444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
Homocystinuria is a rare genetic disorder characterized by the inability of the body to properly process the amino acid methionine. This condition leads to an accumulation of homocysteine, causing various health problems. Homocystinuria therapeutics focus on managing the symptoms and reducing homocysteine levels in affected individuals. The global Homocystinuria Therapeutics market encompasses a range of treatments and interventions aimed at improving the quality of life for patients.
Meaning
Homocystinuria is a metabolic disorder that affects the body’s ability to break down certain proteins. It is caused by mutations in the genes responsible for producing enzymes involved in the metabolism of methionine. These mutations result in the accumulation of homocysteine, leading to a range of symptoms such as developmental delays, skeletal abnormalities, and cardiovascular complications. Effective therapeutics play a crucial role in managing the condition and preventing long-term complications.
Executive Summary
The Homocystinuria Therapeutics market is witnessing steady growth due to increased awareness, advancements in diagnostic techniques, and the development of novel treatment options. The market offers a range of therapeutic approaches, including dietary modifications, vitamin supplementation, and enzyme replacement therapies. Ongoing research and clinical trials hold promise for the development of more effective treatments in the future.

Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
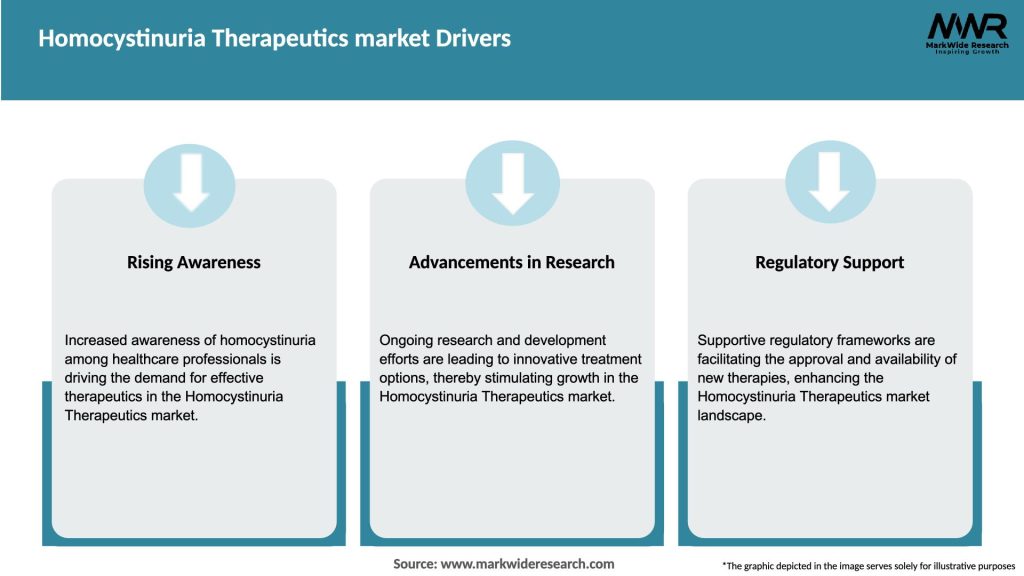
The Homocystinuria Therapeutics market is driven by a combination of factors, including the increasing prevalence of homocystinuria, technological advancements, and government initiatives. The market is also influenced by factors such as limited awareness and diagnosis, high treatment costs, and regulatory hurdles. However, opportunities exist in the form of personalized medicine, expansion in emerging markets, collaborative research efforts, and the adoption of telemedicine and digital health solutions.
Regional Analysis
The Homocystinuria Therapeutics market is segmented into various regions, including North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa. North America currently holds a significant market share due to the presence of well-established healthcare infrastructure, high awareness levels, and government support. Europe and Asia Pacific are also witnessing significant growth, driven by increasing prevalence and improving healthcare systems. Latin America and the Middle East and Africa offer untapped opportunities for market players to expand their presence.
