444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The Pharma 4.0 market has emerged as a revolutionary approach in the pharmaceutical industry, combining advanced technologies with traditional manufacturing processes to enhance efficiency, productivity, and patient outcomes. This transformative concept leverages the power of digitalization, automation, and data analytics to optimize the entire pharmaceutical value chain, from drug discovery and development to manufacturing, distribution, and patient care.
Pharma 4.0 represents a paradigm shift from the traditional pharma industry model, integrating cutting-edge technologies such as artificial intelligence (AI), machine learning (ML), the Internet of Things (IoT), big data analytics, robotics, and 3D printing. By harnessing these technologies, Pharma 4.0 aims to streamline processes, improve quality control, reduce costs, increase operational agility, and enable personalized medicine.
Meaning
Pharma 4.0 refers to the integration of advanced technologies and digitalization in the pharmaceutical industry. It combines the principles of Industry 4.0 with specific applications tailored to the unique needs of the pharmaceutical sector. This approach enables the use of cutting-edge technologies such as artificial intelligence (AI), machine learning (ML), robotics, Internet of Things (IoT), and big data analytics to streamline manufacturing processes, enhance quality control, improve supply chain management, and optimize patient care.
Executive Summary
The Pharma 4.0 market is experiencing significant growth, driven by the increasing need for efficient and cost-effective pharmaceutical production, rising demand for personalized medicine, and the growing emphasis on quality and safety standards. The integration of digital technologies in the pharmaceutical industry has the potential to transform traditional manufacturing processes, enhance productivity, reduce time to market, and improve patient outcomes. This report provides a comprehensive analysis of the Pharma 4.0 market, highlighting key market insights, drivers, restraints, opportunities, and regional trends.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The Pharma 4.0 market is characterized by rapid technological advancements, increasing regulatory scrutiny, and changing market dynamics. Key trends include the convergence of AI, ML, and big data analytics in pharmaceutical applications, the rise of digital twins for process optimization, the adoption of robotics and automation in manufacturing, and the integration of cloud computing for data storage and collaboration.
Regional Analysis
The Pharma 4.0 market is witnessing significant growth across various regions. North America dominates the market due to the presence of established pharmaceutical companies, advanced healthcare infrastructure, and favorable government initiatives to promote digital transformation. Europe is also a key market, driven by the emphasis on quality and safety standards. The Asia Pacific region is expected to experience rapid growth, fueled by the expanding pharmaceutical sector, rising healthcare expenditure, and increasing focus on technology adoption.
Competitive Landscape
Leading Companies in Pharma 4.0 Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
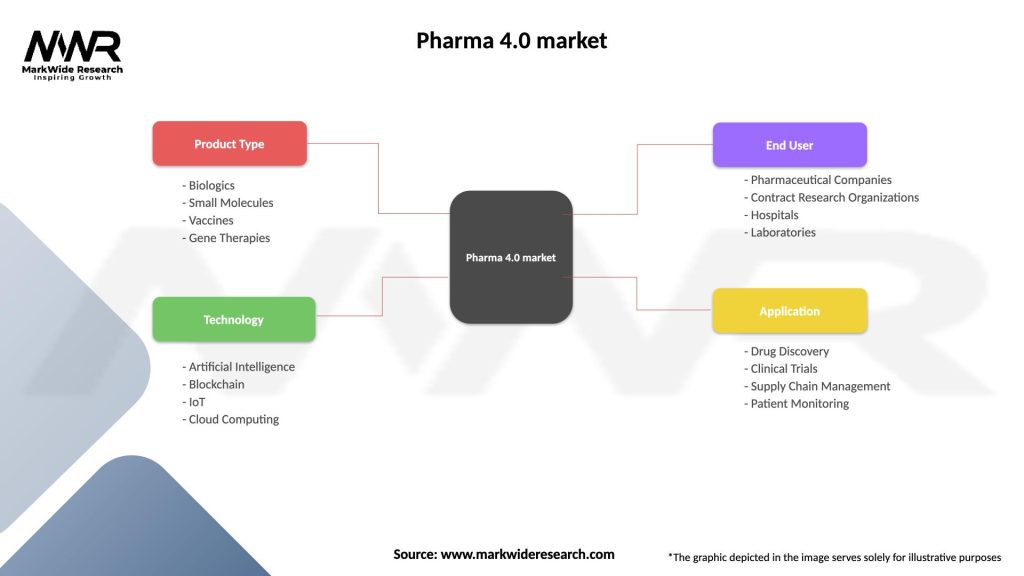
Segmentation
The Pharma 4.0 market can be segmented based on technology, application, and end-user. By technology, the market can be categorized into AI and ML, IoT, big data analytics, robotics, and others. Application-wise, the market can be divided into manufacturing execution systems (MES), supply chain management, laboratory information management systems (LIMS), quality control, and others. Based on end-users, the market can be classified into pharmaceutical companies, contract manufacturing organizations (CMOs), research organizations, and others.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Covid-19 Impact
The COVID-19 pandemic has highlighted the importance of digital transformation in the pharmaceutical industry. The adoption of Pharma 4.0 technologies has accelerated as companies strive to overcome disruptions in the supply chain, ensure continuity of drug production, and develop effective vaccines and treatments. Digital solutions have played a critical role in remote monitoring, telemedicine, virtual clinical trials, and data analysis for drug repurposing and vaccine development.
