444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at

Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
The isolators in pharmaceutical market represents a critical segment of pharmaceutical manufacturing infrastructure, focusing on advanced containment systems that protect both products and personnel from contamination. These sophisticated systems create controlled environments essential for sterile manufacturing, aseptic processing, and handling of potent compounds. Market dynamics indicate robust growth driven by stringent regulatory requirements, increasing demand for sterile pharmaceuticals, and rising focus on operator safety.
Pharmaceutical isolators serve as primary barriers in manufacturing facilities, providing superior contamination control compared to traditional cleanroom environments. The market encompasses various isolator types including positive pressure, negative pressure, and dual-purpose systems designed for specific pharmaceutical applications. Growth projections suggest the market will expand at a CAGR of 8.2% through the forecast period, reflecting increasing adoption across pharmaceutical manufacturing facilities globally.
Regional distribution shows North America maintaining market leadership with approximately 42% market share, followed by Europe and Asia-Pacific regions experiencing rapid expansion. The market benefits from technological advancements in isolator design, integration with automated systems, and enhanced monitoring capabilities that ensure compliance with evolving pharmaceutical manufacturing standards.
The isolators in pharmaceutical market refers to the comprehensive ecosystem of containment systems, technologies, and services designed to create sterile, controlled environments for pharmaceutical manufacturing and research applications. These systems provide physical barriers that isolate processes from external contamination while protecting operators from exposure to hazardous substances.
Pharmaceutical isolators function as enclosed workspaces equipped with glove ports, transfer systems, and advanced filtration mechanisms. They maintain controlled atmospheric conditions through precise air pressure management, HEPA filtration, and environmental monitoring systems. The market encompasses hardware components, software integration, validation services, and ongoing maintenance support required for optimal isolator performance.
Modern isolator systems integrate sophisticated technologies including automated decontamination cycles, real-time environmental monitoring, and data logging capabilities that ensure regulatory compliance. These systems support various pharmaceutical processes from sterile compounding and aseptic filling to cytotoxic drug handling and microbiology testing applications.
Market fundamentals demonstrate strong growth momentum in the isolators in pharmaceutical market, driven by increasing regulatory scrutiny and expanding sterile pharmaceutical production. The market benefits from rising demand for personalized medicines, growing biosimilar development, and enhanced focus on contamination prevention in pharmaceutical manufacturing processes.
Technology evolution continues advancing isolator capabilities through integration with Industry 4.0 technologies, improved automation features, and enhanced user interfaces. These developments enable pharmaceutical manufacturers to achieve higher efficiency levels while maintaining strict contamination control standards. Adoption rates show approximately 73% of major pharmaceutical facilities now utilize isolator technology for critical manufacturing processes.
Competitive dynamics feature established manufacturers expanding product portfolios while emerging companies introduce innovative solutions targeting specific pharmaceutical applications. The market structure supports both standardized isolator systems and customized solutions designed for specialized manufacturing requirements. Investment trends indicate increased funding for isolator technology development, particularly in areas of automation integration and smart monitoring systems.
Strategic insights reveal several critical factors shaping the isolators in pharmaceutical market landscape:
Market intelligence indicates that pharmaceutical companies prioritize isolator systems offering flexible configurations, scalable designs, and comprehensive validation support. These insights guide manufacturers in developing solutions that address evolving pharmaceutical industry requirements while maintaining competitive positioning in the market.
Primary growth drivers propelling the isolators in pharmaceutical market include escalating regulatory requirements for sterile manufacturing environments. Regulatory agencies worldwide mandate strict contamination control measures, making isolator technology essential for pharmaceutical compliance. Enforcement trends show regulatory inspections focusing increasingly on environmental controls and contamination prevention protocols.
Pharmaceutical industry expansion creates substantial demand for isolator systems as companies establish new manufacturing facilities and upgrade existing operations. The growing biosimilar market, personalized medicine development, and increased sterile injectable production drive isolator adoption across diverse pharmaceutical applications. Manufacturing growth in emerging markets contributes significantly to global isolator demand.
