444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The Human Thrombin market is a vital segment of the global biopharmaceutical industry. Thrombin is a crucial enzyme involved in blood coagulation, and its use has found extensive applications in various medical procedures, particularly in surgeries and wound healing. Human thrombin is derived from human plasma and is considered superior to bovine-derived thrombin due to its lower immunogenicity. The rising prevalence of cardiovascular diseases, increasing surgical procedures, and a growing aging population have driven the demand for human thrombin products. This market overview provides insights into the key aspects of the Human Thrombin market and its potential for growth.
Meaning
Human thrombin is a serine protease enzyme that plays a crucial role in the blood coagulation cascade. It is involved in converting fibrinogen to fibrin, an essential step in forming blood clots to stop bleeding. This process, known as thrombin clotting time (TCT), is widely used in medical procedures such as surgeries, wound healing, and hemostasis management. Human thrombin is commercially available in various forms, including topical, powder, and solution, making it suitable for different clinical applications.
Executive Summary
The Human Thrombin market has witnessed significant growth in recent years due to its widespread use in medical procedures. With the increasing number of surgical interventions and a rising aging population, the demand for human thrombin products is expected to surge in the forecast period. This report aims to provide comprehensive insights into the market, including key drivers, restraints, opportunities, and market dynamics, along with a detailed analysis of regional trends, competitive landscape, and key industry developments.
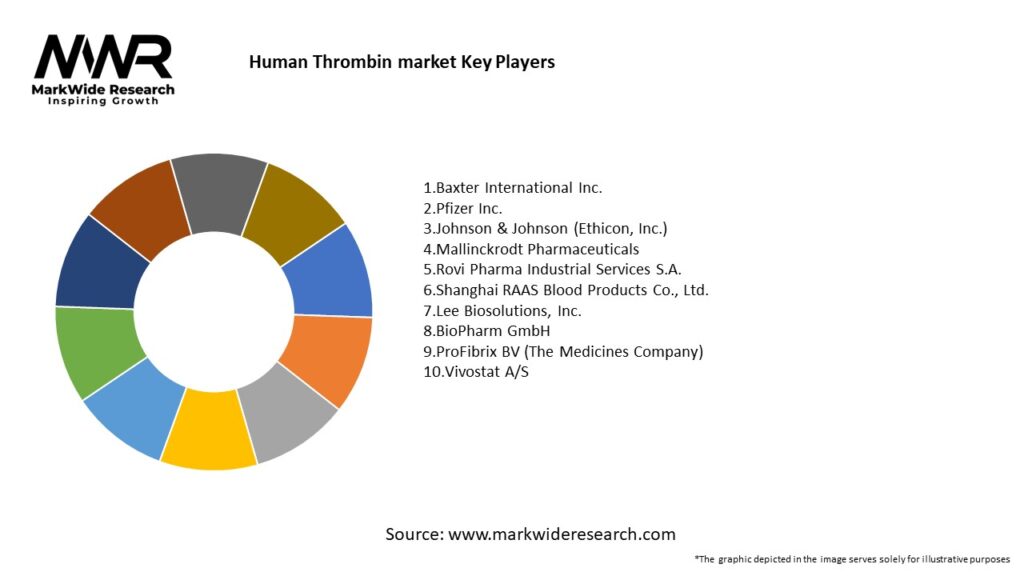
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
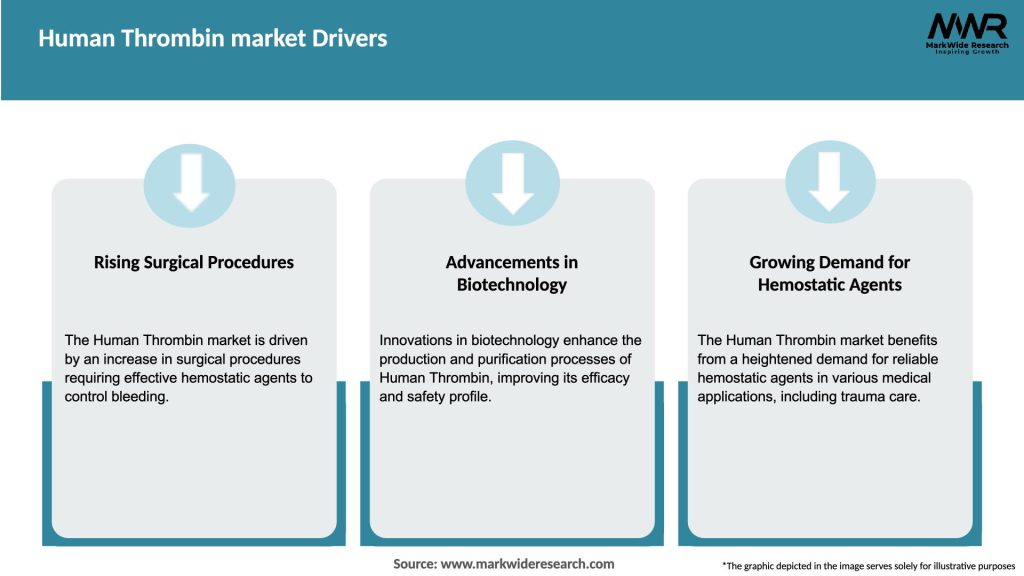
