444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The global in vivo toxicology market is witnessing significant growth due to the increasing emphasis on safety assessment of pharmaceuticals, chemicals, and consumer products. In vivo toxicology refers to the study of the adverse effects of substances on living organisms, typically animals. This comprehensive analysis provides insights into the current state and future prospects of the global in vivo toxicology market.
Meaning
In vivo toxicology involves the assessment of the toxic effects of substances on living organisms, typically animals, to evaluate their safety for human use. It plays a critical role in drug development, chemical risk assessment, and ensuring the safety of consumer products. In vivo toxicology studies involve the administration of substances to animals and monitoring their physiological, biochemical, and histopathological changes to determine their toxicity levels.
Executive Summary
The executive summary offers a concise overview of the key findings and highlights of the global in vivo toxicology market. It outlines the market size, growth rate, and major trends observed in the industry. The summary also includes a snapshot of the competitive landscape and key recommendations for industry participants and stakeholders.
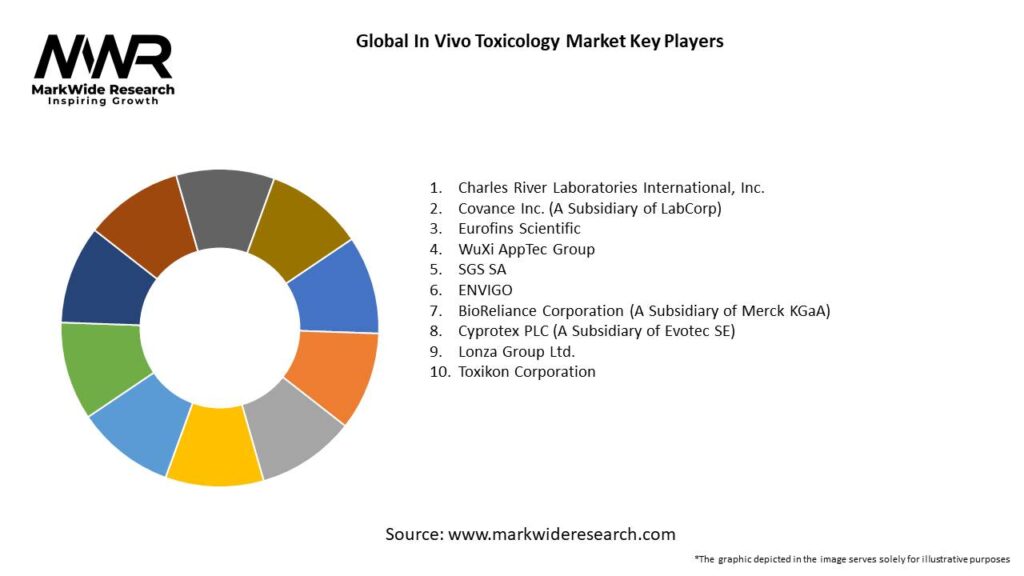
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
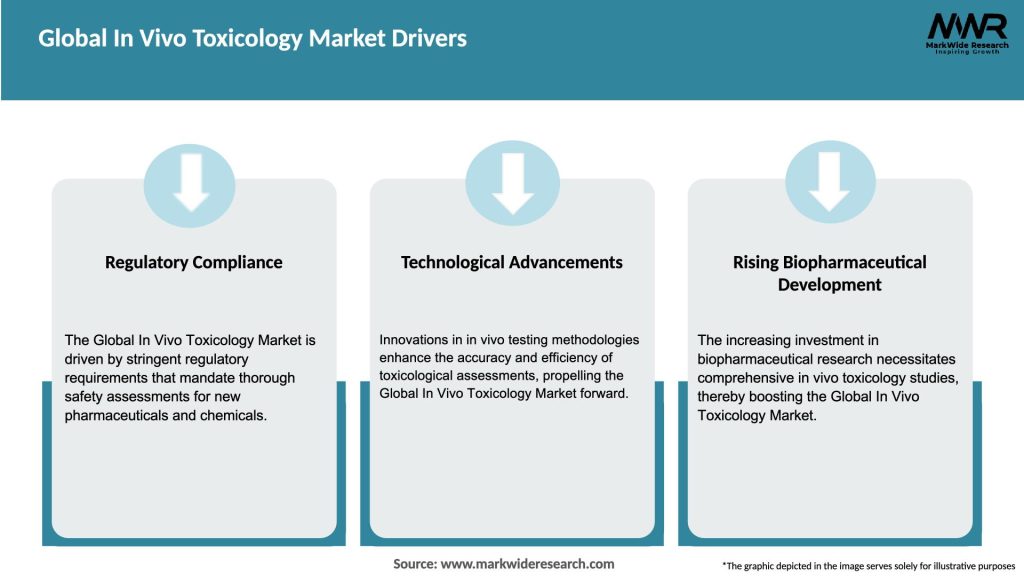
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
Regional Analysis
North America dominates the global in vivo toxicology market, driven by strong pharmaceutical and biotechnology sectors and strict regulatory requirements. The United States, in particular, is a major player in the market due to its extensive pharmaceutical R&D activities and regulatory framework. Europe also holds a significant share, with increasing regulatory pressure and a growing emphasis on product safety. However, the European market faces challenges due to regulations restricting animal testing. The Asia-Pacific region, led by countries like China and India, is expected to witness the highest growth due to the increasing pharmaceutical R&D and improving healthcare infrastructure.