Safety considerations represent another crucial driver as pharmaceutical companies prioritize operator protection from exposure to potent compounds and cytotoxic substances. Enhanced awareness of occupational health risks associated with pharmaceutical manufacturing processes increases isolator adoption rates. Workplace safety regulations continue evolving to require more stringent containment measures in pharmaceutical facilities.
Technological advancement drives market growth through improved isolator capabilities, enhanced automation integration, and superior monitoring systems. These developments enable pharmaceutical manufacturers to achieve higher productivity levels while maintaining strict quality standards. Innovation cycles consistently introduce new features that expand isolator applications across pharmaceutical manufacturing processes.
Capital investment requirements present significant barriers to isolator adoption, particularly for smaller pharmaceutical companies and contract manufacturing organizations. High initial costs associated with isolator procurement, installation, and validation can strain budgets and delay implementation timelines. Financial constraints often require companies to prioritize isolator investments based on critical manufacturing needs.
Technical complexity challenges pharmaceutical facilities in isolator system integration, operation, and maintenance. Specialized training requirements for personnel, complex validation procedures, and ongoing technical support needs can overwhelm organizations lacking adequate resources. Implementation difficulties may result in extended project timelines and increased operational costs.
Space limitations in existing pharmaceutical facilities constrain isolator installation options and may require costly facility modifications. Retrofitting older manufacturing spaces to accommodate modern isolator systems presents engineering challenges and potential production disruptions. Infrastructure requirements including adequate HVAC systems, electrical capacity, and structural support add complexity to isolator projects.
Maintenance demands require ongoing investment in specialized services, replacement parts, and system upgrades throughout isolator lifecycles. Regular calibration, validation, and preventive maintenance activities consume resources and may impact production schedules. Operational costs associated with isolator maintenance can accumulate significantly over system lifespans.
Emerging market expansion presents substantial opportunities for isolator manufacturers as pharmaceutical companies establish operations in developing regions. Growing healthcare infrastructure, increasing pharmaceutical manufacturing capabilities, and rising regulatory standards in these markets drive isolator demand. Geographic expansion strategies enable companies to capture market share in high-growth regions.
Automation integration opportunities emerge as pharmaceutical manufacturers seek to enhance efficiency through Industry 4.0 technologies. Advanced isolator systems incorporating robotics, artificial intelligence, and predictive analytics offer competitive advantages in pharmaceutical manufacturing. Technology convergence creates new market segments for intelligent isolator solutions.
Personalized medicine growth generates demand for flexible isolator systems capable of handling small-batch production and diverse pharmaceutical formulations. Customizable isolator configurations supporting rapid changeover between products address evolving pharmaceutical manufacturing requirements. Market segmentation opportunities exist for specialized isolator solutions targeting specific therapeutic areas.
Service sector expansion offers revenue growth through comprehensive isolator lifecycle support including validation, maintenance, training, and upgrade services. Pharmaceutical companies increasingly prefer integrated service packages that ensure optimal isolator performance throughout system lifecycles. Service opportunities provide recurring revenue streams and strengthen customer relationships.
Supply chain dynamics in the isolators in pharmaceutical market reflect complex interactions between equipment manufacturers, pharmaceutical companies, and regulatory agencies. MarkWide Research analysis indicates that supply chain optimization efforts focus on reducing lead times, improving component quality, and enhancing customer support services. These dynamics influence pricing strategies and competitive positioning across the market.
Demand patterns show cyclical variations based on pharmaceutical industry investment cycles, regulatory changes, and new product launches. Peak demand periods typically coincide with facility expansions, regulatory compliance deadlines, and technology upgrade cycles. Market responsiveness requires manufacturers to maintain flexible production capabilities and adequate inventory levels.
Innovation cycles drive continuous product development as manufacturers compete to introduce advanced features and improved performance capabilities. Research and development investments focus on enhancing isolator efficiency, reducing operational costs, and expanding application possibilities. Technology adoption rates indicate that approximately 68% of new isolator installations incorporate advanced monitoring and automation features.