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The Human Thrombin market is dynamic and influenced by various factors, including technological advancements, changing medical practices, and market competition. The demand for human thrombin is primarily driven by its applications in surgeries, wound healing, and trauma care. The market’s growth is further propelled by the rising prevalence of cardiovascular diseases and the aging population. However, stringent regulatory approvals and the risk of adverse reactions could hinder market growth. To overcome these challenges, industry players should focus on continuous research and development, expanding into emerging markets, and forming strategic collaborations to leverage market opportunities effectively.
Regional Analysis
The Human Thrombin market exhibits a strong global presence, with North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa being the key regions driving market growth. North America, led by the United States, dominates the market due to the high prevalence of cardiovascular diseases and a well-established healthcare infrastructure. Europe follows closely, with significant contributions from countries like Germany, France, and the United Kingdom. The Asia Pacific region is expected to witness rapid growth during the forecast period, driven by increasing healthcare investments, rising surgical procedures, and a large patient pool. Latin America and the Middle East & Africa present untapped potential for market players to explore new growth opportunities.
Competitive Landscape
Leading Companies in the Human Thrombin Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
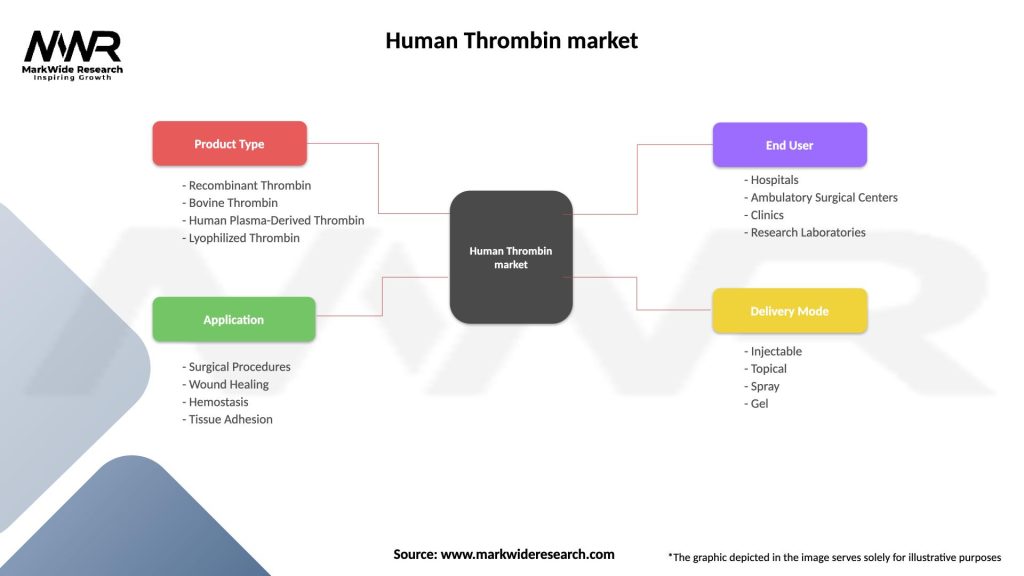
Segmentation
The Human Thrombin market can be segmented based on product type, application, and end-user.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Covid-19 Impact
The COVID-19 pandemic had a multifaceted impact on the Human Thrombin market. While the healthcare system faced overwhelming challenges, elective surgeries and medical procedures were delayed or canceled, leading to a temporary dip in the demand for human thrombin products. However, as the healthcare sector resumed normal operations and surgeries, the market witnessed a rebound in demand. The pandemic also highlighted the importance of effective hemostatic agents like human thrombin in managing emergency medical situations.
