444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The Medical Use Sterile Rubber Stopper market is experiencing steady growth, driven by the increasing demand for high-quality packaging solutions in the pharmaceutical and biotechnology industries. Sterile rubber stoppers play a critical role in sealing medication vials, ampoules, and other containers, ensuring product integrity, stability, and sterility throughout the shelf life. The market is characterized by technological advancements, stringent regulatory requirements, and the emphasis on product safety and quality in healthcare settings.
Meaning
Medical Use Sterile Rubber Stoppers are specialized components designed to seal pharmaceutical containers and maintain product integrity, sterility, and stability. These stoppers are made from high-quality rubber materials such as bromobutyl rubber or chlorobutyl rubber and are subjected to stringent sterilization processes to ensure compliance with regulatory standards. Sterile rubber stoppers are essential for preventing contamination, evaporation, and degradation of medications, vaccines, and biologics, thereby safeguarding patient safety and therapeutic efficacy.
Executive Summary
The global Medical Use Sterile Rubber Stopper market is witnessing robust growth, driven by the increasing demand for sterile packaging solutions in the pharmaceutical and biotechnology sectors. Key factors contributing to market expansion include the growing prevalence of chronic diseases, rising demand for injectable medications, and the proliferation of biologic drugs and vaccines. Market players are focusing on product innovation, quality assurance, and regulatory compliance to meet the evolving needs of pharmaceutical manufacturers and ensure patient safety and product efficacy.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The Medical Use Sterile Rubber Stopper market is characterized by dynamic trends, including technological innovations, regulatory developments, and shifting industry preferences. Companies in the market are investing in research and development to innovate and differentiate their products, improve sealing performance, and address emerging customer needs. Strategic partnerships, mergers and acquisitions, and geographical expansion play a crucial role in driving market growth and maintaining a competitive advantage in the global sterile rubber stopper market.
Regional Analysis
North America dominates the global Medical Use Sterile Rubber Stopper market, driven by the presence of a well-established pharmaceutical industry, stringent regulatory standards, and high adoption of advanced packaging solutions. Europe follows closely, with significant contributions from countries such as Germany, the UK, and France. The Asia-Pacific region is expected to witness the fastest growth, attributed to rapid industrialization, increasing healthcare expenditure, and growing investments in pharmaceutical manufacturing infrastructure in countries such as China, India, and Japan.
Competitive Landscape
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Segmentation
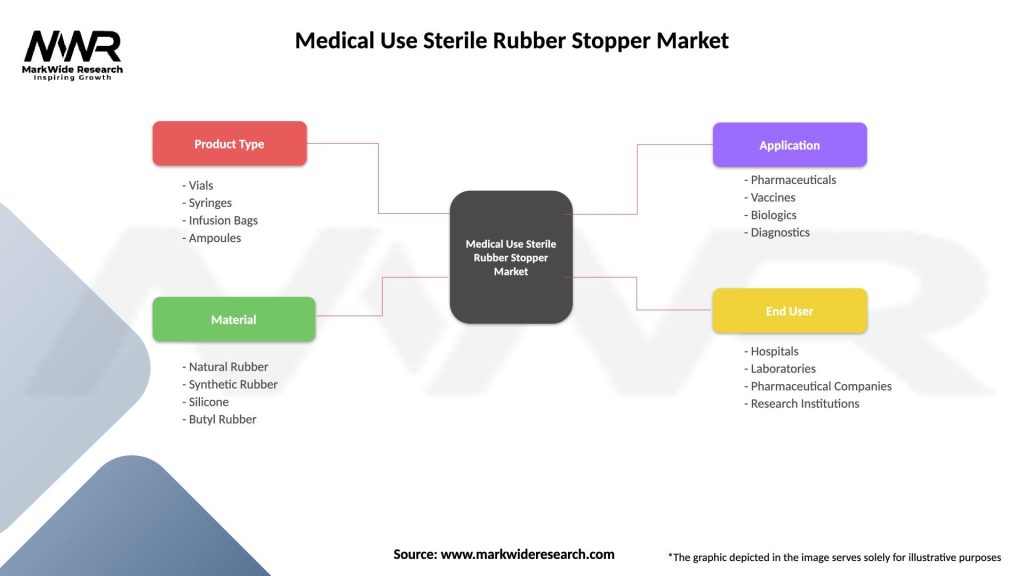
The Medical Use Sterile Rubber Stopper market can be segmented based on material type, closure type, packaging format, end-user, and region.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Market Key Trends
Covid-19 Impact
The Covid-19 pandemic has underscored the importance of sterile packaging solutions, container closure integrity, and infection control in pharmaceutical manufacturing. The pandemic has led to increased demand for sterile rubber stoppers for the packaging of vaccines, antiviral medications, and other critical healthcare products. Manufacturers have ramped up production to meet the surge in demand, while also focusing on ensuring product quality, regulatory compliance, and supply chain resilience in the face of pandemic-related challenges.
