444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
Malaria is a life-threatening disease caused by parasites transmitted through the bite of infected mosquitoes. It poses a significant global health challenge, particularly in regions with high malaria transmission rates. The development of an effective malaria vaccine has been a long-standing goal in the healthcare industry. A malaria vaccine aims to prevent the transmission of the parasite and provide long-lasting immunity against the disease.
Meaning
The malaria vaccine market refers to the commercial market for vaccines designed to prevent malaria infection. This market includes various stakeholders such as pharmaceutical companies, research institutions, healthcare providers, and government organizations working towards the development, manufacturing, and distribution of malaria vaccines. The market is driven by the growing need to combat malaria and reduce its burden on affected populations.
Executive Summary
The malaria vaccine market is witnessing significant growth as various vaccine candidates are progressing through clinical trials and obtaining regulatory approvals. The global focus on eliminating malaria and reducing its impact has led to increased investments in research and development of malaria vaccines. Additionally, public-private partnerships and initiatives by global health organizations have contributed to the advancement of vaccine development and accessibility in malaria-endemic regions.
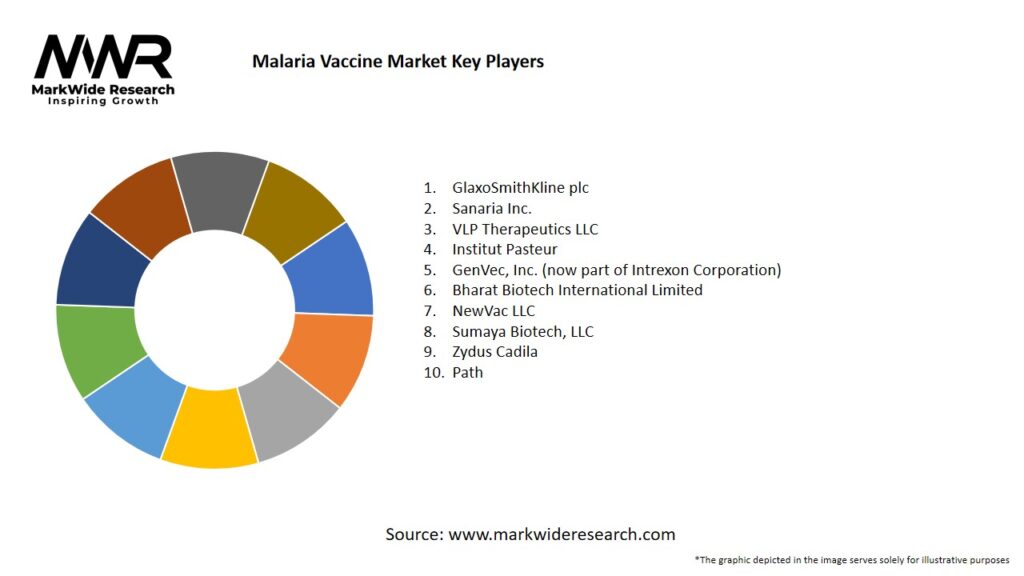
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
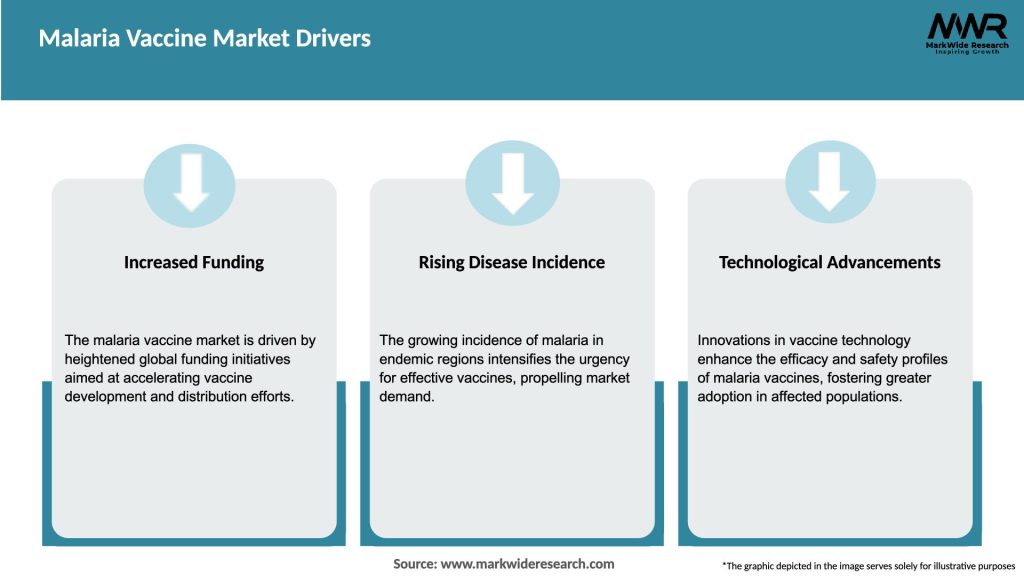
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The malaria vaccine market is characterized by dynamic factors that shape its growth and evolution. These dynamics include technological advancements, government policies, funding opportunities, disease prevalence, and emerging market trends. Continuous research and development efforts, coupled with collaborations among key stakeholders, are key drivers of market growth. However, regulatory challenges, complex parasite biology, and high development costs pose significant barriers to entry and commercialization.
Regional Analysis
The malaria vaccine market exhibits regional variations due to differences in disease prevalence, healthcare infrastructure, and government initiatives. Sub-Saharan Africa, which carries the highest malaria burden, represents a significant market opportunity for malaria vaccines. Other regions with substantial malaria transmission rates, such as Southeast Asia and parts of Latin America, also present growth prospects. Market players need to adapt their strategies to address the unique challenges and requirements of each region.
