444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at

Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$2150
The India API industry market represents one of the most dynamic and rapidly expanding pharmaceutical sectors globally, establishing the country as a dominant force in active pharmaceutical ingredient manufacturing. India’s API sector has evolved from a nascent industry to becoming the world’s largest supplier of generic medicines and a significant contributor to global pharmaceutical supply chains. The market demonstrates exceptional growth potential, driven by increasing domestic healthcare demands, expanding export opportunities, and government initiatives promoting pharmaceutical manufacturing excellence.
Market dynamics indicate robust expansion with the industry experiencing a compound annual growth rate of 9.2%, reflecting strong domestic consumption patterns and international market penetration. The sector benefits from India’s cost-effective manufacturing capabilities, skilled workforce, and established regulatory frameworks that comply with international quality standards. Manufacturing excellence has positioned Indian API companies as preferred suppliers to pharmaceutical companies across developed and emerging markets.
Strategic positioning within the global pharmaceutical ecosystem has enabled India to capture approximately 20% of global generic API production, making it an indispensable component of worldwide healthcare supply chains. The industry’s growth trajectory is supported by increasing investments in research and development, capacity expansion initiatives, and technological advancement programs that enhance production efficiency and product quality.
The India API industry market refers to the comprehensive ecosystem encompassing the research, development, manufacturing, and distribution of active pharmaceutical ingredients within India’s pharmaceutical sector. Active pharmaceutical ingredients represent the biologically active components in medications that produce therapeutic effects, forming the foundation of all pharmaceutical formulations and drug products.
Industry scope encompasses various API categories including synthetic APIs, biotechnology-derived APIs, herbal and natural APIs, and specialty pharmaceutical ingredients. The market includes contract manufacturing organizations, integrated pharmaceutical companies, dedicated API manufacturers, and research-focused biotechnology firms that contribute to India’s pharmaceutical manufacturing capabilities.
Market participants range from large multinational corporations with Indian operations to domestic companies specializing in specific therapeutic areas or manufacturing processes. The industry serves both domestic pharmaceutical companies producing generic and branded medications and international clients requiring cost-effective, high-quality API supplies for global pharmaceutical manufacturing.
India’s API industry stands as a cornerstone of the global pharmaceutical supply chain, demonstrating remarkable resilience and growth potential across diverse therapeutic segments. The market’s expansion is characterized by increasing production capacities, technological innovations, and strategic partnerships that enhance India’s position as a preferred API manufacturing destination.
Key growth drivers include rising healthcare expenditure, expanding generic drug adoption globally, and government initiatives supporting pharmaceutical manufacturing through policy reforms and infrastructure development. The industry benefits from cost advantages of approximately 40-50% compared to developed market manufacturers, making Indian APIs highly competitive in international markets.
Market segmentation reveals strong performance across cardiovascular, anti-diabetic, anti-infective, and central nervous system therapeutic areas, with emerging opportunities in oncology, immunology, and biotechnology-derived APIs. The sector’s diversification strategy has reduced dependency on specific therapeutic areas while expanding market reach across multiple pharmaceutical segments.
Future prospects indicate continued expansion driven by increasing global pharmaceutical demand, patent expiries of branded medications, and India’s growing capabilities in complex API manufacturing including biotechnology products and specialized pharmaceutical ingredients.
Strategic market insights reveal several critical factors shaping India’s API industry landscape and future growth trajectory:
Primary market drivers propelling India’s API industry growth encompass both domestic and international factors that create sustained demand for Indian pharmaceutical ingredients.
Healthcare infrastructure expansion across India drives domestic API consumption as the country’s pharmaceutical market grows to serve an expanding population with increasing healthcare access. Government healthcare initiatives, insurance coverage expansion, and rising health awareness contribute to steady domestic demand growth for pharmaceutical products requiring API inputs.
Global generic drug adoption represents a significant growth driver as healthcare systems worldwide seek cost-effective alternatives to branded medications. Patent expiries of major pharmaceutical products create opportunities for generic drug manufacturers who rely on Indian APIs for cost-competitive production. Generic drug penetration continues expanding globally, with adoption rates reaching 85% in developed markets.