Competitive intensity influences pricing strategies, product differentiation efforts, and market share dynamics. Established manufacturers leverage brand recognition and installed base advantages while emerging companies compete through innovative solutions and competitive pricing. Market consolidation trends show strategic acquisitions and partnerships reshaping competitive landscapes.
Research approach for analyzing the isolators in pharmaceutical market employs comprehensive primary and secondary research methodologies to ensure accurate market assessment and reliable forecasting. Primary research includes structured interviews with industry executives, pharmaceutical manufacturers, isolator suppliers, and regulatory experts to gather firsthand insights into market dynamics and trends.
Data collection methods encompass surveys of pharmaceutical facility managers, technical specialists, and procurement professionals to understand isolator selection criteria, performance requirements, and satisfaction levels. Secondary research involves analysis of industry reports, regulatory documents, patent filings, and company financial statements to validate primary research findings and identify market patterns.
Market sizing methodology utilizes bottom-up and top-down approaches to estimate market dimensions and growth projections. Bottom-up analysis examines individual market segments, regional markets, and application areas to build comprehensive market models. Top-down analysis validates findings through macroeconomic indicators and industry benchmarks.
Quality assurance procedures include data triangulation, expert validation, and statistical analysis to ensure research accuracy and reliability. Multiple data sources are cross-referenced to verify market information and eliminate potential biases. Analytical frameworks incorporate quantitative and qualitative assessment methods to provide balanced market perspectives.
North American market maintains leadership position with approximately 42% global market share, driven by stringent regulatory requirements, advanced pharmaceutical manufacturing infrastructure, and high adoption rates of innovative isolator technologies. The region benefits from established pharmaceutical companies, robust research and development activities, and supportive regulatory frameworks that encourage isolator implementation.
European market represents the second-largest regional segment with strong growth momentum supported by harmonized regulatory standards, increasing pharmaceutical manufacturing activities, and growing focus on operator safety. Key markets including Germany, United Kingdom, and Switzerland demonstrate high isolator adoption rates across pharmaceutical and biotechnology sectors.
Asia-Pacific region exhibits the fastest growth rates with expanding pharmaceutical manufacturing capabilities, increasing regulatory compliance requirements, and rising healthcare infrastructure investments. Countries such as China, India, and Japan drive regional market expansion through facility modernization projects and new pharmaceutical manufacturing establishments.
Latin American markets show emerging opportunities as pharmaceutical companies establish regional manufacturing operations and regulatory standards evolve to align with international requirements. Brazil and Mexico lead regional market development through pharmaceutical industry investments and healthcare infrastructure improvements.
Middle East and Africa represent developing markets with growth potential driven by healthcare sector expansion, pharmaceutical manufacturing investments, and increasing regulatory sophistication. These regions offer long-term opportunities for isolator manufacturers seeking geographic diversification.
Market leadership is characterized by several established manufacturers offering comprehensive isolator solutions and services:
Competitive strategies focus on product innovation, geographic expansion, strategic partnerships, and comprehensive service offerings. Companies invest heavily in research and development to introduce advanced isolator features and maintain technological leadership. Market positioning emphasizes quality, reliability, and regulatory compliance to differentiate offerings in competitive markets.
By Pressure Type:
By Application:
By End User:
Positive pressure isolators dominate market demand due to widespread applications in sterile pharmaceutical manufacturing. These systems provide superior product protection by maintaining higher internal pressure than surrounding environments, preventing contamination ingress. Technology advancements in positive pressure systems focus on energy efficiency, automated monitoring, and integration capabilities.
Negative pressure isolators serve critical roles in cytotoxic drug handling and potent compound processing where operator protection is paramount. These systems contain hazardous substances by maintaining lower internal pressure, ensuring contaminants cannot escape to surrounding areas. Safety regulations increasingly mandate negative pressure isolators for handling oncology drugs and highly potent active pharmaceutical ingredients.