Competitive Landscape
Leading Companies in the Malaria Vaccine Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
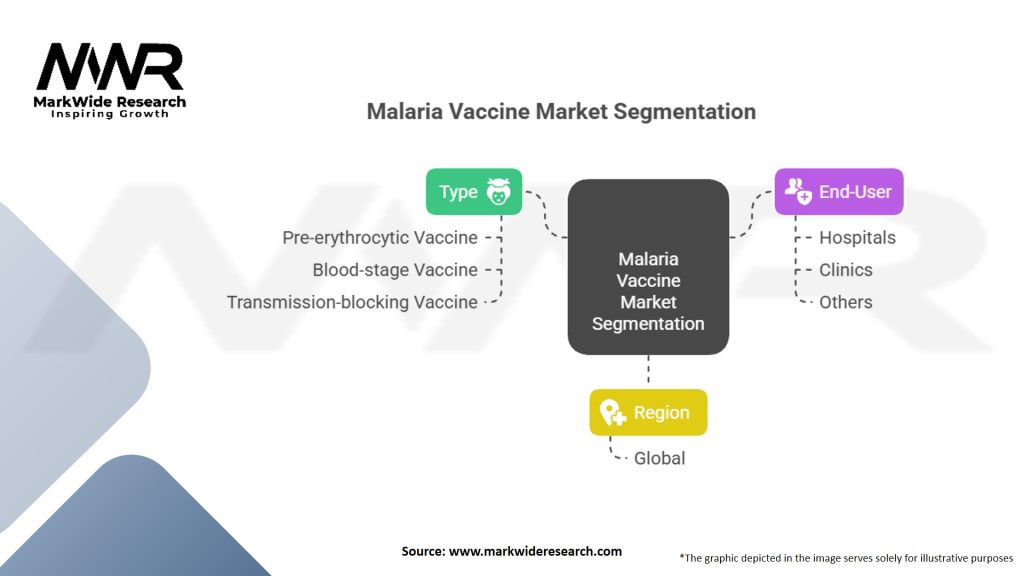
Segmentation
The malaria vaccine market can be segmented based on various factors, including vaccine type, end-user, and region. Common vaccine types include pre-erythrocytic vaccines, blood-stage vaccines, and transmission-blocking vaccines. End-users of malaria vaccines include healthcare facilities, government agencies, and research institutions. Region-wise segmentation helps identify specific market trends and opportunities in different geographic locations.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Market Key Trends
Covid-19 Impact
The COVID-19 pandemic has had a significant impact on the malaria vaccine market. While the resources and focus of the global healthcare community were directed towards combating the pandemic, the development and distribution of malaria vaccines faced challenges. Clinical trials were disrupted, funding priorities shifted, and vaccine rollout efforts were affected. However, the pandemic also highlighted the importance of vaccine research and the need for robust immunization strategies to prevent infectious diseases.
Key Industry Developments
Analyst Suggestions
Future Outlook
The future of the malaria vaccine market looks promising, with increasing investments, advancements in vaccine technology, and global initiatives to combat malaria. As more vaccine candidates progress through clinical trials and obtain regulatory approvals, the market is expected to witness significant growth. Ongoing research and collaborative efforts hold the key to the development of highly effective malaria vaccines that can contribute to the global goal of malaria elimination.
Conclusion
The malaria vaccine market presents immense potential to reduce the burden of malaria and improve public health outcomes. With increasing investments in research and development, strategic collaborations, and advancements in vaccine technology, the market is poised for growth. Overcoming challenges such as complex parasite biology, high development costs, and regulatory hurdles will require continued dedication and collaborative efforts. As the world works towards eliminating malaria, the development and widespread adoption of safe and effective malaria vaccines are crucial steps in achieving this goal.
What is the Malaria Vaccine?
The Malaria Vaccine refers to immunizations designed to prevent malaria, a disease caused by parasites transmitted through the bites of infected mosquitoes. These vaccines aim to stimulate the immune system to recognize and combat the malaria parasites effectively.
Which companies are leading the Malaria Vaccine Market?
Leading companies in the Malaria Vaccine Market include GlaxoSmithKline, Sanaria, and Merck, which are actively involved in the development and distribution of malaria vaccines. These companies focus on innovative research and partnerships to enhance vaccine efficacy and accessibility among others.
What are the key drivers of the Malaria Vaccine Market?
Key drivers of the Malaria Vaccine Market include the rising incidence of malaria in endemic regions, increased funding for vaccine research, and growing awareness of malaria prevention strategies. Additionally, advancements in vaccine technology are contributing to market growth.
What challenges does the Malaria Vaccine Market face?
The Malaria Vaccine Market faces challenges such as the complexity of malaria biology, high development costs, and logistical issues in vaccine distribution in remote areas. Furthermore, vaccine hesitancy in some populations can hinder adoption.
What opportunities exist in the Malaria Vaccine Market?
Opportunities in the Malaria Vaccine Market include the potential for new vaccine candidates that target different malaria strains and the expansion of vaccination programs in high-risk areas. Collaborations with global health organizations can also enhance outreach and effectiveness.
What trends are shaping the Malaria Vaccine Market?
Trends shaping the Malaria Vaccine Market include the integration of digital health technologies for tracking vaccination rates and the development of combination vaccines that address multiple diseases. Additionally, there is a growing emphasis on community engagement in vaccination campaigns.
Malaria Vaccine Market Segmentation:
| Segment | Segmentation Details |
|---|---|
| Type | Pre-erythrocytic Vaccine, Blood-stage Vaccine, Transmission-blocking Vaccine |
| End-User | Hospitals, Clinics, Others |
| Region | Global |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Malaria Vaccine Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at