Manufacturing cost advantages position Indian API producers favorably against international competitors, enabling competitive pricing while maintaining profit margins. Lower labor costs, established infrastructure, and economies of scale contribute to India’s cost leadership in API manufacturing.
Government policy support through initiatives like the Production Linked Incentive scheme, pharmaceutical parks development, and regulatory streamlining creates favorable conditions for industry expansion and investment attraction.
Regulatory compliance challenges pose significant constraints on API industry growth, requiring substantial investments in quality systems, documentation, and facility upgrades to meet evolving international standards. Regulatory complexity across different markets demands specialized expertise and continuous monitoring of changing requirements.
Raw material dependency on imports, particularly from China, creates supply chain vulnerabilities and cost fluctuations that impact production planning and profitability. Supply chain disruptions can significantly affect manufacturing schedules and customer commitments, highlighting the need for supply source diversification.
Environmental compliance costs continue increasing as regulatory authorities implement stricter environmental standards for pharmaceutical manufacturing. Waste treatment, emission control, and sustainable manufacturing practices require ongoing investments that impact operational costs.
Intense price competition in generic API markets pressures profit margins and requires continuous efficiency improvements to maintain competitiveness. Price erosion in mature API segments challenges companies to develop higher-value products and improve operational efficiency.
Skilled workforce shortages in specialized areas like biotechnology, regulatory affairs, and advanced manufacturing technologies limit industry expansion capabilities and innovation potential.
Biotechnology API development presents substantial growth opportunities as the global pharmaceutical industry increasingly focuses on biologics, biosimilars, and personalized medicine. India’s growing biotechnology capabilities position the country to capture significant market share in this high-value segment.
Complex generic APIs offer opportunities for higher margins and reduced competition compared to commodity APIs. Companies developing capabilities in complex molecules, controlled substances, and specialized formulations can command premium pricing and establish competitive advantages.
Contract development and manufacturing services represent expanding opportunities as global pharmaceutical companies seek to outsource API development and production to focus on core competencies. CDMO market growth is projected at 12.5% annually, creating significant opportunities for Indian companies.
Emerging market expansion in Africa, Latin America, and Southeast Asia provides new growth avenues for Indian API exporters. These markets offer less competitive environments and growing pharmaceutical demand driven by improving healthcare access.
Backward integration opportunities in key starting materials and intermediates can reduce supply chain dependencies while improving cost structures and supply security. Companies investing in upstream capabilities can achieve better control over production costs and quality.
Market dynamics within India’s API industry reflect complex interactions between global pharmaceutical trends, regulatory developments, and competitive pressures that shape industry evolution and strategic decision-making.
Supply-demand equilibrium varies significantly across different API categories, with commodity APIs experiencing oversupply and price pressure while specialized and complex APIs maintain stronger pricing power. Market consolidation trends are evident as larger companies acquire smaller players to achieve scale advantages and expand product portfolios.
Technology adoption is accelerating across the industry, with companies investing in continuous manufacturing, process analytical technology, and digitalization initiatives to improve efficiency and quality. Automation implementation has increased productivity by 25-30% in leading facilities while reducing operational costs.
Customer relationship evolution shows increasing preference for long-term partnerships over transactional relationships, with pharmaceutical companies seeking reliable suppliers who can provide consistent quality, regulatory support, and supply security. Strategic partnerships are becoming more common as companies seek to reduce supply chain risks.
Innovation cycles are shortening as companies invest more heavily in research and development to develop next-generation APIs and manufacturing processes. R&D investment levels have increased to 8-12% of revenues among leading API companies, reflecting the importance of innovation in maintaining competitive positions.
Comprehensive research methodology employed in analyzing India’s API industry market combines primary and secondary research approaches to ensure accurate, reliable, and actionable market insights. The methodology encompasses quantitative data analysis, qualitative assessments, and industry expert consultations to provide holistic market understanding.