Competitive Landscape
Leading Companies in Global In Vivo Toxicology Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
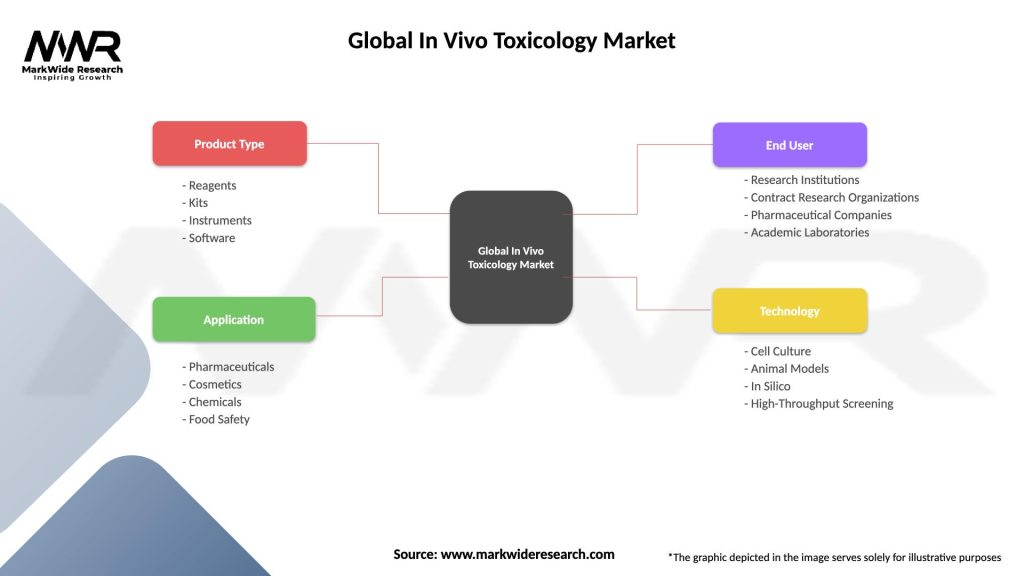
Segmentation
By Type of Test
By Application
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Market Key Trends
Covid-19 Impact
The Covid-19 impact section examines the repercussions of the pandemic on the global in vivo toxicology market. It discusses the short-term and long-term effects, including the disruptions in research activities, the increased focus on drug safety, and the acceleration of digital and remote toxicology approaches. The section offers insights into the market’s resilience and adaptation strategies.
Key Industry Developments
The key industry developments section highlights the recent advancements and innovations in the global in vivo toxicology market. It covers collaborations, research breakthroughs, technological advancements, and regulatory updates. The section provides readers with up-to-date information on the market’s latest developments.
Analyst Suggestions
The analyst suggestions section offers expert recommendations and strategies for industry participants and stakeholders in the global in vivo toxicology market. It provides actionable insights to help companies enhance testing efficiency, improve data interpretation, and optimize regulatory compliance. The section assists stakeholders in making informed decisions and staying ahead of the competition.
Future Outlook
The future outlook section provides a comprehensive analysis of the global in vivo toxicology market’s growth prospects and opportunities. It considers factors such as the increasing demand for alternative testing methods, the advancements in predictive toxicology, and the potential for regulatory acceptance of new approaches. The section helps stakeholders formulate effective strategies for long-term success.
Conclusion
The conclusion summarizes the key findings and insights discussed throughout the analysis of the global in vivo toxicology market. It reiterates the market’s growth potential, highlights the major trends and challenges, and emphasizes the critical role of in vivo toxicology in ensuring the safety of pharmaceuticals, chemicals, and consumer products. The conclusion serves as a comprehensive overview of the market for readers seeking a summary of the report’s key points.
What is In Vivo Toxicology?
In Vivo Toxicology refers to the study of the effects of substances on living organisms, typically using animal models, to assess the safety and efficacy of drugs, chemicals, and other products. This field is crucial for understanding potential health risks and regulatory compliance.
What are the key players in the Global In Vivo Toxicology Market?
Key players in the Global In Vivo Toxicology Market include Charles River Laboratories, Covance, and Eurofins Scientific, which provide a range of toxicology testing services. These companies are known for their expertise in preclinical research and regulatory support, among others.
What are the main drivers of the Global In Vivo Toxicology Market?
The main drivers of the Global In Vivo Toxicology Market include the increasing demand for drug development, the need for regulatory compliance, and the growing focus on safety assessments in pharmaceuticals and chemicals. Additionally, advancements in testing methodologies are contributing to market growth.
What challenges does the Global In Vivo Toxicology Market face?
The Global In Vivo Toxicology Market faces challenges such as ethical concerns regarding animal testing, high costs associated with in vivo studies, and the complexity of interpreting results. These factors can hinder research and development timelines.
What opportunities exist in the Global In Vivo Toxicology Market?
Opportunities in the Global In Vivo Toxicology Market include the development of alternative testing methods, such as in vitro and computational models, which can reduce reliance on animal testing. Additionally, the expansion of biopharmaceuticals presents new avenues for toxicology research.
What trends are shaping the Global In Vivo Toxicology Market?
Trends shaping the Global In Vivo Toxicology Market include the integration of advanced technologies like artificial intelligence and machine learning in toxicology assessments. Furthermore, there is a growing emphasis on personalized medicine and targeted therapies, influencing toxicological evaluations.
Global In Vivo Toxicology Market
| Segmentation Details | Description |
|---|---|
| Product Type | Reagents, Kits, Instruments, Software |
| Application | Pharmaceuticals, Cosmetics, Chemicals, Food Safety |
| End User | Research Institutions, Contract Research Organizations, Pharmaceutical Companies, Academic Laboratories |
| Technology | Cell Culture, Animal Models, In Silico, High-Throughput Screening |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in Global In Vivo Toxicology Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at