Dual-purpose isolators offer operational flexibility by supporting both positive and negative pressure modes within single systems. These versatile solutions enable pharmaceutical facilities to optimize equipment utilization and accommodate diverse manufacturing requirements. Adoption trends show growing preference for dual-purpose systems in facilities handling multiple product types.
Custom isolator solutions address specific pharmaceutical applications requiring specialized configurations, materials, or performance characteristics. These systems command premium pricing but provide optimal solutions for unique manufacturing challenges. Customization demand increases as pharmaceutical companies pursue specialized manufacturing processes and niche therapeutic areas.
Pharmaceutical manufacturers benefit from isolator technology through enhanced product quality, regulatory compliance assurance, and reduced contamination risks. Isolators enable companies to meet stringent manufacturing standards while protecting valuable pharmaceutical products from environmental contamination. Quality improvements result in reduced product recalls, enhanced brand reputation, and increased customer confidence.
Regulatory compliance advantages include simplified validation processes, comprehensive documentation capabilities, and alignment with evolving pharmaceutical manufacturing guidelines. Isolators provide auditable evidence of contamination control measures, facilitating regulatory inspections and approvals. Compliance benefits reduce regulatory risks and support market access for pharmaceutical products.
Operational efficiency gains emerge through reduced cleanroom requirements, streamlined manufacturing processes, and enhanced worker productivity. Isolators eliminate need for extensive gowning procedures and enable more efficient facility utilization. Productivity improvements of up to 35% efficiency gains are achievable through optimized isolator implementations.
Worker safety enhancements protect pharmaceutical personnel from exposure to hazardous substances, reducing occupational health risks and associated liability concerns. Isolators provide physical barriers that eliminate direct contact with potent compounds and cytotoxic drugs. Safety benefits include reduced worker compensation claims and improved employee satisfaction.
Strengths:
Weaknesses:
Opportunities:
Threats:
Automation integration represents a dominant trend as pharmaceutical manufacturers seek to enhance efficiency through robotic systems, automated material handling, and intelligent process control. Modern isolators increasingly incorporate automated features that reduce manual intervention while maintaining strict contamination control. Integration rates show approximately 58% of new isolator projects include automation components.
Smart monitoring systems gain prominence through real-time environmental monitoring, predictive maintenance capabilities, and data analytics integration. These systems provide continuous oversight of isolator performance, enabling proactive maintenance and compliance documentation. Digital transformation initiatives drive adoption of connected isolator technologies across pharmaceutical facilities.
Modular design approaches enable pharmaceutical companies to implement scalable isolator solutions that accommodate changing manufacturing requirements. Modular systems support facility expansion, process modifications, and equipment reconfiguration without major infrastructure changes. Flexibility demands increase as pharmaceutical companies pursue agile manufacturing strategies.
Sustainability considerations influence isolator design through energy-efficient systems, reduced environmental impact, and sustainable manufacturing practices. Pharmaceutical companies prioritize isolators that minimize energy consumption while maintaining performance standards. Environmental initiatives drive development of eco-friendly isolator technologies and operational practices.
Recent innovations in isolator technology include advanced decontamination systems utilizing vaporized hydrogen peroxide, ozone, and UV-C sterilization methods. These developments enhance decontamination effectiveness while reducing cycle times and operational costs. Sterilization advances enable pharmaceutical facilities to achieve higher throughput rates with maintained sterility assurance.
Regulatory updates from FDA, EMA, and other agencies continue shaping isolator requirements and validation protocols. Recent guidance documents emphasize risk-based approaches to contamination control and enhanced documentation requirements. MWR analysis indicates that regulatory evolution drives continuous improvement in isolator design and performance capabilities.
Strategic partnerships between isolator manufacturers and pharmaceutical companies facilitate development of customized solutions and integrated manufacturing systems. These collaborations enable innovation in isolator technology while addressing specific pharmaceutical manufacturing challenges. Partnership trends show increasing cooperation between equipment suppliers and end users.