Key Industry Developments
Analyst Suggestions
Future Outlook
The Pharma 4.0 market is poised for significant growth in the coming years. The increasing adoption of digital technologies, advancements in AI and ML, the rise of personalized medicine, and the focus on patient-centric care will continue to drive the market. However, challenges related to implementation costs, data privacy, and resistance to change need to be addressed. As the industry embraces digital transformation, Pharma 4.0 has the potential to revolutionize the pharmaceutical sector, leading to improved drug development, enhanced patient outcomes, and a more efficient and agile industry.
Conclusion
Pharma 4.0 represents a paradigm shift in the pharmaceutical industry, integrating advanced technologies and digitalization to optimize processes, enhance product quality, and improve patient care. The market is driven by the need for cost-effective production, personalized medicine, and regulatory compliance. Despite challenges, such as implementation costs and data privacy concerns, Pharma 4.0 presents immense opportunities for industry participants. By embracing digital transformation, pharmaceutical companies can unlock significant benefits, including operational efficiency, improved product quality, accelerated time to market, and personalized patient care. The future of the Pharma 4.0 market looks promising, with continued advancements in technology and increasing collaboration between stakeholders.
What is Pharma 4.0?
Pharma 4.0 refers to the integration of advanced technologies such as artificial intelligence, big data, and the Internet of Things in the pharmaceutical industry. This approach aims to enhance efficiency, improve patient outcomes, and streamline drug development processes.
What are the key players in the Pharma 4.0 market?
Key players in the Pharma 4.0 market include companies like Siemens, Roche, and Novartis, which are leveraging digital technologies to transform their operations and improve drug manufacturing processes, among others.
What are the main drivers of growth in the Pharma 4.0 market?
The main drivers of growth in the Pharma 4.0 market include the increasing demand for personalized medicine, the need for operational efficiency, and the rising adoption of automation and data analytics in drug development.
What challenges does the Pharma 4.0 market face?
Challenges in the Pharma 4.0 market include regulatory compliance issues, the high cost of technology implementation, and the need for skilled workforce to manage advanced digital systems.
What opportunities exist in the Pharma 4.0 market?
Opportunities in the Pharma 4.0 market include the potential for enhanced patient engagement through digital health solutions, the development of smart manufacturing processes, and the ability to leverage real-time data for better decision-making.
What trends are shaping the Pharma 4.0 market?
Trends shaping the Pharma 4.0 market include the increasing use of blockchain for supply chain transparency, the rise of telemedicine, and the growing emphasis on data security and patient privacy in digital health initiatives.
Pharma 4.0 market
| Segmentation Details | Description |
|---|---|
| Product Type | Biologics, Small Molecules, Vaccines, Gene Therapies |
| Technology | Artificial Intelligence, Blockchain, IoT, Cloud Computing |
| End User | Pharmaceutical Companies, Contract Research Organizations, Hospitals, Laboratories |
| Application | Drug Discovery, Clinical Trials, Supply Chain Management, Patient Monitoring |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in Pharma 4.0 Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at