444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$2750
Market Overview
The Europe biopsy devices market is a rapidly growing sector within the medical industry. Biopsy devices play a crucial role in the diagnosis and treatment of various diseases, including cancer. These devices enable healthcare professionals to extract tissue samples from the body for laboratory analysis, aiding in the identification of diseases and the formulation of appropriate treatment plans. The market for biopsy devices in Europe is witnessing significant growth due to advancements in technology, increasing prevalence of chronic diseases, and rising awareness among patients and healthcare providers.
Meaning
Biopsy devices refer to medical instruments used to collect tissue samples from the body for diagnostic purposes. These devices are primarily used in the detection and evaluation of cancerous and non-cancerous tumors, as well as other diseases. The tissue samples obtained through biopsies are examined by pathologists, who provide insights into the nature of the disease and guide healthcare professionals in determining the most suitable treatment approach.
Executive Summary
The Europe biopsy devices market is experiencing robust growth, driven by factors such as technological advancements, the increasing incidence of chronic diseases, and rising demand for minimally invasive procedures. The market is characterized by intense competition among key players, who are continuously focusing on product development and innovation to gain a competitive edge. Additionally, favorable reimbursement policies and government initiatives supporting early disease detection and treatment further contribute to the market’s growth.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
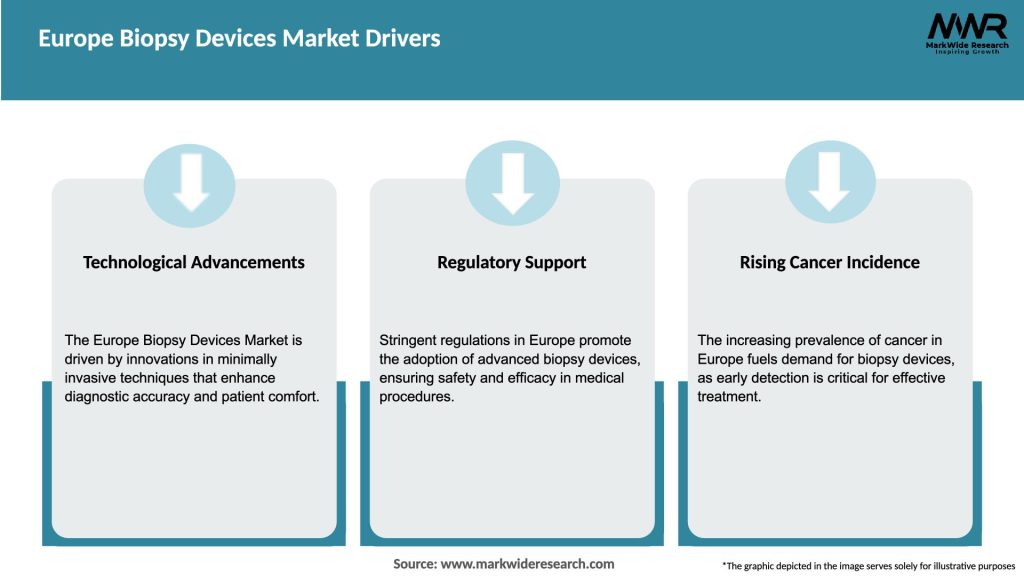
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The Europe biopsy devices market is characterized by intense competition among key players, who are continuously investing in research and development to introduce innovative products. Technological advancements, such as the integration of artificial intelligence and machine learning algorithms, are revolutionizing biopsy procedures, enhancing their accuracy and efficiency. Moreover, strategic collaborations between market players and healthcare providers are further propelling market growth by expanding the reach and accessibility of biopsy devices.
Regional Analysis
Europe is a significant market for biopsy devices, with several countries witnessing substantial growth. Germany, France, and the United Kingdom are the major contributors to the regional market. These countries have well-established healthcare infrastructure, favorable reimbursement policies, and a high prevalence of chronic diseases, driving the demand for biopsy devices. Additionally, the increasing adoption of advanced technologies and the presence of key market players in these countries contribute to their dominance in the European market.