Key Industry Developments
Analyst Suggestions
Future Outlook
The global Medical Use Sterile Rubber Stopper market is poised for continued growth, driven by increasing demand for high-quality and reliable packaging solutions in the pharmaceutical and biotechnology industries. Key opportunities for market players include expansion into emerging markets, development of innovative rubber formulations, and collaboration with pharmaceutical manufacturers and regulatory agencies to address evolving industry needs. Companies that invest in innovation, quality assurance, and customer-centric strategies will be well-positioned to capitalize on these opportunities and drive growth in the global sterile rubber stopper market.
Conclusion
In conclusion, the Medical Use Sterile Rubber Stopper market presents significant opportunities for industry participants to address the growing demand for sterile packaging solutions and ensure product integrity, stability, and safety in pharmaceutical manufacturing. By leveraging advancements in rubber technology, manufacturing processes, and quality control, stakeholders can drive sustainable growth, improve patient outcomes, and maintain regulatory compliance in healthcare settings worldwide. As the market continues to evolve, a focus on innovation, collaboration, and customer satisfaction will be essential to unlocking the full potential of sterile rubber stoppers in safeguarding public health and supporting global healthcare initiatives.
What is Medical Use Sterile Rubber Stopper?
Medical Use Sterile Rubber Stopper refers to a type of closure used in pharmaceutical and medical applications to seal vials and containers, ensuring sterility and preventing contamination. These stoppers are essential for maintaining the integrity of medications and are commonly used in injectable drugs and laboratory settings.
What are the key players in the Medical Use Sterile Rubber Stopper Market?
Key players in the Medical Use Sterile Rubber Stopper Market include companies such as West Pharmaceutical Services, Schott AG, and AptarGroup, which are known for their innovative solutions in drug delivery and packaging. These companies focus on providing high-quality, compliant products to meet the needs of the healthcare industry, among others.
What are the growth factors driving the Medical Use Sterile Rubber Stopper Market?
The Medical Use Sterile Rubber Stopper Market is driven by the increasing demand for injectable drugs, the rise in chronic diseases requiring long-term medication, and advancements in drug formulation technologies. Additionally, the growing emphasis on patient safety and product integrity further fuels market growth.
What challenges does the Medical Use Sterile Rubber Stopper Market face?
Challenges in the Medical Use Sterile Rubber Stopper Market include stringent regulatory requirements, potential contamination risks during manufacturing, and the need for continuous innovation to meet evolving healthcare standards. These factors can impact production efficiency and product availability.
What opportunities exist in the Medical Use Sterile Rubber Stopper Market?
Opportunities in the Medical Use Sterile Rubber Stopper Market include the development of new materials that enhance sterility and compatibility with various drug formulations. Additionally, the increasing trend towards personalized medicine and biologics presents significant growth potential for specialized rubber stoppers.
What trends are shaping the Medical Use Sterile Rubber Stopper Market?
Trends in the Medical Use Sterile Rubber Stopper Market include the adoption of sustainable materials and eco-friendly manufacturing processes. There is also a growing focus on smart packaging solutions that incorporate technology for better tracking and monitoring of pharmaceutical products.
Medical Use Sterile Rubber Stopper Market
| Segmentation Details | Description |
|---|---|
| Product Type | Vials, Syringes, Infusion Bags, Ampoules |
| Material | Natural Rubber, Synthetic Rubber, Silicone, Butyl Rubber |
| Application | Pharmaceuticals, Vaccines, Biologics, Diagnostics |
| End User | Hospitals, Laboratories, Pharmaceutical Companies, Research Institutions |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at