Primary research activities include structured interviews with industry executives, regulatory officials, and market participants across the API value chain. Survey methodologies capture quantitative data on market trends, growth projections, and competitive dynamics from representative industry samples.
Secondary research sources encompass government publications, industry reports, regulatory filings, company annual reports, and trade association data to validate primary findings and provide comprehensive market context. Data triangulation ensures accuracy and reliability of market insights through multiple source verification.
Analytical frameworks include market sizing methodologies, competitive positioning analysis, and trend identification techniques that provide strategic insights for industry participants. MarkWide Research employs proprietary analytical models to assess market dynamics and forecast future trends with high accuracy levels.
Quality assurance processes ensure data accuracy through multiple validation stages, expert review panels, and continuous monitoring of market developments that might impact research findings and conclusions.
Regional distribution of India’s API industry reveals significant geographic concentration in specific states that offer favorable manufacturing conditions, infrastructure access, and regulatory support for pharmaceutical operations.
Gujarat state leads API manufacturing with approximately 35% market share, benefiting from excellent port connectivity, industrial infrastructure, and supportive state policies. The region hosts major API manufacturing clusters in Ahmedabad, Vadodara, and Ankleshwar, providing comprehensive ecosystem support for pharmaceutical companies.
Andhra Pradesh and Telangana collectively account for 25% of national API production, with Hyderabad emerging as a major biotechnology and pharmaceutical hub. The region’s focus on biotechnology APIs and research-intensive manufacturing has attracted significant investments from domestic and international companies.
Maharashtra contributes approximately 20% of API production, with Mumbai and Pune serving as major pharmaceutical centers. The state’s proximity to major ports, skilled workforce availability, and established pharmaceutical ecosystem support continued industry growth.
Tamil Nadu represents 10% of national production, with Chennai serving as a key manufacturing and export hub. The state’s focus on biotechnology and specialty chemicals complements its API manufacturing capabilities.
Other regions including Karnataka, West Bengal, and Himachal Pradesh contribute the remaining 10% of production, with each state developing specialized capabilities in specific API categories or manufacturing processes.
Competitive landscape within India’s API industry features a diverse mix of large integrated pharmaceutical companies, specialized API manufacturers, and emerging biotechnology firms that compete across different market segments and therapeutic areas.
Market competition intensifies as companies invest in capacity expansion, technology upgrades, and product portfolio diversification to maintain competitive positions and capture emerging opportunities in high-growth segments.
Market segmentation of India’s API industry reveals diverse categories based on therapeutic applications, manufacturing processes, and target markets that demonstrate varying growth patterns and competitive dynamics.
By Therapeutic Area:
By Manufacturing Process:
By End Market:
Cardiovascular APIs maintain market leadership due to high global prevalence of cardiovascular diseases and established manufacturing expertise among Indian companies. Market maturity in this segment drives focus on cost optimization and manufacturing efficiency improvements rather than premium pricing strategies.
Anti-diabetic APIs demonstrate exceptional growth potential as diabetes prevalence increases globally, particularly in emerging markets. Indian manufacturers benefit from cost advantages and established production capabilities in key molecules like metformin, glimepiride, and newer generation diabetes medications.
Oncology APIs represent the highest-value segment with premium pricing potential due to complex manufacturing requirements and limited competition. Companies investing in oncology API capabilities can achieve significantly higher margins compared to commodity pharmaceutical ingredients.
Biotechnology APIs emerge as a transformative category requiring substantial investments in specialized manufacturing facilities, skilled workforce, and regulatory expertise. Biosimilar API development offers opportunities to participate in the growing biologics market with potentially high returns on investment.
Anti-infective APIs maintain steady demand driven by ongoing global health challenges and antibiotic resistance concerns that create opportunities for new molecule development. The segment benefits from India’s established expertise in fermentation and synthetic chemistry.
Manufacturing companies benefit from India’s cost-competitive production environment, skilled workforce availability, and established supply chain infrastructure that enable efficient API production and global market access. Operational advantages include lower manufacturing costs, flexible production capabilities, and proximity to key raw material sources.
