444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The plasma protease C1-inhibitor market is witnessing significant growth due to the increasing prevalence of hereditary angioedema (HAE) and other related disorders. C1-inhibitor, a protein produced in the liver, plays a crucial role in regulating the activity of the complement system, a part of the immune system. Deficiency or malfunctioning of C1-inhibitor can lead to the overactivation of the complement system, resulting in HAE.
Meaning
Plasma protease C1-inhibitor refers to a therapeutic protein that is derived from human blood plasma. It is primarily used for the treatment of HAE, a rare genetic disorder characterized by recurrent episodes of swelling in various body parts, such as the face, hands, feet, or gastrointestinal tract. Plasma protease C1-inhibitor helps to regulate the activity of the complement system and prevent uncontrolled swelling attacks in HAE patients.
Executive Summary
The plasma protease C1-inhibitor market is experiencing steady growth, driven by the rising incidence of HAE and the increasing awareness and diagnosis of the condition. The market is witnessing advancements in therapeutic options, with the development of recombinant and subcutaneous formulations of plasma protease C1-inhibitor, offering improved convenience and ease of administration. The market is highly competitive, with several key players vying for market share through product development and strategic collaborations.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The plasma protease C1-inhibitor market is dynamic, driven by the interplay of various factors. The market is influenced by the prevalence and awareness of HAE, technological advancements, regulatory landscape, and competitive dynamics. The availability of reimbursement and the overall economic environment also impact market growth. Continuous research and development activities, along with strategic collaborations, contribute to the evolving market landscape.
Regional Analysis
The plasma protease C1-inhibitor market is segmented into several regions, including North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa. North America currently holds the largest market share, attributed to the high prevalence of HAE and well-established healthcare infrastructure. Europe follows closely, driven by increasing awareness and diagnosis rates. The Asia Pacific region presents significant growth potential due to the large population base and improving healthcare infrastructure.
Competitive Landscape
Leading Companies in the Plasma Protease C1-inhibitor Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
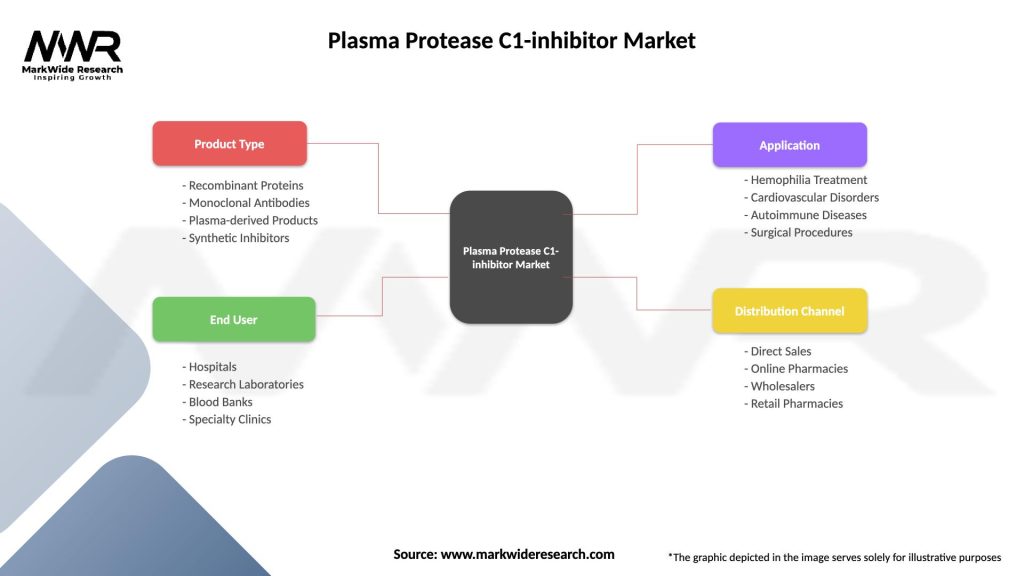
Segmentation
The plasma protease C1-inhibitor market can be segmented based on product type, application, and end-user.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Market Key Trends
Covid-19 Impact
The COVID-19 pandemic has had varying effects on the plasma protease C1-inhibitor market. On one hand, the healthcare system’s focus on managing the pandemic has led to delays in diagnosis and treatment initiation for non-emergent conditions like HAE. This has temporarily impacted the market growth. On the other hand, the pandemic has highlighted the importance of ensuring access to essential therapies, including plasma protease C1-inhibitor, for patients with chronic conditions. The industry has adapted by implementing telemedicine solutions and home delivery services to ensure continuity of care.
Key Industry Developments
Analyst Suggestions
Future Outlook
The plasma protease C1-inhibitor market is expected to grow at a steady pace in the coming years. Increasing awareness and diagnosis rates of HAE, along with technological advancements, will drive market expansion. The introduction of novel therapies and personalized treatment approaches will further enhance patient outcomes. However, challenges such as high treatment costs and stringent regulatory processes need to be addressed to ensure wider accessibility and market growth.
Conclusion
The plasma protease C1-inhibitor market is witnessing significant growth due to the rising prevalence of HAE and related disorders. Technological advancements have led to the development of more convenient and effective treatment options, improving patient outcomes. The market is highly competitive, with key players focusing on product development and strategic collaborations. Emerging markets and continuous research and development efforts present opportunities for future market expansion. However, addressing the high cost of treatment and increasing awareness in low-resource settings remain challenges that need to be overcome for sustained market growth.
What is Plasma Protease C1-inhibitor?
Plasma Protease C1-inhibitor is a protein that plays a crucial role in regulating the immune system and blood coagulation. It inhibits various proteases, thereby preventing excessive inflammation and clotting, making it essential in treating conditions like hereditary angioedema.
What are the key players in the Plasma Protease C1-inhibitor Market?
Key players in the Plasma Protease C1-inhibitor Market include CSL Behring, Shire (now part of Takeda), and Octapharma, among others. These companies are involved in the development and distribution of therapies that utilize C1-inhibitor for various medical conditions.
What are the growth factors driving the Plasma Protease C1-inhibitor Market?
The Plasma Protease C1-inhibitor Market is driven by the increasing prevalence of hereditary angioedema and rising awareness about treatment options. Additionally, advancements in biotechnology and the development of new therapies are contributing to market growth.
What challenges does the Plasma Protease C1-inhibitor Market face?
The Plasma Protease C1-inhibitor Market faces challenges such as high treatment costs and potential side effects associated with therapies. Regulatory hurdles and the need for extensive clinical trials can also impede market growth.
What opportunities exist in the Plasma Protease C1-inhibitor Market?
Opportunities in the Plasma Protease C1-inhibitor Market include the development of novel therapies and expanding applications in other inflammatory conditions. Additionally, increasing investment in research and development can lead to innovative treatment options.
What trends are shaping the Plasma Protease C1-inhibitor Market?
Trends in the Plasma Protease C1-inhibitor Market include a shift towards personalized medicine and the use of biologics. There is also a growing focus on patient-centric approaches and improving access to treatment for patients with rare diseases.
Plasma Protease C1-inhibitor Market
| Segmentation Details | Description |
|---|---|
| Product Type | Recombinant Proteins, Monoclonal Antibodies, Plasma-derived Products, Synthetic Inhibitors |
| End User | Hospitals, Research Laboratories, Blood Banks, Specialty Clinics |
| Application | Hemophilia Treatment, Cardiovascular Disorders, Autoimmune Diseases, Surgical Procedures |
| Distribution Channel | Direct Sales, Online Pharmacies, Wholesalers, Retail Pharmacies |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Plasma Protease C1-inhibitor Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at