444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
Fetal Valproate Syndrome (FVS) is a rare but serious condition caused by prenatal exposure to valproic acid (VPA), a medication commonly prescribed for epilepsy, bipolar disorder, and migraine prophylaxis. FVS is characterized by a range of congenital abnormalities, developmental delays, and cognitive impairments in children exposed to VPA during pregnancy. Despite its rarity, FVS poses significant challenges for affected individuals, families, and healthcare systems due to its lifelong impact on health and well-being.
Meaning
Fetal Valproate Syndrome (FVS) refers to a constellation of physical, cognitive, and behavioral abnormalities observed in individuals exposed to valproic acid (VPA) in utero. Prenatal exposure to VPA, a medication used to treat epilepsy, bipolar disorder, and migraine, can result in various developmental abnormalities and neurocognitive deficits in affected individuals. FVS represents a significant public health concern due to its long-term implications for affected individuals and their families.
Executive Summary
Fetal Valproate Syndrome (FVS) is a rare but serious condition that occurs in children exposed to valproic acid (VPA) during pregnancy. The syndrome is characterized by a spectrum of developmental abnormalities, including congenital malformations, cognitive impairments, and behavioral difficulties. Despite efforts to raise awareness about the risks of VPA exposure during pregnancy, FVS remains a significant clinical challenge with lifelong consequences for affected individuals. Multidisciplinary interventions and support services are essential to mitigate the impact of FVS on affected individuals and their families.
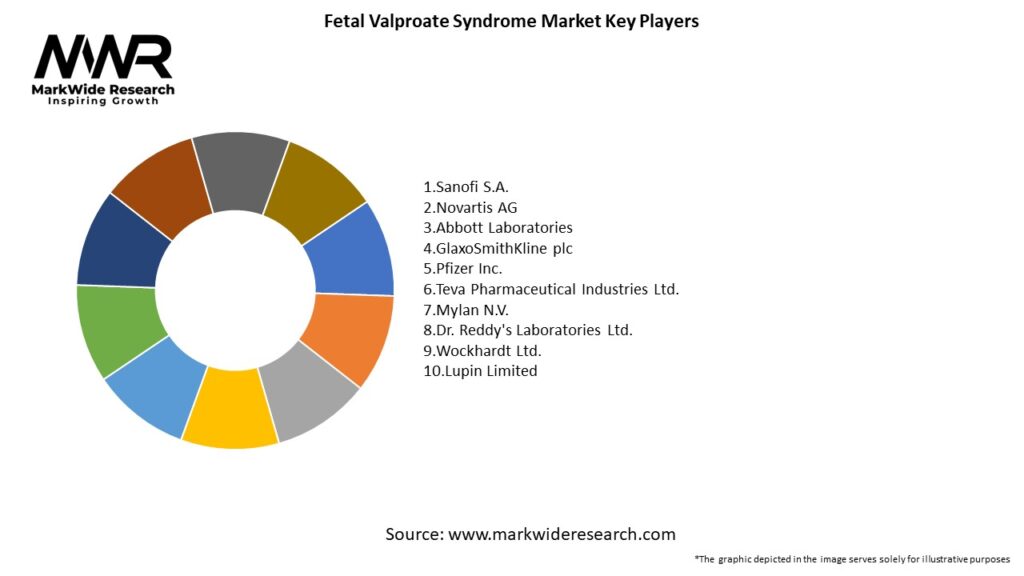
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
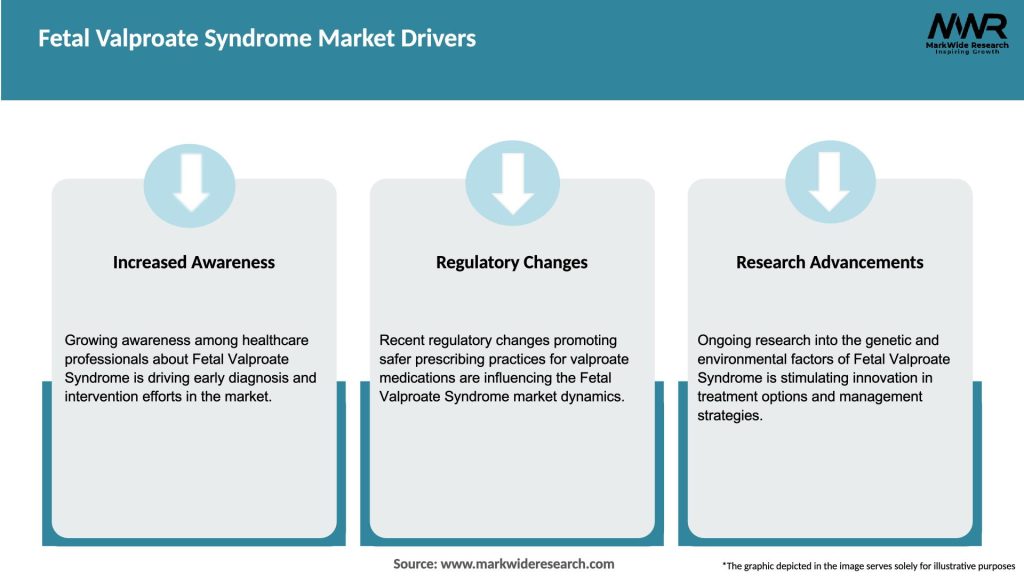
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The market for Fetal Valproate Syndrome (FVS) is influenced by complex interactions between clinical, regulatory, ethical, and socioeconomic factors. The dynamic nature of these interactions underscores the need for ongoing research, surveillance, and advocacy efforts to address the unmet needs of individuals affected by FVS and to prevent future cases through risk mitigation strategies and improved patient care pathways.
