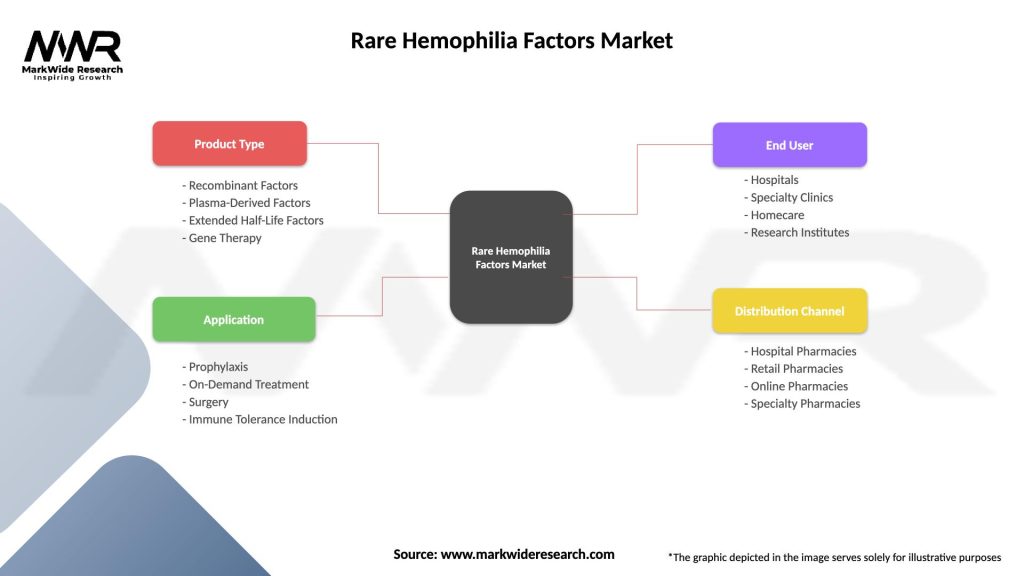
Segmentation: The Rare Hemophilia Factors Market can be segmented based on various factors, including:
Segmentation allows for a more nuanced understanding of the market dynamics, enabling healthcare providers and pharmaceutical companies to tailor their strategies to specific patient needs and market conditions.
Category-wise Insights:
Key Benefits for Industry Participants and Stakeholders:
SWOT Analysis: A SWOT analysis provides an overview of the Rare Hemophilia Factors Market’s strengths, weaknesses, opportunities, and threats:
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends:
Covid-19 Impact: The global Covid-19 pandemic has had notable effects on the Rare Hemophilia Factors Market:
Key Industry Developments:
Analyst Suggestions:
Future Outlook: The future outlook for the Rare Hemophilia Factors Market is characterized by a balance between challenges and opportunities. The market is poised for significant advancements, driven by ongoing research, technological innovations, and a growing emphasis on personalized medicine. Key factors shaping the future outlook include:
Conclusion: The Rare Hemophilia Factors Market stands at the intersection of medical innovation, patient care, and global collaboration. While the challenges associated with a limited patient pool and complex treatment decisions persist, the market is witnessing remarkable advancements that offer hope for individuals with rare hemophilia factors. The future holds the promise of transformative therapies, increased patient engagement, and a collective effort to address the unique needs of this underserved patient population. As the healthcare landscape evolves, stakeholders in the Rare Hemophilia Factors Market are poised to contribute significantly to improving the lives of those affected by these rare genetic conditions.
What is Rare Hemophilia Factors?
Rare Hemophilia Factors refer to specific proteins in the blood that are deficient or absent in individuals with rare types of hemophilia, such as Factor VII, Factor XI, and Factor XIII. These factors are crucial for proper blood clotting and managing bleeding episodes in affected patients.
What are the key players in the Rare Hemophilia Factors Market?
Key players in the Rare Hemophilia Factors Market include companies like Novo Nordisk, Baxter International, and BioMarin Pharmaceutical, which are known for their innovative therapies and products for hemophilia treatment, among others.
What are the growth factors driving the Rare Hemophilia Factors Market?
The Rare Hemophilia Factors Market is driven by factors such as the increasing prevalence of rare hemophilia types, advancements in biotechnology leading to new treatment options, and a growing awareness of hemophilia management among healthcare professionals and patients.
What challenges does the Rare Hemophilia Factors Market face?
Challenges in the Rare Hemophilia Factors Market include the high cost of treatment, limited availability of specific factor concentrates, and the need for ongoing research to develop effective therapies for rare hemophilia types.
What opportunities exist in the Rare Hemophilia Factors Market?
Opportunities in the Rare Hemophilia Factors Market include the potential for developing gene therapies, expanding access to treatment in underserved regions, and increasing collaboration between pharmaceutical companies and research institutions to innovate new solutions.
What trends are shaping the Rare Hemophilia Factors Market?
Trends in the Rare Hemophilia Factors Market include a shift towards personalized medicine, the development of long-acting factor products, and the integration of digital health technologies to improve patient monitoring and adherence to treatment.
Rare Hemophilia Factors Market
| Segmentation Details | Description |
|---|---|
| Product Type | Recombinant Factors, Plasma-Derived Factors, Extended Half-Life Factors, Gene Therapy |
| Application | Prophylaxis, On-Demand Treatment, Surgery, Immune Tolerance Induction |
| End User | Hospitals, Specialty Clinics, Homecare, Research Institutes |
| Distribution Channel | Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Specialty Pharmacies |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at