444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The Global DT-Based Combined Vaccines market refers to the market for vaccines that provide protection against diphtheria and tetanus, often in combination with other antigens. These vaccines are crucial in preventing these infectious diseases and are widely used across the world. The market for DT-based combined vaccines has experienced significant growth in recent years, driven by the increasing emphasis on immunization programs, rising awareness about the importance of vaccination, and the introduction of innovative vaccine formulations.
Meaning
DT-based combined vaccines are immunization products that contain antigens for both diphtheria and tetanus. These vaccines are typically administered through intramuscular injection and are designed to stimulate the immune system to produce protective antibodies against the respective diseases. By combining the antigens in a single vaccine, healthcare providers can simplify immunization schedules and reduce the number of injections required for individuals. This approach has proven effective in reducing the burden of diphtheria and tetanus in many regions worldwide.
Executive Summary
The Global DT-Based Combined Vaccines market has witnessed substantial growth in recent years, driven by the increasing focus on immunization programs and the need to prevent diphtheria and tetanus infections. The market is characterized by the presence of established vaccine manufacturers and research institutions that are continuously working towards developing advanced vaccine formulations. The market offers significant opportunities for both existing players and new entrants to capitalize on the growing demand for DT-based combined vaccines. However, challenges such as vaccine hesitancy and supply chain issues need to be addressed to ensure the sustained growth of the market.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
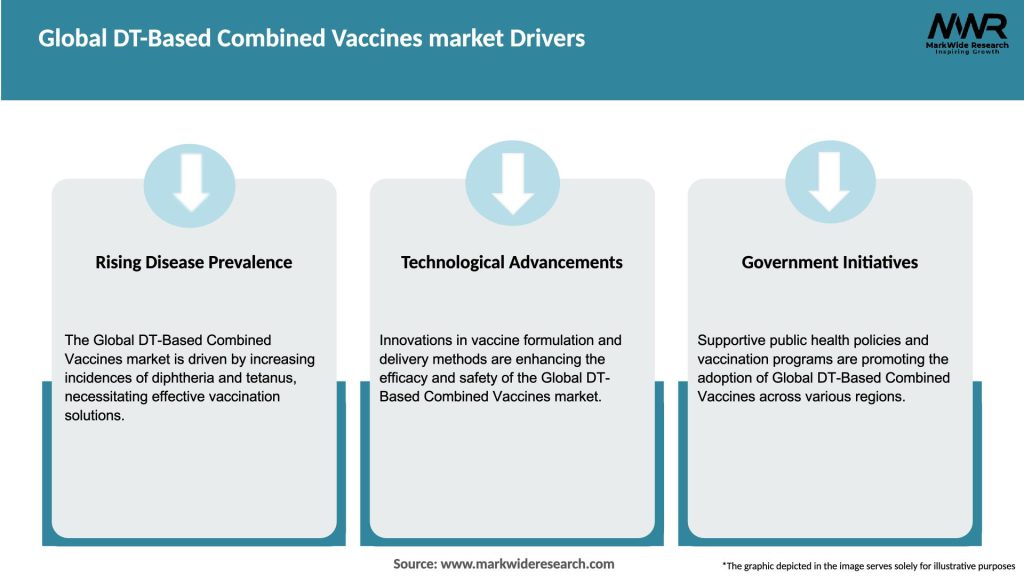
Market Dynamics
The Global DT-Based Combined Vaccines market is dynamic and influenced by various factors. The market is driven by the increasing emphasis on immunization programs and rising awareness about the importance of vaccination. Technological advancements in vaccine production and strong regulatory frameworks also contribute to market growth. However, vaccine hesitancy, supply chain issues, and the high cost of vaccines act as significant restraints. The market offers opportunities for expansion in emerging markets and the development of combination vaccines. Continuous investments in research and development are expected to shape the market’s future trajectory.
Regional Analysis
The Global DT-Based Combined Vaccines market can be analyzed across various regions, including North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa. Each region has unique characteristics and factors that influence the market dynamics. North America and Europe have well-established healthcare systems and robust immunization programs, driving the demand for DT-based combined vaccines. Asia Pacific presents significant growth opportunities due to the large population, increasing healthcare expenditure, and expanding immunization initiatives. Latin America, the Middle East, and Africa are witnessing improvements in healthcare infrastructure, which can contribute to market growth.
