444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
The HPV Testing market is a rapidly growing sector in the healthcare industry, driven by the increasing prevalence of Human Papillomavirus (HPV) infections worldwide. HPV is a sexually transmitted infection that can lead to various types of cancers, including cervical, anal, vaginal, and oropharyngeal cancer. HPV testing plays a crucial role in early detection and prevention of these cancers, making it an essential component of women’s healthcare.
HPV testing involves the identification and analysis of HPV DNA or RNA in the patient’s sample, such as cervical swabs or biopsies. This diagnostic test helps detect the presence of high-risk HPV strains that are more likely to cause cancer. By detecting HPV infections early, healthcare providers can initiate appropriate interventions and monitor patients for any signs of precancerous or cancerous changes.
Executive Summary
The HPV Testing market has witnessed significant growth over the past few years, driven by rising awareness about HPV-related cancers and the importance of early detection. The market is characterized by the presence of several key players offering a wide range of HPV testing products and services. The increasing adoption of HPV testing in routine screening programs, advancements in testing technologies, and government initiatives promoting cervical cancer prevention have contributed to the market’s expansion.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
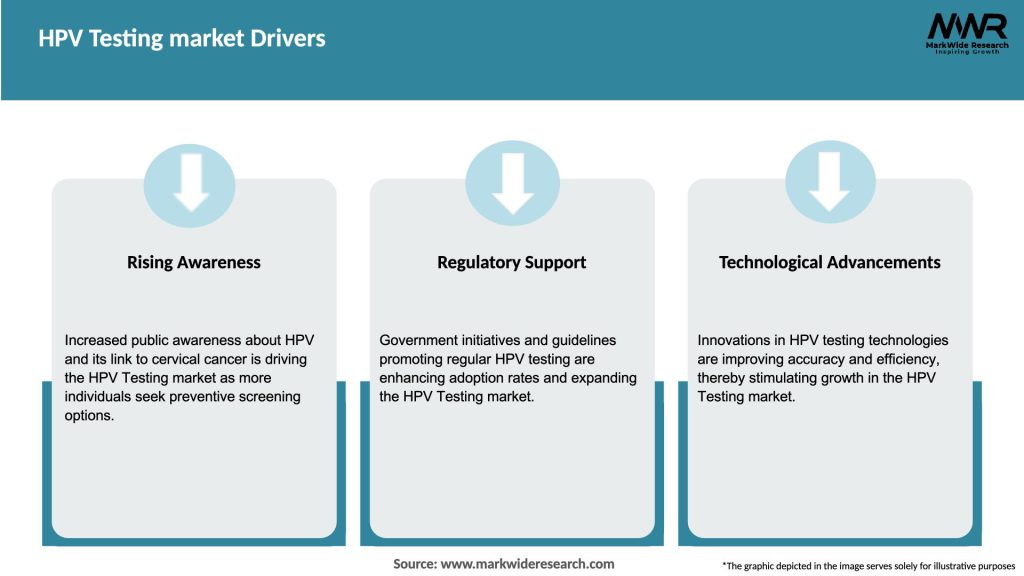
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The HPV Testing market is highly dynamic, driven by various factors such as increasing awareness, technological advancements, government initiatives, and market competition. The market is characterized by intense competition among key players, resulting in ongoing product innovation and the launch of advanced testing platforms. The evolving regulatory landscape and changing reimbursement policies also influence market dynamics. Moreover, the COVID-19 pandemic has had an impact on the market, leading to changes in testing patterns and priorities.
Regional Analysis
The HPV Testing market exhibits regional variations in terms of market size, testing rates, and adoption of different testing methods. North America and Europe dominate the market due to well-established screening programs, higher awareness levels, and favorable reimbursement policies. Asia Pacific is a rapidly growing market, driven by increasing awareness and government initiatives to tackle cervical cancer. Latin America and Africa present opportunities for market expansion, given the high burden of HPV-related cancers and the need for improved testing infrastructure.
