444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
The hepatitis C testing market is experiencing significant growth due to the rising prevalence of hepatitis C virus (HCV) infections globally. Hepatitis C is a viral infection that primarily affects the liver, leading to chronic liver disease, cirrhosis, and in some cases, hepatocellular carcinoma. The market for hepatitis C testing is driven by the need for early detection, accurate diagnosis, and effective treatment options to combat this life-threatening disease.
Hepatitis C is a bloodborne infection caused by the hepatitis C virus (HCV). It is mainly transmitted through exposure to infected blood, such as through sharing needles during drug use, receiving contaminated blood transfusions or organ transplants, or through unsafe medical procedures. The virus can also be transmitted from an infected mother to her baby during childbirth. Hepatitis C can range from a mild illness lasting a few weeks to a chronic condition that leads to severe liver damage.
Executive Summary
The hepatitis C testing market is witnessing rapid growth due to the increasing number of HCV infections worldwide. The market is driven by advancements in diagnostic technologies, growing awareness about the importance of early detection, and the introduction of effective antiviral therapies. Key market players are focusing on strategic collaborations, product innovations, and geographical expansion to gain a competitive edge in the market.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The hepatitis C testing market is highly dynamic, driven by technological advancements, evolving treatment guidelines, and changing market dynamics. Market players are investing in research and development to introduce innovative diagnostic tools and therapies. The market is witnessing a transition from traditional laboratory-based tests to point-of-care devices, enabling rapid and decentralized testing. Strategic collaborations and partnerships between diagnostic companies and pharmaceutical firms are fostering innovation and market growth.
Regional Analysis
The hepatitis C testing market exhibits regional variations, with North America and Europe currently dominating the market due to well-established healthcare infrastructure, favorable reimbursement policies, and high awareness levels. However, the Asia Pacific region is anticipated to witness significant growth during the forecast period due to the increasing prevalence of HCV infections, rising healthcare expenditure, and government initiatives to control and eliminate hepatitis C.
Competitive Landscape
Leading companies in the Hepatitis C Testing Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
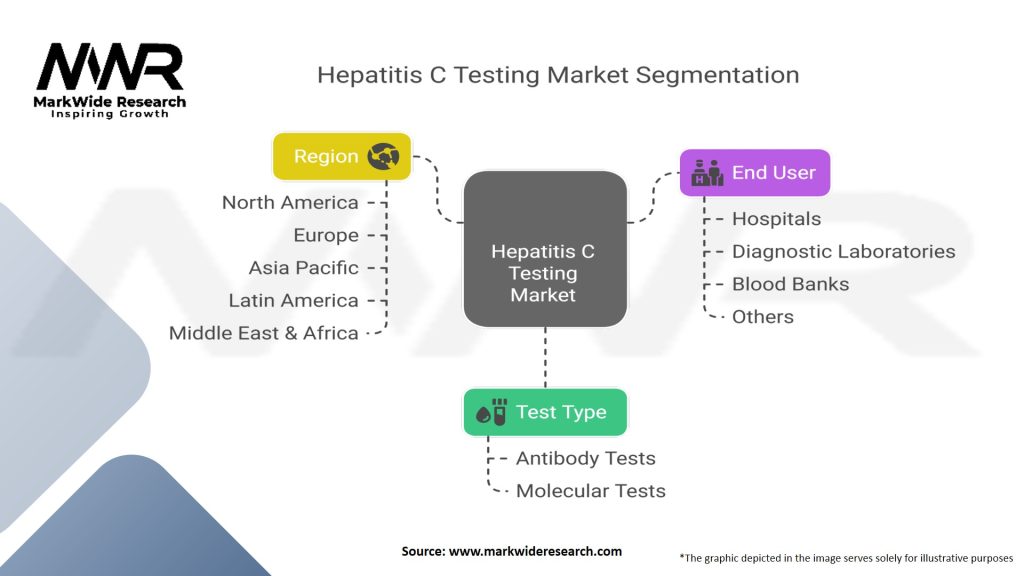
Segmentation
The hepatitis C testing market can be segmented based on test type, end-user, and region.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
The hepatitis C testing market offers several benefits for industry participants and stakeholders:
SWOT Analysis
Market Key Trends
Covid-19 Impact
The COVID-19 pandemic has had a significant impact on the hepatitis C testing market. The diversion of healthcare resources and the implementation of infection control measures have led to disruptions in HCV testing and treatment services. Routine screening programs were temporarily halted, and individuals with chronic hepatitis C faced challenges in accessing healthcare facilities for testing and treatment. However, healthcare systems have adapted to the situation by implementing telemedicine services, home-based testing options, and ensuring continuity of care for individuals with chronic conditions. The pandemic has also highlighted the importance of resilient healthcare systems and the need for innovative approaches to ensure access to essential healthcare services, including hepatitis C testing.
Key Industry Developments
Analyst Suggestions
Future Outlook
The hepatitis C testing market is poised for significant growth in the coming years. Increasing awareness, technological advancements, and the availability of effective antiviral therapies will drive market expansion. The market will witness a shift towards non-invasive testing methods and the integration of AI and ML algorithms into diagnostic platforms. Targeted testing strategies and public health initiatives will play a crucial role in reducing the burden of hepatitis C. Continued investment in research and development, collaborations, and regulatory support will shape the future of hepatitis C testing and treatment.
Conclusion
The hepatitis C testing market is experiencing rapid growth due to the increasing prevalence of HCV infections worldwide. Advancements in diagnostic technologies, the availability of effective antiviral therapies, and growing awareness about the importance of early detection and treatment are driving market expansion. Industry participants and stakeholders have significant opportunities to tap into this market by developing innovative diagnostic tools, expanding access to testing, and collaborating to improve patient outcomes. Continued efforts in education, research, and public health initiatives will be crucial in controlling and eliminating hepatitis C and improving the overall health outcomes for individuals affected by this disease.
What is Hepatitis C Testing?
Hepatitis C Testing refers to the medical procedures used to detect the presence of the Hepatitis C virus in an individual’s blood. This testing is crucial for diagnosing infection, monitoring liver health, and determining the appropriate treatment options.
What are the key companies in the Hepatitis C Testing Market?
Key companies in the Hepatitis C Testing Market include Abbott Laboratories, Roche Diagnostics, and Siemens Healthineers, among others.
What are the drivers of growth in the Hepatitis C Testing Market?
The growth of the Hepatitis C Testing Market is driven by increasing awareness of Hepatitis C, advancements in testing technologies, and the rising prevalence of the virus globally. Additionally, government initiatives to promote testing contribute to market expansion.
What challenges does the Hepatitis C Testing Market face?
The Hepatitis C Testing Market faces challenges such as the stigma associated with the disease, limited access to healthcare in certain regions, and the high cost of some testing methods. These factors can hinder widespread testing and diagnosis.
What opportunities exist in the Hepatitis C Testing Market?
Opportunities in the Hepatitis C Testing Market include the development of rapid testing kits, increased funding for public health initiatives, and the potential for integration of testing with telehealth services. These advancements can enhance accessibility and efficiency in testing.
What trends are shaping the Hepatitis C Testing Market?
Trends in the Hepatitis C Testing Market include the shift towards point-of-care testing, the use of artificial intelligence in diagnostic processes, and the growing emphasis on personalized medicine. These trends aim to improve patient outcomes and streamline testing procedures.
Hepatitis C Testing Market
| Segmentation | Details |
|---|---|
| Test Type | Antibody Tests, Molecular Tests |
| End User | Hospitals, Diagnostic Laboratories, Blood Banks, Others |
| Region | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading companies in the Hepatitis C Testing Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at