444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at

Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$2750
The MEA bladder cancer therapeutics and diagnostics market represents a rapidly evolving healthcare sector addressing one of the most prevalent urological malignancies across the Middle East and Africa region. Bladder cancer continues to pose significant challenges for healthcare systems throughout these regions, with increasing incidence rates driving demand for advanced therapeutic interventions and diagnostic solutions. The market encompasses a comprehensive range of treatment modalities including chemotherapy, immunotherapy, targeted therapy, and surgical interventions, alongside sophisticated diagnostic technologies such as cystoscopy, imaging systems, and biomarker testing.
Regional healthcare infrastructure improvements across the MEA region have created substantial opportunities for market expansion, with countries like Saudi Arabia, UAE, South Africa, and Turkey leading adoption of innovative bladder cancer management solutions. The market demonstrates robust growth potential, with projections indicating a compound annual growth rate of 8.2% through the forecast period. Government healthcare initiatives and increasing healthcare expenditure across key MEA countries are driving market accessibility and treatment adoption rates.
Technological advancement in both therapeutic and diagnostic segments continues to reshape treatment paradigms, with personalized medicine approaches gaining significant traction. The integration of artificial intelligence in diagnostic imaging and the development of novel immunotherapeutic agents represent key growth catalysts for the regional market.
The MEA bladder cancer therapeutics and diagnostics market refers to the comprehensive healthcare sector encompassing all medical products, services, and technologies specifically designed for the prevention, diagnosis, treatment, and management of bladder cancer across Middle Eastern and African countries. This market includes pharmaceutical interventions such as chemotherapy agents, immunotherapy drugs, targeted therapies, and adjuvant treatments, alongside diagnostic technologies including cystoscopy equipment, imaging systems, urine-based tests, and molecular diagnostic tools.
Market scope extends beyond traditional treatment modalities to include emerging therapeutic approaches such as intravesical therapy, photodynamic therapy, and combination treatment protocols. The diagnostic component encompasses both invasive and non-invasive detection methods, ranging from conventional cystoscopy to advanced molecular biomarker analysis and liquid biopsy technologies.
Regional specificity of this market reflects unique healthcare challenges, regulatory environments, and economic conditions prevalent across MEA countries, influencing treatment accessibility, adoption patterns, and market dynamics distinct from global trends.
Market dynamics within the MEA bladder cancer therapeutics and diagnostics sector demonstrate significant growth momentum driven by increasing disease prevalence, improving healthcare infrastructure, and rising awareness of early detection benefits. The regional market benefits from substantial government healthcare investments, with healthcare spending increases of 12% annually across key MEA markets supporting expanded access to advanced treatment options.
Therapeutic segment dominance characterizes current market structure, with chemotherapy and immunotherapy representing the largest revenue contributors. However, diagnostic technologies are experiencing rapid growth, particularly in countries with established healthcare systems. South Africa and Saudi Arabia lead regional market development, accounting for significant market share due to advanced healthcare infrastructure and favorable reimbursement policies.
Innovation pipeline strength indicates robust future growth potential, with numerous clinical trials underway across the region testing novel therapeutic combinations and diagnostic technologies. The market demonstrates increasing adoption of personalized medicine approaches, with biomarker-guided therapy adoption rates reaching 35% in leading regional healthcare centers.
Competitive landscape features both international pharmaceutical companies and emerging regional players, creating dynamic market conditions that benefit healthcare providers and patients through improved treatment options and competitive pricing structures.
Primary market drivers include escalating bladder cancer incidence rates across the MEA region, with environmental factors and lifestyle changes contributing to disease burden increases. Healthcare infrastructure modernization across key regional markets creates expanding opportunities for advanced therapeutic and diagnostic solution deployment.
Market segmentation reveals distinct growth patterns across therapeutic categories, with targeted therapies demonstrating the highest growth rates despite smaller current market share. Diagnostic segment expansion reflects increasing emphasis on early detection and personalized treatment approaches.
Epidemiological trends represent the fundamental driver of MEA bladder cancer market growth, with regional cancer registries reporting consistent increases in bladder cancer diagnoses. Environmental risk factors prevalent across many MEA countries, including occupational exposures and water contamination, contribute to sustained disease incidence growth.