Competitive Landscape
Leading Companies in the Global DT-Based Combined Vaccines Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
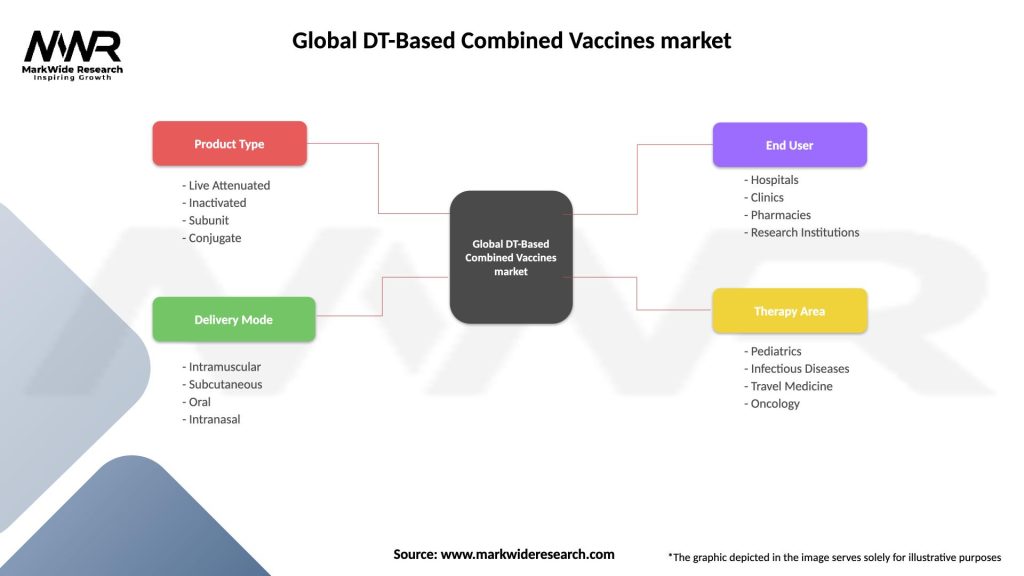
Segmentation
The Global DT-Based Combined Vaccines market can be segmented based on various factors, including vaccine type, end-user, and distribution channel. Vaccine types may include DTaP (Diphtheria, Tetanus, and Pertussis) vaccines, DT (Diphtheria and Tetanus) vaccines, and Tdap (Tetanus, Diphtheria, and Pertussis) vaccines. End-users may comprise pediatric population, adult population, and healthcare institutions. Distribution channels can include hospitals, clinics, retail pharmacies, and online pharmacies.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
A SWOT analysis of the Global DT-Based Combined Vaccines market can provide insights into the market’s strengths, weaknesses, opportunities, and threats.
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Covid-19 Impact
The COVID-19 pandemic has had a significant impact on the Global DT-Based Combined Vaccines market. While the primary focus has been on the development and distribution of COVID-19 vaccines, routine immunization programs, including DT-based combined vaccines, have faced disruptions. Vaccination campaigns were temporarily halted or scaled back in many countries, leading to a decline in vaccine coverage rates for diphtheria and tetanus. The pandemic also highlighted the importance of vaccine development and manufacturing capabilities, leading to increased investments in vaccine research and production infrastructure. As the world recovers from the pandemic, efforts are being made to restore and strengthen routine immunization programs, which will drive the demand for DT-based combined vaccines.
Key Industry Developments
The Global DT-Based Combined Vaccines market has witnessed significant developments in recent years, driven by various factors such as increasing awareness about the benefits of vaccination, rising government initiatives, and advancements in healthcare infrastructure. DT-based combined vaccines, which offer protection against diphtheria and tetanus, have gained considerable popularity due to their effectiveness and convenience.
One of the key industry developments is the growing demand for DT-based combined vaccines in both developed and developing regions. The rising prevalence of diphtheria and tetanus, coupled with the need for comprehensive immunization programs, has fueled the market growth. Governments and healthcare organizations are actively promoting the use of these vaccines to prevent the spread of these infectious diseases.
In addition, advancements in vaccine technology have contributed to the growth of the DT-based combined vaccines market. Manufacturers are investing in research and development to improve the efficacy and safety of these vaccines. The development of newer adjuvants and delivery systems has enhanced the immune response and reduced the side effects associated with vaccination.
