444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The human acellular vessels (HAVs) market is at the forefront of regenerative medicine, offering innovative solutions for vascular reconstruction and replacement. These acellular vessels are derived from human donor tissues, processed to remove cellular components while preserving the extracellular matrix (ECM) structure. HAVs hold immense potential in various clinical applications, including vascular surgery, dialysis access, and tissue engineering. As the demand for durable, biocompatible vascular grafts continues to rise, the HAVs market is experiencing significant growth and technological advancements.
Meaning
Human acellular vessels (HAVs) represent a novel approach to vascular grafts, leveraging the natural ECM architecture of human tissues to provide biocompatible and durable conduits for vascular reconstruction. By removing cellular components, HAVs minimize the risk of immune rejection and thrombosis while maintaining structural integrity and promoting tissue regeneration. These acellular vessels offer advantages such as improved patency rates, reduced infection risk, and enhanced biocompatibility compared to synthetic grafts, making them a promising alternative for patients requiring vascular interventions.
Executive Summary
The global human acellular vessels (HAVs) market is witnessing rapid growth driven by factors such as increasing prevalence of vascular diseases, growing demand for biocompatible graft materials, and advancements in tissue engineering technology. Key market players are investing in research and development to expand product offerings, improve manufacturing processes, and address unmet clinical needs. Regulatory approvals, clinical trials, and strategic collaborations are shaping market dynamics and driving innovation in the HAVs market.

Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The human acellular vessels (HAVs) market is characterized by dynamic factors such as technological innovation, market competition, regulatory compliance, and customer preferences, which influence market dynamics and shape industry trends. These dynamics drive product development, market segmentation, and strategic partnerships among manufacturers, distributors, and end-users, shaping the competitive landscape and market positioning of HAVs in the global regenerative medicine market.
Regional Analysis
The global human acellular vessels (HAVs) market is segmented into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa. North America and Europe lead the market in terms of market share and revenue due to well-established healthcare infrastructure, regulatory frameworks, and clinical adoption of HAVs in vascular surgery and tissue engineering applications. However, Asia Pacific is expected to witness rapid growth in the coming years due to increasing healthcare investments, rising prevalence of chronic diseases, and growing demand for innovative regenerative medicine solutions in emerging economies such as China, India, and Southeast Asia.
Competitive Landscape
Leading Companies in Human Acellular Vessels (HAVs) Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Segmentation
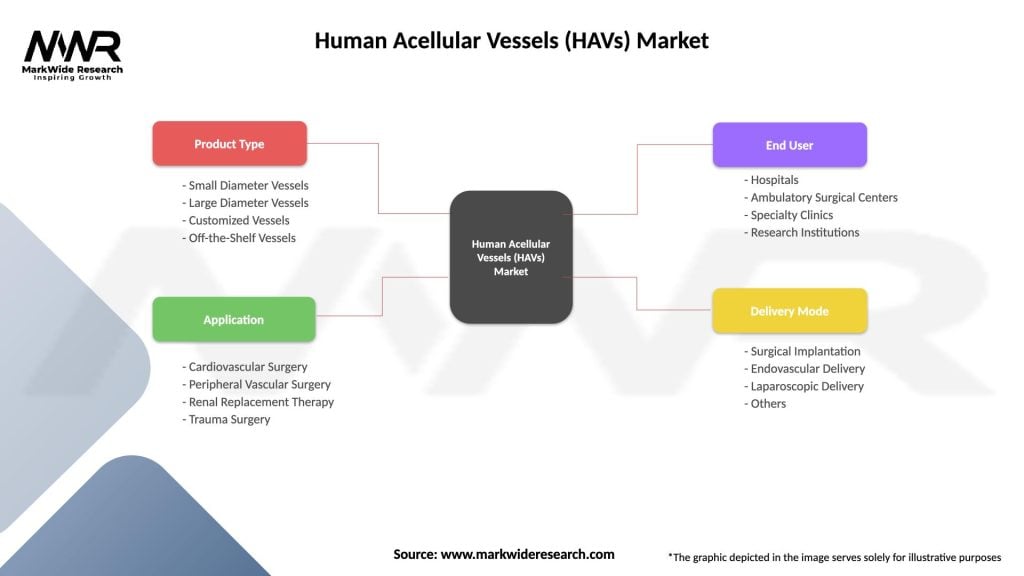
The human acellular vessels (HAVs) market can be segmented based on product type, application, end-user, and geography. By product type, the market includes vascular grafts, conduits, tissue-engineered constructs, and other HAVs products. By application, the market encompasses vascular surgery, dialysis access, tissue engineering, and regenerative medicine. By end-user, the market comprises hospitals, clinics, ambulatory surgical centers, and research laboratories.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Market Key Trends
Covid-19 Impact
The Covid-19 pandemic has had a significant impact on the human acellular vessels (HAVs) market, disrupting supply chains, clinical workflows, and research activities in regenerative medicine. The pandemic has underscored the importance of resilient healthcare systems, innovative medical technologies, and collaborative research efforts in addressing global health challenges and emerging infectious threats. Despite initial disruptions and delays in clinical trials, regulatory approvals, and product launches, the HAVs market has rebounded with renewed focus on pandemic preparedness, healthcare resilience, and digital health solutions. Telemedicine, remote monitoring, and virtual clinical trials have emerged as essential tools for delivering healthcare services, conducting research, and accelerating innovation in regenerative medicine, driving market growth and adoption of HAVs in vascular surgery, tissue engineering, and regenerative medicine applications.