Pharmaceutical customers gain access to high-quality APIs at competitive prices, reliable supply chains, and comprehensive regulatory support that facilitates product development and market entry. Partnership benefits include reduced procurement costs, supply security, and access to specialized manufacturing capabilities.
Investors find attractive opportunities in a growing industry with strong fundamentals, increasing global demand, and government policy support. Investment advantages include market growth potential, export earnings, and opportunities for value creation through operational improvements and market expansion.
Government stakeholders benefit from increased export earnings, employment generation, and technology development that contribute to economic growth and industrial development objectives. Economic benefits include foreign exchange earnings, tax revenues, and industrial ecosystem development.
Healthcare systems globally benefit from access to affordable, high-quality APIs that enable cost-effective pharmaceutical production and improved healthcare accessibility. System advantages include reduced medication costs, supply security, and access to essential medicines.
Strengths:
Weaknesses:
Opportunities:
Threats:
Digitalization adoption accelerates across API manufacturing as companies implement advanced process control systems, data analytics, and automation technologies to improve efficiency and quality. Digital transformation initiatives focus on predictive maintenance, real-time quality monitoring, and supply chain optimization.
Sustainability focus intensifies as companies invest in green chemistry, waste reduction, and environmental compliance to meet evolving regulatory requirements and customer expectations. Sustainable manufacturing practices become competitive advantages in securing long-term customer relationships.
Supply chain localization trends emerge as companies seek to reduce dependencies on single-source suppliers and geographic concentrations. Supply diversification strategies include backward integration, alternative supplier development, and strategic inventory management.
Quality by design principles gain adoption as manufacturers implement systematic approaches to product development and manufacturing that ensure consistent quality and regulatory compliance. QbD implementation improves process understanding and reduces manufacturing risks.
Continuous manufacturing technologies attract increasing interest as companies seek to improve efficiency, reduce costs, and enhance quality control compared to traditional batch manufacturing processes. Process innovation drives competitive advantages in cost and quality performance.
Government policy initiatives including the Production Linked Incentive scheme provide substantial support for API manufacturing capacity expansion and technology upgrades. Policy support focuses on reducing import dependencies and strengthening domestic pharmaceutical supply chains.
Capacity expansion projects across major API manufacturers indicate strong confidence in market growth prospects and increasing global demand. Investment commitments total substantial amounts directed toward new facility construction and existing facility upgrades.
Strategic partnerships between Indian API companies and global pharmaceutical firms create opportunities for technology transfer, market access, and collaborative product development. Partnership agreements focus on long-term supply arrangements and joint development programs.
Regulatory approvals for new manufacturing facilities and product registrations demonstrate continued industry expansion and market access improvements. Approval trends indicate growing acceptance of Indian API manufacturing capabilities by international regulatory authorities.
Technology investments in biotechnology capabilities, continuous manufacturing, and advanced analytics position Indian companies for participation in next-generation pharmaceutical manufacturing. Innovation focus drives competitive positioning in high-value market segments.
Strategic recommendations for API industry participants focus on building sustainable competitive advantages through innovation, quality excellence, and market diversification strategies that address evolving industry dynamics.
Investment priorities should emphasize biotechnology capabilities development, complex generic APIs, and backward integration initiatives that reduce supply chain dependencies while creating higher-value product portfolios. MarkWide Research analysis indicates that companies investing in these areas achieve superior growth rates of 15-20% compared to industry averages.
Market expansion strategies should target emerging markets with growing pharmaceutical demand while maintaining strong positions in established markets through quality excellence and customer relationship management. Geographic diversification reduces market concentration risks and provides growth opportunities.
Operational excellence initiatives including digitalization, automation, and continuous improvement programs are essential for maintaining cost competitiveness and quality standards in increasingly competitive markets. Efficiency improvements directly impact profitability and market positioning.
