444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The printer for capsules market is witnessing substantial growth, fueled by the pharmaceutical industry’s demand for efficient and precise printing solutions to mark and identify capsules for quality assurance, compliance, and brand recognition purposes. Printers designed specifically for capsules offer pharmaceutical companies a reliable and customizable solution for printing essential information such as dosage, batch numbers, and expiration dates directly onto capsule surfaces with accuracy and consistency.
Meaning
A printer for capsules is a specialized printing device used in the pharmaceutical industry to mark or print essential information directly onto the surface of capsules. These printers employ advanced printing technologies such as inkjet or laser printing to apply text, symbols, or codes onto capsule shells, ensuring clear and legible markings for identification, tracking, and compliance with regulatory requirements.
Executive Summary
The printer for capsules market is experiencing rapid expansion, driven by the pharmaceutical sector’s need for precise and efficient printing solutions to enhance product safety, quality, and traceability. With printers for capsules offering pharmaceutical manufacturers customizable printing capabilities, high-speed operation, and compliance with industry standards and regulations, they play a crucial role in ensuring product integrity and brand recognition in the competitive pharmaceutical market.

Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The printer for capsules market is characterized by evolving regulatory landscapes, technological advancements, and competitive dynamics. Key market players are adapting to changing market trends, customer needs, and industry standards to maintain competitiveness, drive innovation, and sustain growth in the dynamic pharmaceutical manufacturing industry.
Regional Analysis
The printer for capsules market is geographically segmented into North America, Europe, Asia-Pacific, Latin America, and the Middle East and Africa. The North America region leads the global market, driven by a large pharmaceutical manufacturing sector, strict regulatory standards, and growing adoption of automated printing solutions for capsule identification and serialization. However, other regions such as Europe, Asia-Pacific, and Latin America also witness significant market growth, fueled by expanding pharmaceutical markets, increasing generic drug production, and rising demand for quality control and compliance solutions in emerging economies.
Competitive Landscape
Leading Companies in the Printer for Capsules Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Segmentation
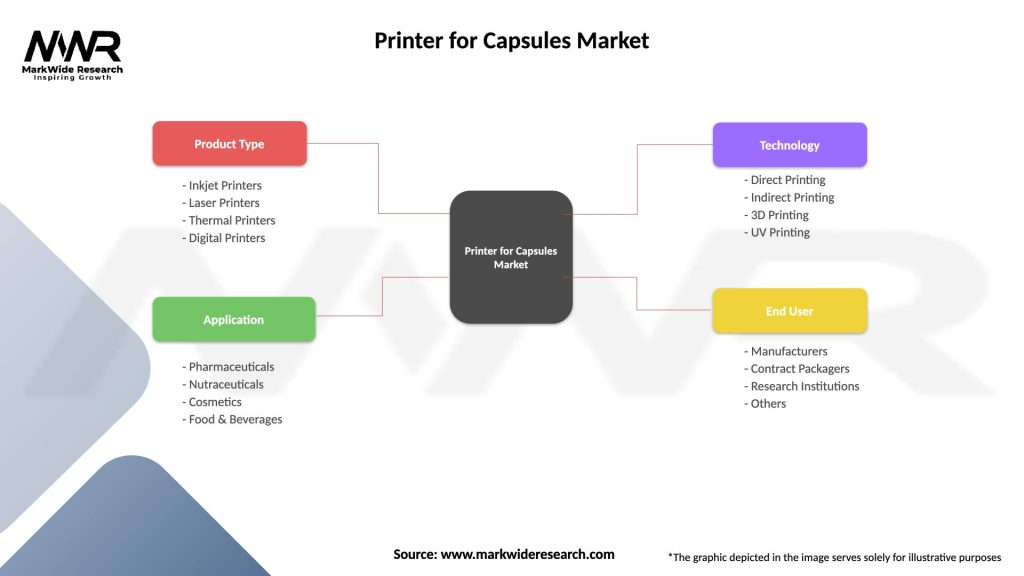
The printer for capsules market can be segmented based on printing technology, printing capacity, and geographic region. By printing technology, the market includes options such as inkjet printing, laser printing, thermal transfer printing, and digital printing, each offering different capabilities, speeds, and resolutions for printing text, symbols, or codes onto capsule surfaces. By printing capacity, the market comprises options such as high-speed printers, mid-range printers, and tabletop printers, each tailored to specific production volumes, throughput requirements, and space constraints in pharmaceutical manufacturing facilities.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Market Key Trends
Covid-19 Impact
The Covid-19 pandemic has influenced the printer for capsules market by accelerating digital transformation, automation initiatives, and supply chain resilience efforts in the pharmaceutical industry. As pharmaceutical companies prioritize operational efficiency, quality assurance, and compliance with regulatory requirements, there has been growing interest in printers for capsules that enable remote monitoring, real-time data analytics, and seamless integration with production systems to enhance agility, visibility, and control in pharmaceutical manufacturing operations.