Healthcare system modernization across the region creates expanding infrastructure capable of supporting advanced cancer care delivery. Countries like UAE, Saudi Arabia, and South Africa have invested substantially in comprehensive cancer centers, creating demand for sophisticated therapeutic and diagnostic technologies. Medical tourism growth within the region further drives demand for world-class bladder cancer treatment capabilities.
Awareness campaign effectiveness has significantly improved early detection rates, with screening program participation increasing by 28% across major regional healthcare systems. Professional education initiatives enhance healthcare provider capabilities in bladder cancer management, supporting adoption of evidence-based treatment protocols.
Pharmaceutical industry investment in regional market development includes local manufacturing facilities, clinical research centers, and distribution networks. Regulatory environment improvements facilitate faster approval processes for innovative treatments, reducing time-to-market for breakthrough therapies.
Economic disparities across MEA countries create significant challenges for uniform market development, with treatment accessibility varying substantially between developed and developing regional markets. Healthcare financing limitations in certain countries restrict patient access to expensive therapeutic interventions, particularly novel immunotherapy and targeted therapy options.
Healthcare infrastructure gaps in rural and remote areas limit diagnostic capabilities and specialized treatment access. Skilled healthcare professional shortages across many regional markets constrain treatment capacity and quality of care delivery. Regulatory complexity and varying approval timelines across different MEA countries create market entry challenges for pharmaceutical companies.
Cultural and social barriers may delay diagnosis and treatment initiation, particularly in conservative societies where discussion of urological conditions remains sensitive. Limited health insurance coverage for advanced cancer treatments in certain markets restricts patient access to optimal care options.
Supply chain vulnerabilities can impact treatment continuity, particularly for specialized medications requiring cold chain storage and distribution. Currency fluctuations and economic instability in some regional markets create pricing pressures and market uncertainty for international pharmaceutical companies.
Emerging market potential across developing MEA countries presents substantial growth opportunities as healthcare infrastructure continues expanding. Public-private partnership development creates innovative financing models for cancer care delivery, improving treatment accessibility while generating sustainable business opportunities.
Digital health integration offers transformative opportunities for bladder cancer management, including telemedicine consultations, remote monitoring systems, and artificial intelligence-powered diagnostic support. Precision medicine advancement creates opportunities for companion diagnostic development and personalized treatment protocols tailored to regional patient populations.
Local manufacturing expansion presents opportunities for cost reduction and improved supply chain reliability. Biosimilar development for established bladder cancer therapeutics offers accessible treatment alternatives while maintaining clinical efficacy. Clinical research collaboration between regional institutions and international pharmaceutical companies generates valuable data while accelerating innovative treatment development.
Medical tourism growth positions leading regional healthcare centers as destinations for comprehensive bladder cancer care, creating revenue opportunities while improving regional treatment capabilities. Educational program expansion supports healthcare professional development and improves treatment outcomes across the region.
Competitive intensity within the MEA bladder cancer market continues increasing as both established pharmaceutical companies and emerging biotechnology firms compete for market share. Innovation cycles are accelerating, with new therapeutic approaches and diagnostic technologies regularly entering the market, creating dynamic competitive conditions.
Pricing pressures from government healthcare programs and insurance providers drive companies to demonstrate clear value propositions and cost-effectiveness for their products. Market consolidation trends through mergers and acquisitions reshape competitive landscapes while potentially improving treatment access through expanded distribution networks.
Regulatory evolution across MEA countries increasingly emphasizes patient safety and treatment efficacy, requiring companies to invest in comprehensive clinical evidence generation. Technology convergence between therapeutic and diagnostic segments creates opportunities for integrated treatment solutions and comprehensive patient management platforms.
Patient advocacy influence grows stronger across the region, with organized patient groups advocating for improved treatment access and healthcare policy changes. Healthcare provider consolidation creates larger purchasing entities with increased negotiating power, influencing market dynamics and pricing strategies.
Comprehensive market analysis employs multiple research methodologies to ensure accurate and reliable market insights. Primary research activities include extensive interviews with healthcare professionals, pharmaceutical executives, regulatory officials, and patient advocacy representatives across key MEA markets.
Secondary research components encompass analysis of published clinical studies, regulatory filings, company financial reports, and healthcare statistics from regional health ministries. Market modeling techniques utilize both bottom-up and top-down approaches to validate market size estimates and growth projections.
Data triangulation methods ensure research accuracy by comparing findings across multiple sources and methodologies. Regional market surveys capture local market dynamics, treatment patterns, and healthcare provider preferences specific to different MEA countries.