Competitive Landscape
Leading Companies in the HPV Testing Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
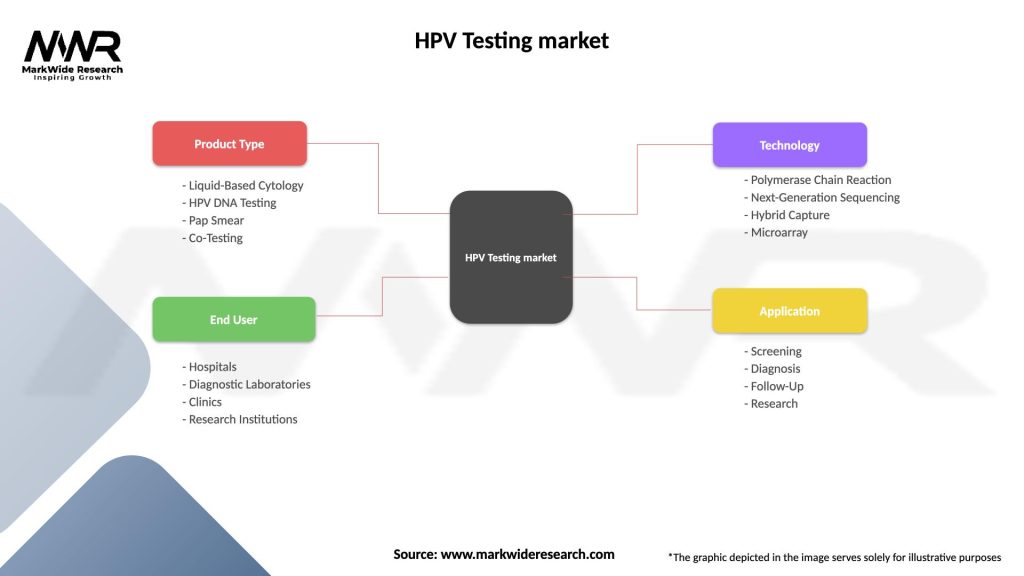
Segmentation
The HPV Testing market can be segmented based on test type, end-user, and region. By test type, the market can be divided into molecular tests (PCR, hybrid capture assays), cytology tests (Pap smears), and others. End-users of HPV testing include hospitals, diagnostic laboratories, and research institutions. Geographically, the market can be segmented into North America, Europe, Asia Pacific, Latin America, and Africa.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
The SWOT analysis for the HPV Testing market is as follows:
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Covid-19 Impact
The COVID-19 pandemic has had an impact on the HPV Testing market. Due to the strain on healthcare resources and the focus on managing the pandemic, routine screening and testing programs were temporarily disrupted in some regions. However, as the situation improves, healthcare systems are gradually resuming HPV testing services. The pandemic has also highlighted the need for resilient testing infrastructure and the integration of testing services into emergency response protocols.
Key Industry Developments
Analyst Suggestions
Future Outlook
The HPV Testing market is expected to continue its growth trajectory in the coming years. Factors such as increasing awareness, advancements in testing technologies, and government initiatives will drive market expansion. The integration of HPV testing into primary healthcare settings and personalized medicine approaches will further enhance testing rates and accuracy. However, addressing cost barriers, improving accessibility in underserved regions, and overcoming social and cultural barriers remain crucial for the market’s future success.
Conclusion
The HPV Testing market is experiencing significant growth, driven by rising awareness about HPV-related cancers and the importance of early detection. Molecular-based testing methods are gaining popularity due to their higher accuracy, while traditional cytology tests continue to play a role in screening programs. Industry participants should focus on product innovation, collaborations, and expanding their reach to tap into emerging markets. By addressing barriers and improving accessibility, the market can contribute to reducing the burden of HPV-related cancers and improving patient outcomes.
What is HPV Testing?
HPV Testing refers to the medical procedures used to detect the presence of human papillomavirus (HPV) in individuals, primarily to assess the risk of cervical cancer and other HPV-related diseases.
What are the key players in the HPV Testing market?
Key players in the HPV Testing market include Roche, Hologic, and Qiagen, which are known for their innovative testing solutions and contributions to HPV research and diagnostics, among others.
What are the main drivers of growth in the HPV Testing market?
The main drivers of growth in the HPV Testing market include increasing awareness of cervical cancer prevention, advancements in testing technologies, and rising healthcare expenditure focused on women’s health.
What challenges does the HPV Testing market face?
Challenges in the HPV Testing market include the high costs associated with testing, varying guidelines for screening across regions, and the need for better education on HPV and its implications.
What opportunities exist in the HPV Testing market?
Opportunities in the HPV Testing market include the development of more accessible testing methods, integration of HPV testing with other screening processes, and potential expansion into emerging markets.
What trends are shaping the HPV Testing market?
Trends shaping the HPV Testing market include the shift towards self-collection methods, the use of molecular testing technologies, and increasing collaborations between public health organizations and private companies.
HPV Testing market
| Segmentation Details | Description |
|---|---|
| Product Type | Liquid-Based Cytology, HPV DNA Testing, Pap Smear, Co-Testing |
| End User | Hospitals, Diagnostic Laboratories, Clinics, Research Institutions |
| Technology | Polymerase Chain Reaction, Next-Generation Sequencing, Hybrid Capture, Microarray |
| Application | Screening, Diagnosis, Follow-Up, Research |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the HPV Testing Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at