Key Industry Developments
Analyst Suggestions
Future Outlook
The future outlook for the printer for capsules market is optimistic, with continued growth expected from increasing pharmaceutical production volumes, regulatory compliance requirements, and technological advancements in printing technologies and automation solutions. As pharmaceutical manufacturers continue to prioritize product safety, quality, and compliance, printers for capsules are poised to play a vital role in enabling pharmaceutical companies to meet regulatory requirements, enhance operational efficiency, and ensure patient safety and supply chain security in the dynamic and evolving pharmaceutical manufacturing industry.
Conclusion
In conclusion, the printer for capsules market is experiencing significant growth driven by the pharmaceutical industry’s demand for efficient, precise, and compliant printing solutions for capsule identification and serialization. With printers for capsules offering pharmaceutical manufacturers customizable printing capabilities, high-speed operation, and compliance with regulatory standards, they play a crucial role in ensuring product integrity, safety, and brand recognition in the competitive pharmaceutical market. By addressing market trends, customer needs, and regulatory requirements, printer manufacturers can capitalize on emerging opportunities and drive growth in the dynamic and evolving market for printers for capsules and other pharmaceutical printing solutions.
What is Printer for Capsules?
Printer for Capsules refers to specialized printing technology used to print on capsule surfaces, often utilized in the pharmaceutical and nutraceutical industries for branding and information purposes.
What are the key players in the Printer for Capsules Market?
Key players in the Printer for Capsules Market include companies like Videojet Technologies, Markem-Imaje, and Domino Printing Sciences, among others.
What are the main drivers of growth in the Printer for Capsules Market?
The growth of the Printer for Capsules Market is driven by the increasing demand for personalized medicine, the rise in the nutraceutical sector, and advancements in printing technology that enhance efficiency and quality.
What challenges does the Printer for Capsules Market face?
Challenges in the Printer for Capsules Market include stringent regulatory requirements, the need for high-quality printing standards, and competition from alternative labeling methods.
What opportunities exist in the Printer for Capsules Market?
Opportunities in the Printer for Capsules Market include the expansion of e-commerce in pharmaceuticals, the growing trend of customization in product packaging, and innovations in eco-friendly printing materials.
What trends are shaping the Printer for Capsules Market?
Trends in the Printer for Capsules Market include the adoption of digital printing technologies, increased focus on sustainability, and the integration of smart technologies for better tracking and traceability.
Printer for Capsules Market
| Segmentation Details | Description |
|---|---|
| Product Type | Inkjet Printers, Laser Printers, Thermal Printers, Digital Printers |
| Application | Pharmaceuticals, Nutraceuticals, Cosmetics, Food & Beverages |
| Technology | Direct Printing, Indirect Printing, 3D Printing, UV Printing |
| End User | Manufacturers, Contract Packagers, Research Institutions, Others |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Printer for Capsules Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at