Expert panel consultations with leading oncologists, urologists, and healthcare economists provide clinical and economic insights essential for comprehensive market understanding. Regulatory analysis examines approval pathways, reimbursement policies, and healthcare regulations across major regional markets.
Saudi Arabia leads the MEA bladder cancer therapeutics and diagnostics market, accounting for approximately 22% of regional market share due to substantial healthcare investments and comprehensive cancer care infrastructure. Vision 2030 initiatives continue driving healthcare sector modernization, creating expanding opportunities for advanced treatment technologies.
South Africa represents the second-largest regional market, with well-established healthcare systems and strong clinical research capabilities. Private healthcare sector strength supports adoption of innovative treatments, while public sector initiatives improve treatment accessibility across diverse population groups.
United Arab Emirates demonstrates rapid market growth driven by medical tourism development and healthcare infrastructure investments. Dubai and Abu Dhabi serve as regional healthcare hubs, attracting patients from across the MEA region for specialized bladder cancer treatment.
Turkey benefits from strategic geographic positioning and growing pharmaceutical manufacturing capabilities. Healthcare system reforms improve treatment accessibility while maintaining cost-effectiveness. Egypt shows significant growth potential with population-driven demand increases of 15% annually, though infrastructure development remains ongoing.
Emerging markets including Morocco, Jordan, and Kenya demonstrate increasing treatment adoption rates as healthcare infrastructure continues expanding. Regional cooperation initiatives facilitate knowledge sharing and treatment protocol standardization across MEA countries.
Market leadership within the MEA bladder cancer therapeutics and diagnostics sector features a combination of established international pharmaceutical companies and emerging regional players. Competitive positioning depends on factors including product portfolio breadth, regional market presence, and local partnership strength.
Strategic partnerships between international companies and regional healthcare providers create competitive advantages through improved market access and local expertise. Research and development investments continue driving innovation, with companies focusing on region-specific treatment needs and patient populations.
Emerging competitors include regional pharmaceutical companies developing biosimilar products and specialized diagnostic technology providers offering cost-effective solutions tailored to MEA market requirements.
By Treatment Type:
By Diagnostic Method:
By End User:
Therapeutic Categories:
Immunotherapy segment demonstrates the highest growth rates within the MEA market, with adoption rates increasing by 42% annually across leading regional cancer centers. Checkpoint inhibitors such as pembrolizumab and atezolizumab gain increasing acceptance among oncologists, supported by strong clinical evidence and improving reimbursement coverage.
Chemotherapy remains the backbone of bladder cancer treatment across the region, with cisplatin-based combinations maintaining clinical standard of care status. Targeted therapy adoption grows steadily, particularly in countries with advanced healthcare infrastructure and personalized medicine capabilities.
Diagnostic Categories:
Non-invasive diagnostic methods experience rapid growth as healthcare providers seek to improve patient comfort while maintaining diagnostic accuracy. Liquid biopsy technologies show particular promise for treatment monitoring and recurrence detection. Artificial intelligence integration in imaging analysis improves diagnostic accuracy while reducing interpretation time.
Cystoscopy equipment modernization continues across the region, with flexible cystoscopes and enhanced imaging capabilities improving patient experience and diagnostic yield. Point-of-care testing development addresses healthcare access challenges in remote areas.
Healthcare Providers:
Patients:
Pharmaceutical Companies:
Strengths:
Weaknesses:
Opportunities:
Threats:
Personalized Medicine Adoption represents the most significant trend shaping the MEA bladder cancer market, with biomarker testing adoption rates reaching 38% in leading regional cancer centers. Companion diagnostic development supports targeted therapy selection, improving treatment outcomes while optimizing healthcare resource utilization.
Digital Health Integration transforms patient care delivery through telemedicine platforms, remote monitoring systems, and artificial intelligence-powered diagnostic support. Mobile health applications improve patient engagement and treatment adherence, particularly valuable in geographically diverse regional markets.
Combination Therapy Approaches gain increasing clinical acceptance as oncologists seek to maximize treatment efficacy while managing side effect profiles. Immunotherapy combinations with chemotherapy or targeted agents show promising clinical results in regional patient populations.
Minimally Invasive Procedures continue advancing, with robotic surgery adoption and enhanced endoscopic techniques improving patient outcomes while reducing recovery times. Outpatient treatment protocols become more prevalent, reducing healthcare costs while maintaining treatment quality.