Regulatory compliance investments remain critical for maintaining market access and avoiding disruptions that can significantly impact business operations and customer relationships. Compliance excellence becomes a competitive advantage in securing long-term customer partnerships.
Long-term prospects for India’s API industry remain highly positive, driven by increasing global pharmaceutical demand, patent expiries creating generic opportunities, and India’s strengthening position as a preferred manufacturing destination for complex pharmaceutical ingredients.
Growth projections indicate sustained expansion at compound annual growth rates exceeding 10% over the next decade, supported by domestic market growth, export expansion, and increasing participation in high-value pharmaceutical segments. Market evolution toward more sophisticated products and manufacturing processes creates opportunities for premium pricing and improved profitability.
Technology advancement will continue driving industry transformation as companies adopt continuous manufacturing, biotechnology capabilities, and digital technologies that improve efficiency and enable participation in next-generation pharmaceutical manufacturing. Innovation investment becomes increasingly critical for maintaining competitive positions.
Regulatory landscape evolution toward more stringent quality requirements and environmental standards will favor companies with strong compliance capabilities and operational excellence. Regulatory leadership creates competitive advantages in securing market access and customer relationships.
Market consolidation trends may accelerate as companies seek scale advantages, technology capabilities, and market access through strategic acquisitions and partnerships. Industry structure evolution toward larger, more capable organizations will drive competitive dynamics and market leadership positions.
India’s API industry market represents a dynamic and rapidly evolving sector that has established itself as a critical component of the global pharmaceutical supply chain. The industry’s remarkable growth trajectory, supported by cost advantages, quality capabilities, and government policy support, positions India as a dominant force in global API manufacturing.
Market fundamentals remain strong with increasing global pharmaceutical demand, patent expiries creating generic opportunities, and India’s expanding capabilities in complex and biotechnology APIs. The industry’s evolution toward higher-value products and advanced manufacturing processes creates opportunities for sustained growth and improved profitability.
Strategic positioning for future success requires continued investment in innovation, quality excellence, and operational efficiency while addressing challenges related to supply chain dependencies and regulatory compliance. Companies that successfully navigate these dynamics while capitalizing on emerging opportunities in biotechnology and complex generics will achieve superior performance.
Industry outlook indicates continued expansion and evolution as India strengthens its position as the world’s preferred API manufacturing destination, contributing to global healthcare accessibility while driving economic growth and industrial development within the country.
What is API?
API, or Application Programming Interface, refers to a set of rules and protocols for building and interacting with software applications. In the context of the India API industry, it enables different software systems to communicate and share data seamlessly.
What are the key players in the India API Industry Market?
Key players in the India API Industry Market include companies like Postman, which provides API development tools, and Razorpay, known for its payment gateway APIs. Other notable companies include Zoho and Freshworks, among others.
What are the growth factors driving the India API Industry Market?
The growth of the India API Industry Market is driven by the increasing demand for digital transformation, the rise of cloud computing, and the need for seamless integration of applications across various sectors such as finance, healthcare, and e-commerce.
What challenges does the India API Industry Market face?
The India API Industry Market faces challenges such as security concerns related to data breaches, the complexity of API management, and the need for standardization across different platforms and services.
What opportunities exist in the India API Industry Market?
Opportunities in the India API Industry Market include the growing adoption of microservices architecture, the expansion of IoT applications, and the increasing focus on automation and AI-driven solutions across various industries.
What trends are shaping the India API Industry Market?
Trends shaping the India API Industry Market include the rise of serverless computing, the increasing use of RESTful APIs, and the growing emphasis on API security and management solutions to enhance performance and reliability.