Key Industry Developments
Analyst Suggestions
Future Outlook
The future outlook for the human acellular vessels (HAVs) market is promising, with continued growth expected in response to increasing demand for biocompatible vascular grafts, rising prevalence of vascular diseases, and advancements in tissue engineering technology. Key drivers such as personalized medicine, precision engineering, and next-generation biomaterials shape market dynamics and drive investment in research, development, and commercialization of HAVs products for vascular surgery, tissue engineering, and regenerative medicine applications. Looking ahead, market players are poised to capitalize on emerging opportunities in telehealth, digital health integration, and regenerative medicine to drive innovation, market expansion, and value creation in the dynamic and evolving HAVs market.
Conclusion
In conclusion, the human acellular vessels (HAVs) market represents a transformative approach to vascular reconstruction and tissue engineering, offering innovative solutions for patients with vascular diseases, chronic kidney disease, and tissue defects. HAVs provide biocompatible, durable conduits for vascular surgery, dialysis access, and regenerative medicine applications, leveraging the natural ECM architecture of human tissues to promote tissue integration, remodeling, and functional recovery in vivo. Market players are focused on innovation, collaboration, and regulatory compliance to address unmet clinical needs, expand market access, and drive value creation in the dynamic and evolving HAVs market. By investing in innovation, strengthening regulatory compliance, and fostering collaboration with healthcare stakeholders, market players can drive sustainable growth and market leadership in the competitive and rapidly evolving human acellular vessels (HAVs) market.
What is Human Acellular Vessels?
Human Acellular Vessels (HAVs) are bioengineered vascular grafts designed to replace or repair damaged blood vessels. They are made from human tissue and are intended to promote natural healing and integration within the body.
What are the key players in the Human Acellular Vessels (HAVs) Market?
Key players in the Human Acellular Vessels (HAVs) Market include companies like Humacyte, Inc., Terumo Corporation, and Corline Biomedical, among others. These companies are involved in the development and commercialization of HAVs for various medical applications.
What are the growth factors driving the Human Acellular Vessels (HAVs) Market?
The Human Acellular Vessels (HAVs) Market is driven by factors such as the increasing prevalence of cardiovascular diseases, advancements in tissue engineering, and the growing demand for minimally invasive surgical procedures.
What challenges does the Human Acellular Vessels (HAVs) Market face?
Challenges in the Human Acellular Vessels (HAVs) Market include regulatory hurdles, high manufacturing costs, and the need for extensive clinical trials to ensure safety and efficacy.
What opportunities exist in the Human Acellular Vessels (HAVs) Market?
Opportunities in the Human Acellular Vessels (HAVs) Market include the potential for expanding applications in regenerative medicine, partnerships with healthcare providers, and innovations in graft design and manufacturing processes.
What trends are shaping the Human Acellular Vessels (HAVs) Market?
Trends in the Human Acellular Vessels (HAVs) Market include the increasing focus on personalized medicine, the integration of advanced biomaterials, and the rise of bioprinting technologies for creating customized vascular grafts.
Human Acellular Vessels (HAVs) Market
| Segmentation Details | Description |
|---|---|
| Product Type | Small Diameter Vessels, Large Diameter Vessels, Customized Vessels, Off-the-Shelf Vessels |
| Application | Cardiovascular Surgery, Peripheral Vascular Surgery, Renal Replacement Therapy, Trauma Surgery |
| End User | Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Research Institutions |
| Delivery Mode | Surgical Implantation, Endovascular Delivery, Laparoscopic Delivery, Others |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at