Regional Manufacturing Expansion addresses supply chain challenges while reducing treatment costs. Local pharmaceutical production capabilities improve treatment accessibility across price-sensitive markets within the MEA region.
Regulatory Milestone Achievements across key MEA markets facilitate faster approval processes for innovative bladder cancer treatments. Saudi FDA and South African Health Products Regulatory Authority implement streamlined review procedures for breakthrough therapies, reducing time-to-market for critical treatments.
Clinical Trial Expansion across the region generates valuable real-world evidence while providing patients access to experimental treatments. MarkWide Research analysis indicates clinical trial activity increased by 45% across major MEA markets over the past two years, reflecting growing research infrastructure and international collaboration.
Healthcare Infrastructure Projects including comprehensive cancer centers in Dubai, Riyadh, and Cape Town enhance regional treatment capabilities. Public-private partnerships facilitate sustainable financing models for advanced cancer care delivery.
Technology Transfer Agreements between international pharmaceutical companies and regional manufacturers improve local production capabilities while reducing treatment costs. Biosimilar approvals for established bladder cancer therapeutics provide cost-effective treatment alternatives.
Professional Education Initiatives including fellowship programs and continuing medical education enhance healthcare provider capabilities in bladder cancer management. Regional oncology societies facilitate knowledge sharing and treatment protocol standardization.
Market Entry Strategies for pharmaceutical companies should prioritize partnership development with established regional healthcare providers and distributors. Local market expertise proves essential for navigating regulatory requirements and cultural considerations across diverse MEA markets.
Investment Priorities should focus on digital health technologies and personalized medicine capabilities, as these represent the highest growth potential segments. Diagnostic technology development offers particular opportunities given the emphasis on early detection and treatment monitoring.
Pricing Strategy Optimization requires careful consideration of economic disparities across the region, with tiered pricing models potentially improving market access while maintaining profitability. Value-based pricing approaches aligned with treatment outcomes can support reimbursement negotiations.
Clinical Development Focus should emphasize regional patient population characteristics and treatment patterns. Real-world evidence generation through post-market studies can support expanded indications and improved treatment protocols.
Partnership Development with regional academic institutions and research centers can accelerate innovation while building local market presence. Technology transfer initiatives may provide sustainable competitive advantages in price-sensitive markets.
Market growth trajectory for the MEA bladder cancer therapeutics and diagnostics sector remains strongly positive, with sustained expansion expected across all major regional markets. Healthcare infrastructure development continues accelerating, creating expanding opportunities for advanced treatment and diagnostic technology deployment.
Innovation pipeline strength indicates continued market evolution, with novel therapeutic approaches and diagnostic technologies regularly entering clinical development. Precision medicine advancement will likely transform treatment paradigms, with personalized therapy adoption projected to reach 65% in leading regional markets within the next five years.
Digital health integration will fundamentally reshape patient care delivery, with artificial intelligence, telemedicine, and remote monitoring becoming standard components of comprehensive bladder cancer management. MWR projections suggest digital health solutions will capture significant market share growth of 25% annually.
Regional market consolidation may occur as healthcare systems seek economies of scale and improved treatment standardization. Cross-border collaboration will likely increase, facilitating knowledge sharing and resource optimization across MEA countries.
Regulatory harmonization efforts across the region should streamline market access for innovative treatments while maintaining safety standards. Biosimilar market expansion will provide cost-effective treatment alternatives, improving accessibility across diverse economic conditions.
The MEA bladder cancer therapeutics and diagnostics market presents substantial growth opportunities driven by increasing disease prevalence, improving healthcare infrastructure, and advancing treatment technologies. Regional market dynamics reflect unique challenges and opportunities that require tailored strategies for successful market participation.
Innovation continues driving market evolution, with personalized medicine, digital health integration, and combination therapy approaches reshaping treatment paradigms. Healthcare investment growth across key regional markets supports sustained market expansion while improving patient access to advanced care options.
Competitive landscape evolution creates opportunities for both established pharmaceutical companies and emerging regional players to contribute to improved bladder cancer outcomes. Strategic partnerships and local market expertise prove essential for navigating diverse regulatory environments and cultural considerations across MEA countries.
Future market success will depend on companies’ ability to balance innovation with accessibility, ensuring advanced treatments reach diverse patient populations across the economically varied MEA region. Sustainable growth strategies must address both clinical efficacy and cost-effectiveness to maximize market impact and patient benefit.