Regional Analysis
The prevalence and management of Fetal Valproate Syndrome (FVS) may vary across different regions due to differences in healthcare infrastructure, regulatory frameworks, clinical practices, and public health policies. Regional disparities in access to prenatal care, genetic counseling services, and specialized interventions may impact the diagnosis and management of FVS in affected populations.
Competitive Landscape
Leading Companies in the Fetal Valproate Syndrome Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
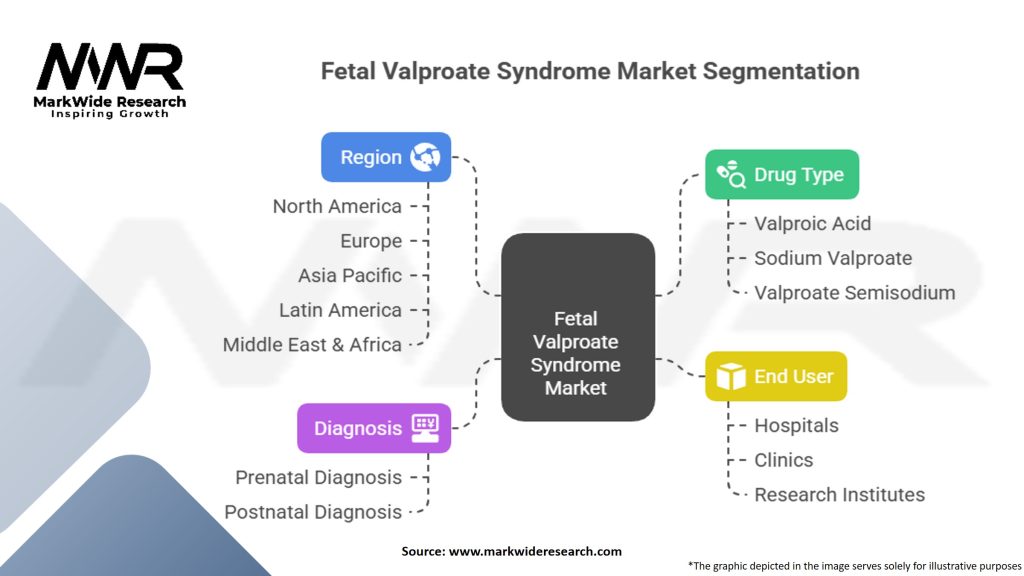
Segmentation
The market for Fetal Valproate Syndrome (FVS) can be segmented based on various factors, including:
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Market Key Trends
Covid-19 Impact
The COVID-19 pandemic has had profound implications for the diagnosis, treatment, and support of individuals with Fetal Valproate Syndrome (FVS), including disruptions to healthcare services, delays in diagnostic evaluations, and challenges in accessing supportive resources. Key impacts of COVID-19 on the FVS market include:
Key Industry Developments
Analyst Suggestions
Future Outlook
The future outlook for the Fetal Valproate Syndrome (FVS) market is characterized by ongoing efforts to improve risk awareness, enhance prenatal counseling, advance genetic research, and optimize clinical management strategies. Despite challenges posed by the COVID-19 pandemic and evolving regulatory landscapes, the collective commitment of stakeholders to address the unmet needs of individuals affected by FVS offers hope for improved outcomes, increased resilience, and greater societal awareness of the importance of medication safety during pregnancy.
Conclusion
Fetal Valproate Syndrome (FVS) represents a complex clinical entity with significant implications for affected individuals, families, and healthcare systems. Despite advances in prenatal care, genetic testing, and therapeutic interventions, FVS remains a challenging condition with lifelong consequences for neurodevelopmental health and well-being. Multidisciplinary collaboration, patient advocacy, and public health initiatives are essential to raise awareness, promote prevention, and optimize care pathways for individuals affected by FVS. By fostering a culture of innovation, inclusivity, and evidence-based practice, stakeholders can work together to improve outcomes, enhance quality of life, and support the needs of individuals living with Fetal Valproate Syndrome (FVS) and their families.
What is Fetal Valproate Syndrome?
Fetal Valproate Syndrome is a condition that results from exposure to the medication valproate during pregnancy. It is characterized by a range of physical, cognitive, and developmental disabilities in the child.
What are the key players in the Fetal Valproate Syndrome Market?
Key players in the Fetal Valproate Syndrome Market include pharmaceutical companies such as Sanofi, AbbVie, and Teva, which are involved in the production and distribution of valproate and related medications, among others.
What are the growth factors driving the Fetal Valproate Syndrome Market?
The growth of the Fetal Valproate Syndrome Market is driven by increasing awareness of the syndrome, advancements in prenatal care, and the rising incidence of epilepsy among women of childbearing age.
What challenges does the Fetal Valproate Syndrome Market face?
The Fetal Valproate Syndrome Market faces challenges such as regulatory scrutiny regarding the use of valproate during pregnancy, potential legal liabilities for manufacturers, and the need for better patient education.
What opportunities exist in the Fetal Valproate Syndrome Market?
Opportunities in the Fetal Valproate Syndrome Market include the development of safer alternative treatments for epilepsy, increased research funding for understanding the syndrome, and enhanced support services for affected families.
What trends are emerging in the Fetal Valproate Syndrome Market?
Emerging trends in the Fetal Valproate Syndrome Market include a focus on personalized medicine, the integration of genetic testing in prenatal care, and growing advocacy for awareness and support for affected individuals.
Fetal Valproate Syndrome Market
| Segmentation | Details |
|---|---|
| Drug Type | Valproic Acid, Sodium Valproate, Valproate Semisodium |
| Diagnosis | Prenatal Diagnosis, Postnatal Diagnosis |
| End User | Hospitals, Clinics, Research Institutes |
| Region | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Fetal Valproate Syndrome Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at