444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The Global Rift Valley Fever Vaccines market is experiencing significant growth and is expected to expand at a steady rate in the coming years. Rift Valley Fever (RVF) is a viral zoonotic disease that primarily affects animals but can also infect humans. The disease is prevalent in regions of Africa, such as Kenya, Sudan, and South Africa, but cases have also been reported in the Middle East. RVF can have severe economic consequences due to its impact on livestock, as it causes high mortality rates among animals and leads to significant losses in the agricultural sector.
Meaning
Rift Valley Fever is caused by the Rift Valley Fever virus (RVFV), which belongs to the Bunyaviridae family. The virus is primarily transmitted through mosquito bites or contact with infected animals or their tissues. RVFV can cause a range of symptoms in humans, including fever, headache, muscle and joint pain, and in severe cases, it can lead to hemorrhagic fever or encephalitis. In animals, the virus can cause abortion in pregnant livestock and high mortality rates among young animals.
Executive Summary
The Global Rift Valley Fever Vaccines market is expected to witness substantial growth due to the increasing incidence of RVF and the rising demand for effective vaccines. The market is driven by the need to protect livestock and prevent the spread of the virus to humans. Several companies are investing in the development of RVF vaccines, focusing on both inactivated and live attenuated vaccines. The market is also supported by government initiatives and funding for disease control and prevention programs.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
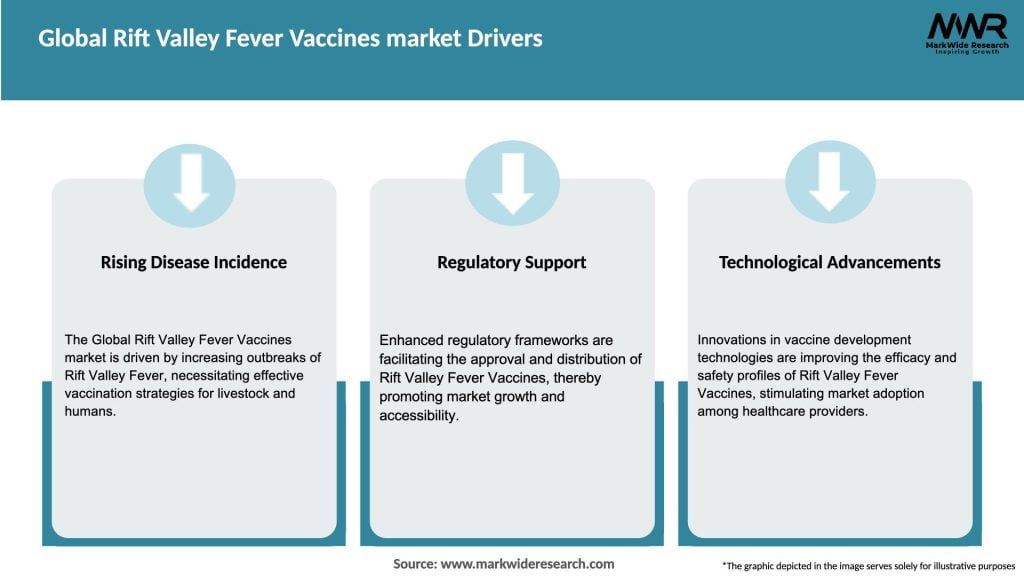
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The Global Rift Valley Fever Vaccines market is characterized by intense competition among key players, with a focus on research and development activities to develop innovative and effective vaccines. Market dynamics are influenced by factors such as government regulations, disease prevalence, public health concerns, and technological advancements in vaccine development. The market is expected to witness consolidation through strategic alliances, mergers, and acquisitions, enabling companies to strengthen their product portfolios and expand their geographic presence.
Regional Analysis
The Global Rift Valley Fever Vaccines market is segmented into several regions, including North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa. Africa is expected to dominate the market due to the high prevalence of RVF in the region and the significant economic impact of the disease. North America and Europe are also anticipated to witness substantial growth due to increasing awareness and investments in RVF control measures.