Analyst Suggestions
Based on the market analysis, industry experts suggest several strategies for stakeholders in the Global DT-Based Combined Vaccines market. Firstly, manufacturers should focus on expanding their production capacities to meet the growing demand for these vaccines. Collaborations with government bodies and healthcare organizations can help ensure a steady supply and distribution of vaccines.
Furthermore, research and development efforts should be directed towards the development of next-generation DT-based combined vaccines. The incorporation of additional antigens, such as pertussis, can provide broader protection against multiple diseases in a single shot. Moreover, continuous monitoring and surveillance of vaccine efficacy and safety are essential to address any concerns and maintain public trust.
Market players should also prioritize improving the accessibility and affordability of DT-based combined vaccines, particularly in low-income countries. This can be achieved through partnerships with international organizations, local manufacturers, and the implementation of cost-effective production processes.
Future Outlook
The future outlook for the Global DT-Based Combined Vaccines market appears promising. The increasing emphasis on immunization programs and the need for comprehensive protection against diphtheria and tetanus are expected to drive the market growth. Technological advancements in vaccine development and the introduction of innovative delivery systems will further contribute to market expansion.
Additionally, the ongoing research on combination vaccines, incorporating DT-based protection with other antigens, holds great potential. This approach can simplify vaccination schedules, reduce the number of injections required, and improve patient compliance. Such developments are likely to attract significant investments and collaborations in the coming years.
Conclusion
In conclusion, the Global DT-Based Combined Vaccines market is witnessing substantial growth due to key industry developments, such as increasing demand, advancements in vaccine technology, and government initiatives. Analysts suggest that stakeholders should focus on expanding production capacities, investing in research and development, and ensuring accessibility and affordability. The future outlook for the market is positive, driven by the growing emphasis on immunization programs and the potential for innovative combination vaccines. The DT-based combined vaccines market plays a vital role in preventing the spread of diphtheria and tetanus, and continuous efforts are needed to address the evolving healthcare needs and ensure global vaccination coverage.
What is DT-Based Combined Vaccines?
DT-Based Combined Vaccines refer to vaccines that combine diphtheria and tetanus toxoids with other vaccines to provide immunity against multiple diseases. These vaccines are essential in immunization programs to prevent serious infections in children and adults.
What are the key players in the Global DT-Based Combined Vaccines market?
Key players in the Global DT-Based Combined Vaccines market include Sanofi Pasteur, GlaxoSmithKline, Merck & Co., and Pfizer, among others. These companies are involved in the development and distribution of various combined vaccines to enhance public health.
What are the growth factors driving the Global DT-Based Combined Vaccines market?
The Global DT-Based Combined Vaccines market is driven by increasing vaccination rates, rising awareness about preventive healthcare, and government initiatives to control infectious diseases. Additionally, advancements in vaccine technology contribute to market growth.
What challenges does the Global DT-Based Combined Vaccines market face?
The Global DT-Based Combined Vaccines market faces challenges such as vaccine hesitancy, logistical issues in vaccine distribution, and regulatory hurdles. These factors can impact the effectiveness of immunization programs.
What opportunities exist in the Global DT-Based Combined Vaccines market?
Opportunities in the Global DT-Based Combined Vaccines market include the development of new combination vaccines, expansion into emerging markets, and increasing partnerships between public health organizations and vaccine manufacturers. These factors can enhance vaccine accessibility and coverage.
What trends are shaping the Global DT-Based Combined Vaccines market?
Trends shaping the Global DT-Based Combined Vaccines market include the integration of digital health technologies for vaccination tracking, the rise of personalized vaccines, and a focus on improving vaccine formulations. These trends aim to enhance immunization efficiency and effectiveness.
Global DT-Based Combined Vaccines market
| Segmentation Details | Description |
|---|---|
| Product Type | Live Attenuated, Inactivated, Subunit, Conjugate |
| Delivery Mode | Intramuscular, Subcutaneous, Oral, Intranasal |
| End User | Hospitals, Clinics, Pharmacies, Research Institutions |
| Therapy Area | Pediatrics, Infectious Diseases, Travel Medicine, Oncology |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Global DT-Based Combined Vaccines Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at