What is Bladder Cancer Therapeutics & Diagnostics?
Bladder Cancer Therapeutics & Diagnostics refers to the medical treatments and diagnostic tools used to manage and detect bladder cancer. This includes various therapies such as chemotherapy, immunotherapy, and targeted therapies, as well as diagnostic methods like imaging and biomarker tests.
What are the key players in the MEA Bladder Cancer Therapeutics & Diagnostics Market?
Key players in the MEA Bladder Cancer Therapeutics & Diagnostics Market include companies like Roche, Merck, and Bristol-Myers Squibb, which are known for their innovative cancer therapies and diagnostic solutions, among others.
What are the growth factors driving the MEA Bladder Cancer Therapeutics & Diagnostics Market?
The growth of the MEA Bladder Cancer Therapeutics & Diagnostics Market is driven by factors such as the increasing prevalence of bladder cancer, advancements in diagnostic technologies, and the rising demand for personalized medicine in cancer treatment.
What challenges does the MEA Bladder Cancer Therapeutics & Diagnostics Market face?
Challenges in the MEA Bladder Cancer Therapeutics & Diagnostics Market include high treatment costs, regulatory hurdles for new therapies, and the need for more effective diagnostic methods to improve early detection rates.
What opportunities exist in the MEA Bladder Cancer Therapeutics & Diagnostics Market?
Opportunities in the MEA Bladder Cancer Therapeutics & Diagnostics Market include the development of novel therapies, the integration of artificial intelligence in diagnostics, and expanding access to healthcare services in emerging markets.
What trends are shaping the MEA Bladder Cancer Therapeutics & Diagnostics Market?
Trends shaping the MEA Bladder Cancer Therapeutics & Diagnostics Market include the increasing focus on immunotherapy, the rise of liquid biopsy technologies, and the growing emphasis on patient-centric care in treatment approaches.
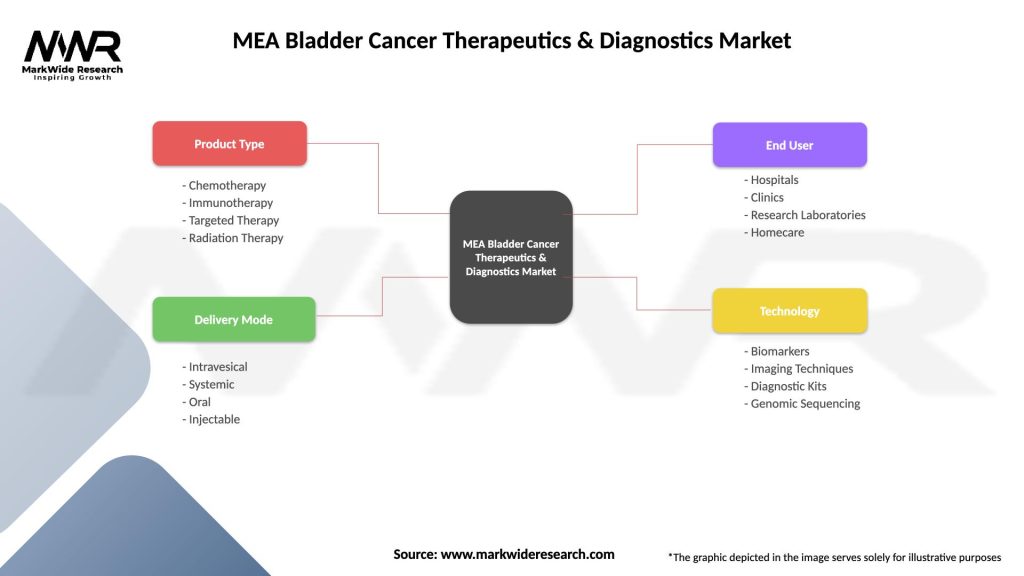
MEA Bladder Cancer Therapeutics & Diagnostics Market
| Segmentation Details | Description |
|---|---|
| Product Type | Chemotherapy, Immunotherapy, Targeted Therapy, Radiation Therapy |
| Delivery Mode | Intravesical, Systemic, Oral, Injectable |
| End User | Hospitals, Clinics, Research Laboratories, Homecare |
| Technology | Biomarkers, Imaging Techniques, Diagnostic Kits, Genomic Sequencing |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading companies in the MEA Bladder Cancer Therapeutics & Diagnostics Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at