Competitive Landscape
Leading Companies in the Homocystinuria Therapeutics Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
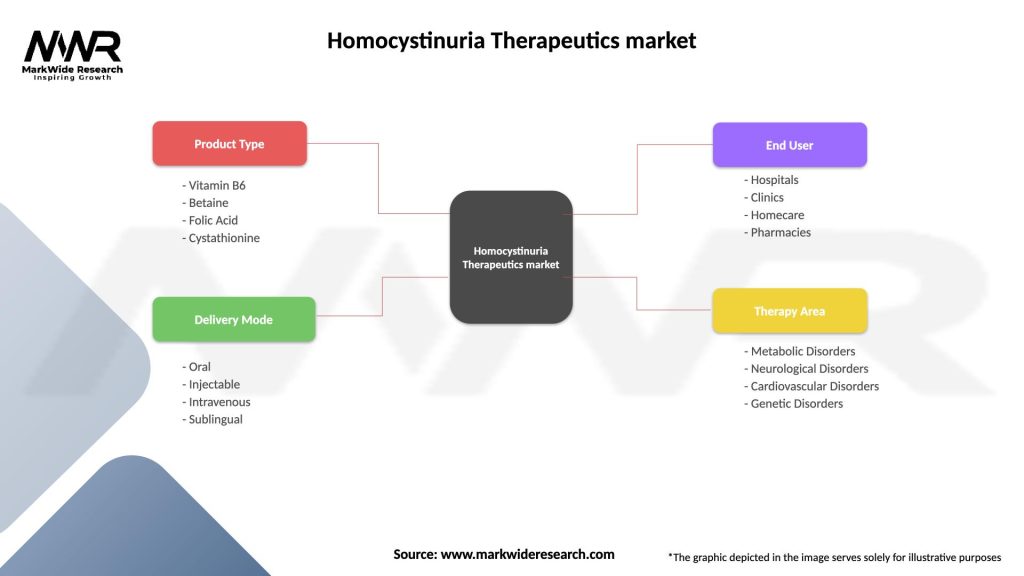
Segmentation
The Homocystinuria Therapeutics market can be segmented based on treatment type, including dietary modifications, vitamin supplementation, enzyme replacement therapies, and gene therapies. It can also be segmented based on end-users, such as hospitals, clinics, and research institutes.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Covid-19 Impact
The global COVID-19 pandemic has had a significant impact on the healthcare industry, including the Homocystinuria Therapeutics market. The pandemic disrupted supply chains, delayed clinical trials, and diverted healthcare resources to address the immediate needs of COVID-19 patients. However, the market demonstrated resilience, with companies adapting to remote work, virtual consultations, and increased telemedicine adoption. The long-term impact of the pandemic on the market will depend on factors such as the duration of the pandemic, vaccination rates, and healthcare system recovery.
Key Industry Developments
Analyst Suggestions
Future Outlook
The Homocystinuria Therapeutics market is expected to witness continued growth in the coming years, driven by increased awareness, advancements in treatment approaches, and growing research and development activities. The development of personalized medicine, expansion into emerging markets, and the adoption of telemedicine and digital health solutions will shape the future of the market. However, challenges such as limited awareness, high treatment costs, and regulatory hurdles need to be addressed to unlock the full potential of homocystinuria therapeutics.
Conclusion
The Homocystinuria Therapeutics market is a dynamic and evolving landscape, driven by the increasing prevalence of the condition and advancements in diagnostic techniques and treatment approaches. While challenges such as limited awareness, high treatment costs, and regulatory hurdles exist, opportunities for market expansion through personalized medicine, collaboration, and technological advancements are on the horizon. Stakeholders must prioritize patient education, research collaboration, and innovation to improve the quality of life for individuals with homocystinuria and drive positive outcomes in the field of therapeutics.
What is Homocystinuria Therapeutics?
Homocystinuria Therapeutics refers to the treatments and interventions designed to manage homocystinuria, a genetic disorder that affects the metabolism of the amino acid methionine. These therapeutics aim to reduce homocysteine levels in the body and prevent complications associated with the condition.
What are the key players in the Homocystinuria Therapeutics market?
Key players in the Homocystinuria Therapeutics market include companies such as Horizon Therapeutics, Recordati, and Cambrooke Therapeutics, which focus on developing specialized treatments and dietary management solutions for patients with this condition, among others.
What are the growth factors driving the Homocystinuria Therapeutics market?
The growth of the Homocystinuria Therapeutics market is driven by increasing awareness of genetic disorders, advancements in genetic testing, and the development of targeted therapies that improve patient outcomes. Additionally, the rising prevalence of homocystinuria is contributing to market expansion.
What challenges does the Homocystinuria Therapeutics market face?
The Homocystinuria Therapeutics market faces challenges such as the high cost of treatment, limited patient population, and the need for ongoing research to develop effective therapies. Regulatory hurdles and the complexity of treatment protocols also pose significant challenges.
What opportunities exist in the Homocystinuria Therapeutics market?
Opportunities in the Homocystinuria Therapeutics market include the potential for innovative drug development, increased investment in rare disease research, and the expansion of telemedicine for patient management. Collaborations between biotech firms and research institutions may also enhance therapeutic options.
What trends are shaping the Homocystinuria Therapeutics market?
Trends in the Homocystinuria Therapeutics market include a growing focus on personalized medicine, advancements in gene therapy, and the integration of digital health technologies for better patient monitoring. There is also an increasing emphasis on dietary management as part of comprehensive treatment plans.
Homocystinuria Therapeutics market
| Segmentation Details | Description |
|---|---|
| Product Type | Vitamin B6, Betaine, Folic Acid, Cystathionine |
| Delivery Mode | Oral, Injectable, Intravenous, Sublingual |
| End User | Hospitals, Clinics, Homecare, Pharmacies |
| Therapy Area | Metabolic Disorders, Neurological Disorders, Cardiovascular Disorders, Genetic Disorders |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Homocystinuria Therapeutics Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at