Market consolidation activities include acquisitions, mergers, and strategic alliances that reshape competitive dynamics and expand technological capabilities. These developments create larger, more comprehensive solution providers capable of addressing diverse pharmaceutical manufacturing requirements. Industry restructuring continues influencing market competition and innovation directions.
Investment priorities should focus on isolator systems offering superior automation integration, comprehensive validation support, and flexible configuration options. Pharmaceutical companies should evaluate total cost of ownership including initial investment, operational costs, and lifecycle expenses when selecting isolator solutions. Strategic planning requires consideration of future manufacturing needs and technology evolution trends.
Technology adoption strategies should emphasize gradual implementation approaches that minimize operational disruption while maximizing benefits. Pilot projects and phased rollouts enable pharmaceutical companies to validate isolator performance before full-scale deployment. Implementation success depends on adequate planning, training, and change management processes.
Vendor selection criteria should prioritize suppliers offering comprehensive support services, proven track records, and ongoing innovation capabilities. Long-term partnerships with isolator manufacturers provide advantages in system optimization, upgrades, and technical support. Supplier relationships significantly influence isolator project success and operational performance.
Regulatory preparation requires proactive engagement with evolving guidelines and standards affecting isolator applications. Pharmaceutical companies should maintain awareness of regulatory trends and participate in industry discussions shaping future requirements. Compliance strategies must anticipate regulatory changes and ensure isolator systems meet emerging standards.
Growth projections indicate sustained expansion in the isolators in pharmaceutical market driven by increasing pharmaceutical manufacturing activities, regulatory requirements, and technology advancement. MarkWide Research forecasts suggest the market will experience robust growth with CAGR of 8.2% through the next decade, reflecting strong demand across pharmaceutical industry segments.
Technology evolution will continue advancing isolator capabilities through artificial intelligence integration, enhanced automation features, and improved user interfaces. Future isolator systems will offer greater intelligence, predictive capabilities, and seamless integration with pharmaceutical manufacturing execution systems. Innovation cycles will accelerate as manufacturers compete to introduce breakthrough technologies.
Market expansion opportunities exist in emerging pharmaceutical markets, personalized medicine applications, and specialized therapeutic areas requiring advanced containment solutions. Geographic diversification and application expansion will drive market growth beyond traditional pharmaceutical manufacturing segments. Diversification strategies will enable isolator manufacturers to capture new market opportunities.
Industry transformation through digitalization, sustainability initiatives, and regulatory evolution will reshape isolator market dynamics and competitive landscapes. Companies adapting to these changes while maintaining focus on core containment capabilities will achieve competitive advantages. Market leadership will depend on balancing innovation with proven performance in pharmaceutical applications.
The isolators in pharmaceutical market demonstrates strong growth potential driven by regulatory requirements, safety considerations, and technological advancement. Market dynamics favor solutions offering superior containment capabilities, automation integration, and comprehensive support services. Strategic opportunities exist for manufacturers developing innovative isolator technologies that address evolving pharmaceutical manufacturing needs.
Success factors in this market include maintaining regulatory compliance, delivering reliable performance, and providing exceptional customer support throughout isolator lifecycles. Companies positioning themselves as comprehensive solution providers with strong technical capabilities and service excellence will capture market leadership positions. Market evolution continues favoring isolator systems that enhance pharmaceutical manufacturing efficiency while ensuring strict contamination control.
Future market development will be shaped by continued pharmaceutical industry growth, regulatory evolution, and technology innovation. The isolators in pharmaceutical market represents a critical component of modern pharmaceutical manufacturing infrastructure, with sustained demand expected across global markets and diverse application areas.
What is an Isolator in Pharmaceutical?
Isolators in pharmaceutical contexts are controlled environments designed to prevent contamination during the manufacturing and handling of sterile products. They are crucial for maintaining aseptic conditions and are commonly used in the production of injectable drugs and biologics.
What are the key players in the Isolators in Pharmaceutical Market?