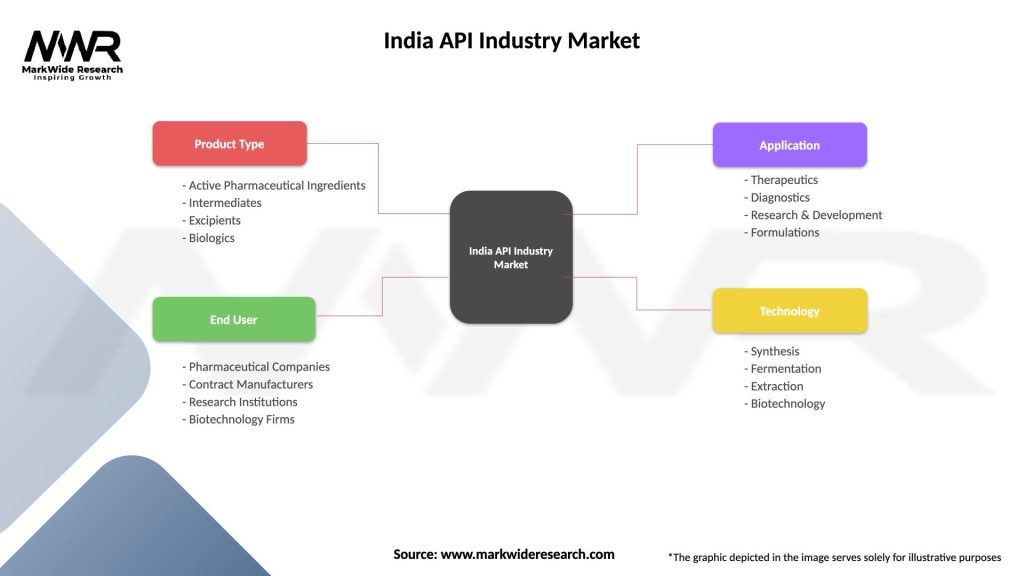
India API Industry Market
| Segmentation Details | Description |
|---|---|
| Product Type | Active Pharmaceutical Ingredients, Intermediates, Excipients, Biologics |
| End User | Pharmaceutical Companies, Contract Manufacturers, Research Institutions, Biotechnology Firms |
| Application | Therapeutics, Diagnostics, Research & Development, Formulations |
| Technology | Synthesis, Fermentation, Extraction, Biotechnology |
Please note: The segmentation can be entirely customized to align with our client’s needs.
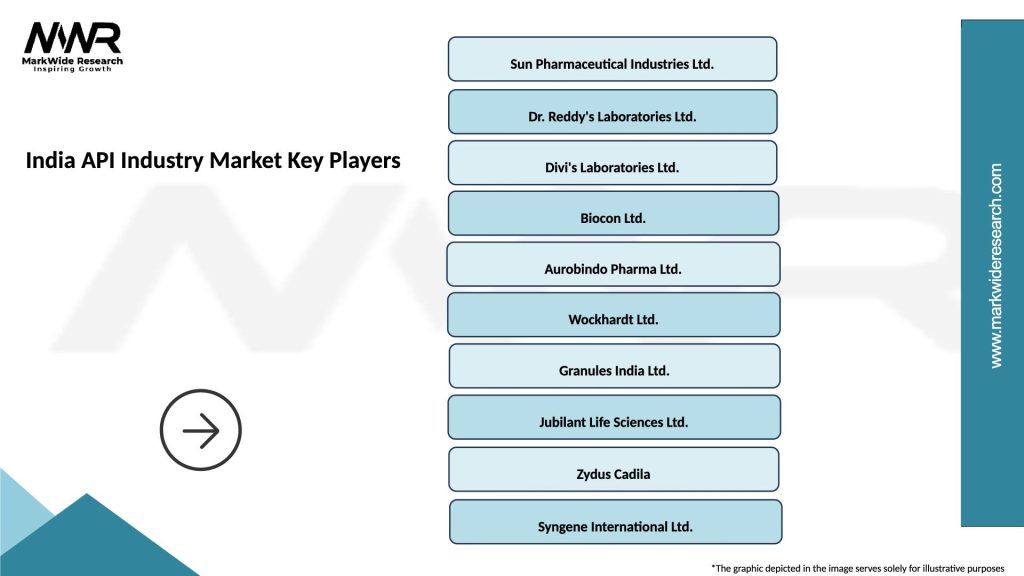
Leading companies in the India API Industry Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at