Key Industry Developments
Analyst Suggestions
Future Outlook
The Human Thrombin market is expected to experience steady growth in the coming years, driven by factors such as an aging population, increasing surgical procedures, and growing demand for advanced hemostatic agents. The market’s growth will be further bolstered by ongoing research and development efforts to introduce innovative human thrombin products. Strategic collaborations and expansions into emerging markets will play a crucial role in shaping the market’s future landscape.
Conclusion
The Human Thrombin market serves as a critical component of the biopharmaceutical industry, offering effective solutions for blood coagulation management in various medical applications. With increasing surgical interventions, trauma cases, and wound healing requirements, the demand for human thrombin is expected to surge in the forecast period. However, market players must navigate challenges related to safety concerns, cost-effectiveness, and competition from alternative hemostatic agents. By capitalizing on emerging opportunities, investing in research and development, and forming strategic partnerships, stakeholders can unlock the market’s potential and contribute to improved patient outcomes in the medical field.
What is Human Thrombin?
Human Thrombin is a serine protease enzyme that plays a crucial role in the coagulation process, converting fibrinogen to fibrin, which is essential for blood clot formation. It is used in various medical applications, including surgical procedures and wound healing.
What are the key companies in the Human Thrombin market?
Key companies in the Human Thrombin market include Baxter International, Johnson & Johnson, and Grifols, among others. These companies are involved in the development and production of thrombin products for clinical use.
What are the growth factors driving the Human Thrombin market?
The Human Thrombin market is driven by the increasing number of surgical procedures, the rising prevalence of bleeding disorders, and advancements in medical technology. Additionally, the growing demand for effective hemostatic agents in surgeries contributes to market growth.
What challenges does the Human Thrombin market face?
The Human Thrombin market faces challenges such as regulatory hurdles, the risk of contamination in blood-derived products, and competition from synthetic alternatives. These factors can impact product availability and market dynamics.
What opportunities exist in the Human Thrombin market?
Opportunities in the Human Thrombin market include the development of novel thrombin formulations and the expansion of applications in minimally invasive surgeries. Additionally, increasing investments in research and development present avenues for growth.
What trends are shaping the Human Thrombin market?
Trends in the Human Thrombin market include a shift towards personalized medicine and the integration of biotechnology in thrombin production. Furthermore, there is a growing focus on sustainability and ethical sourcing of blood products.
Human Thrombin market
| Segmentation Details | Description |
|---|---|
| Product Type | Recombinant Thrombin, Bovine Thrombin, Human Plasma-Derived Thrombin, Lyophilized Thrombin |
| Application | Surgical Procedures, Wound Healing, Hemostasis, Tissue Adhesion |
| End User | Hospitals, Ambulatory Surgical Centers, Clinics, Research Laboratories |
| Delivery Mode | Injectable, Topical, Spray, Gel |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Human Thrombin Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at