Key players in the Isolators in Pharmaceutical Market include companies like Getinge AB, STERIS plc, and Merck KGaA, which provide a range of isolator solutions for sterile manufacturing. These companies focus on innovation and compliance with stringent regulatory standards, among others.
What are the growth factors driving the Isolators in Pharmaceutical Market?
The growth of the Isolators in Pharmaceutical Market is driven by the increasing demand for sterile products, advancements in biopharmaceutical manufacturing, and the rising focus on contamination control. Additionally, the expansion of the pharmaceutical industry in emerging markets contributes to this growth.
What challenges does the Isolators in Pharmaceutical Market face?
Challenges in the Isolators in Pharmaceutical Market include high initial investment costs, the complexity of integration into existing manufacturing processes, and the need for ongoing maintenance and validation. These factors can hinder adoption, especially among smaller manufacturers.
What future opportunities exist in the Isolators in Pharmaceutical Market?
Future opportunities in the Isolators in Pharmaceutical Market include the development of more advanced isolator technologies, such as robotic automation and real-time monitoring systems. Additionally, the growing trend towards personalized medicine and biologics presents new avenues for isolator applications.
What trends are shaping the Isolators in Pharmaceutical Market?
Trends in the Isolators in Pharmaceutical Market include the increasing adoption of single-use isolators, advancements in materials that enhance sterility, and the integration of digital technologies for better process control. These trends are aimed at improving efficiency and compliance in pharmaceutical manufacturing.
Isolators in Pharmaceutical Market
| Segmentation Details | Description |
|---|---|
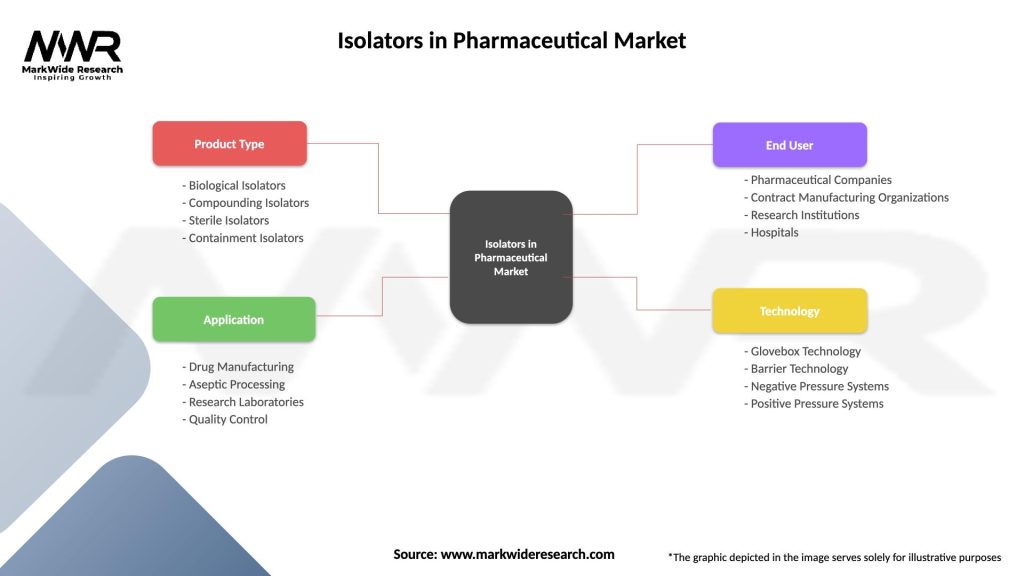
| Product Type | Biological Isolators, Compounding Isolators, Sterile Isolators, Containment Isolators |
| Application | Drug Manufacturing, Aseptic Processing, Research Laboratories, Quality Control |
| End User | Pharmaceutical Companies, Contract Manufacturing Organizations, Research Institutions, Hospitals |
| Technology | Glovebox Technology, Barrier Technology, Negative Pressure Systems, Positive Pressure Systems |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading companies in the Isolators in Pharmaceutical Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at