Competitive Landscape
Leading companies in the Global Rift Valley Fever Vaccines Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
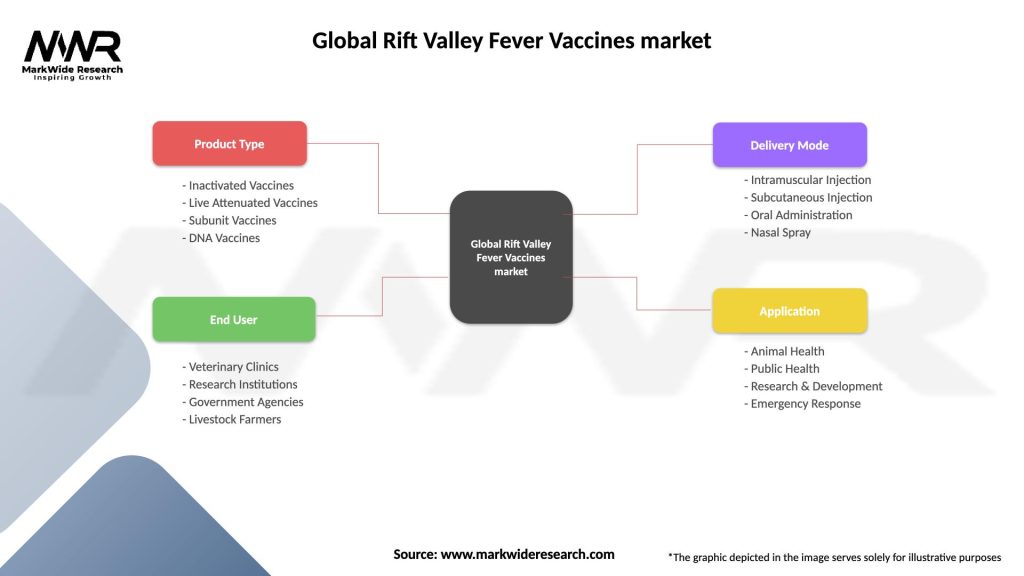
Segmentation
The Global Rift Valley Fever Vaccines market can be segmented based on vaccine type, animal type, and distribution channel. By vaccine type, the market can be categorized into inactivated vaccines, live attenuated vaccines, and others. By animal type, the market can be divided into cattle, sheep and goats, camels, and others. Based on the distribution channel, the market can be classified into veterinary clinics, pharmacies, and online platforms.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Covid-19 Impact
The Covid-19 pandemic has had a significant impact on the Global Rift Valley Fever Vaccines market. While RVF is unrelated to Covid-19, the pandemic has highlighted the importance of preparedness and the need for robust vaccination strategies. The focus on vaccine development and distribution during the pandemic has accelerated research and development efforts for RVF vaccines as well. However, the diversion of resources and disruptions in supply chains caused by the pandemic may have temporarily affected the availability and distribution of RVF vaccines.
Key Industry Developments
Advances in Vaccine Research: Recent advancements in the research and development of RVF vaccines have resulted in more effective and safer options. Companies are focusing on enhancing the stability and shelf life of vaccines, making them easier to distribute and store in low-resource environments.
Increased Government Support: Governments and international health organizations are increasing their investments in RVF control and prevention, leading to a greater demand for vaccines. This support is helping to overcome barriers such as high production costs and distribution challenges.
Public-Private Partnerships: Collaborations between the public and private sectors are helping to reduce the cost of vaccine development and ensure that vaccines are distributed in endemic regions where they are most needed.
Analyst Suggestions
Future Outlook
The Global Rift Valley Fever Vaccines market is expected to grow at a steady pace in the coming years, driven by increasing awareness about the disease and the need for effective preventive measures. Technological advancements, such as the use of novel vaccine platforms and adjuvants, will contribute to the development of more efficient and targeted vaccines. Collaboration between industry players, research institutions, and governments will be crucial for addressing challenges and improving vaccine accessibility. The market is expected to witness significant opportunities in emerging markets, fueled by rising demand for livestock products and increasing investments in animal health.
Conclusion
The Global Rift Valley Fever Vaccines market is poised for significant growth, driven by the increasing prevalence of RVF and the need to protect both animals and humans from the disease. The market offers opportunities for vaccine manufacturers, research institutions, and governments to collaborate and develop innovative and cost-effective solutions. However, challenges such as high development costs, limited accessibility, and regulatory requirements need to be addressed. With continuous investments in research and development, improved vaccine distribution strategies, and strong collaborations, the market is expected to witness steady growth and contribute to the prevention and control of Rift Valley Fever globally.
What is Rift Valley Fever Vaccines?
Rift Valley Fever Vaccines are immunizations designed to protect against Rift Valley Fever, a viral disease that primarily affects livestock and can also infect humans. These vaccines are crucial for controlling outbreaks and ensuring animal health in affected regions.
What are the key players in the Global Rift Valley Fever Vaccines market?
Key players in the Global Rift Valley Fever Vaccines market include companies like Zoetis, Merck Animal Health, and Boehringer Ingelheim, which are involved in the development and distribution of vaccines for livestock and public health, among others.
What are the growth factors driving the Global Rift Valley Fever Vaccines market?
The growth of the Global Rift Valley Fever Vaccines market is driven by increasing incidences of Rift Valley Fever outbreaks, rising awareness about zoonotic diseases, and the need for effective vaccination programs in livestock management.
What challenges does the Global Rift Valley Fever Vaccines market face?
The Global Rift Valley Fever Vaccines market faces challenges such as regulatory hurdles, the complexity of vaccine development, and the need for extensive field trials to ensure safety and efficacy in diverse animal populations.
What opportunities exist in the Global Rift Valley Fever Vaccines market?
Opportunities in the Global Rift Valley Fever Vaccines market include advancements in vaccine technology, increasing investment in veterinary health, and the potential for new markets in developing regions where livestock farming is prevalent.
What trends are shaping the Global Rift Valley Fever Vaccines market?
Trends shaping the Global Rift Valley Fever Vaccines market include the development of more effective and safer vaccine formulations, the integration of digital technologies in vaccine distribution, and a growing focus on sustainable livestock management practices.
Global Rift Valley Fever Vaccines market
| Segmentation Details | Description |
|---|---|
| Product Type | Inactivated Vaccines, Live Attenuated Vaccines, Subunit Vaccines, DNA Vaccines |
| End User | Veterinary Clinics, Research Institutions, Government Agencies, Livestock Farmers |
| Delivery Mode | Intramuscular Injection, Subcutaneous Injection, Oral Administration, Nasal Spray |
| Application | Animal Health, Public Health, Research & Development, Emergency Response |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading companies in the Global Rift Valley Fever Vaccines Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at