Competitive Landscape
Leading Companies in the Europe Biopsy Devices Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
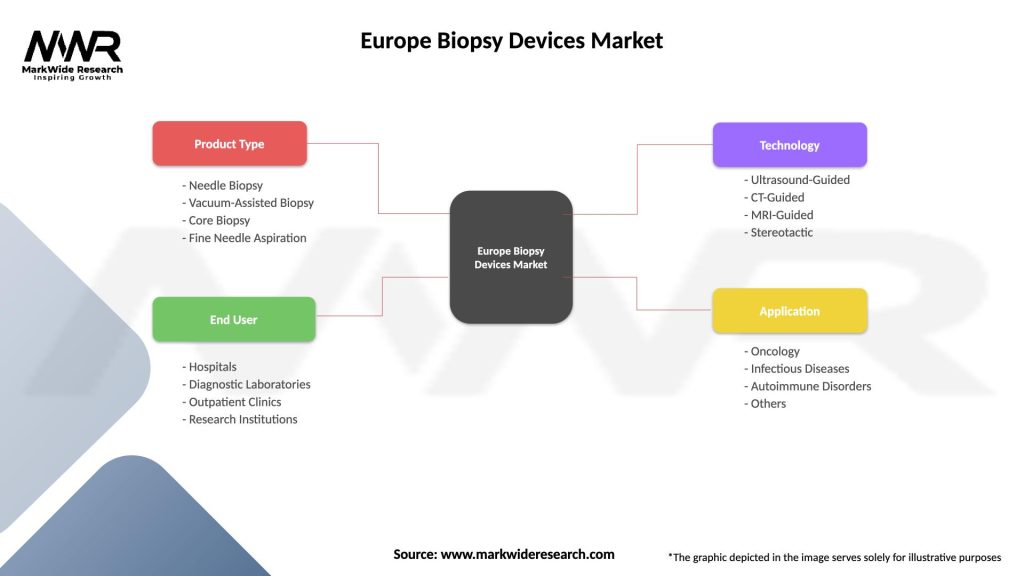
Segmentation
The Europe biopsy devices market can be segmented based on product type, application, end-user, and geography. By product type, the market can be categorized into needle-based biopsy devices, surgical biopsy devices, and others. Based on application, the market can be divided into breast biopsy, lung biopsy, prostate biopsy, liver biopsy, and others. The end-users of biopsy devices include hospitals, diagnostic centers, and research institutions.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Covid-19 Impact
The Covid-19 pandemic had a significant impact on the Europe biopsy devices market. The prioritization of resources and healthcare infrastructure towards managing the pandemic led to a temporary slowdown in elective procedures, including biopsies. However, as the situation improves and healthcare services resume normal operations, the demand for biopsy devices is expected to rebound. The importance of early disease detection and treatment has been emphasized during the pandemic, further driving the need for biopsy procedures.
Key Industry Developments
Analyst Suggestions
Future Outlook
The Europe biopsy devices market is poised for substantial growth in the coming years. Technological advancements, increasing prevalence of chronic diseases, and the focus on personalized medicine are expected to drive market expansion. Furthermore, the integration of artificial intelligence and machine learning algorithms in biopsy devices, along with the development of liquid biopsy techniques, will shape the future of the market. Collaborations and strategic partnerships among key industry players will continue to play a crucial role in driving innovation and market growth.
Conclusion
The Europe biopsy devices market is witnessing significant growth, driven by factors such as technological advancements, increasing prevalence of chronic diseases, and rising awareness among patients and healthcare providers. The market presents numerous opportunities for industry participants, including medical device manufacturers and distributors. However, challenges such as high costs and regulatory requirements need to be addressed to ensure market sustainability. With continued investment in research and development and a focus on personalized medicine, the Europe biopsy devices market is poised for a promising future.
What are biopsy devices in the context of the Europe Biopsy Devices Market?
Biopsy devices are medical instruments used to extract tissue samples from the body for diagnostic purposes. In the Europe Biopsy Devices Market, these devices play a crucial role in detecting diseases such as cancer and infections.
Who are the key players in the Europe Biopsy Devices Market?
Key players in the Europe Biopsy Devices Market include companies like Medtronic, Boston Scientific, and B. Braun, which are known for their innovative biopsy solutions and technologies, among others.
What are the main drivers of growth in the Europe Biopsy Devices Market?
The growth of the Europe Biopsy Devices Market is driven by factors such as the increasing prevalence of cancer, advancements in minimally invasive procedures, and the rising demand for early diagnosis and personalized medicine.
What challenges does the Europe Biopsy Devices Market face?
The Europe Biopsy Devices Market faces challenges such as stringent regulatory requirements, high costs associated with advanced biopsy technologies, and the need for skilled professionals to perform procedures.
What opportunities exist in the Europe Biopsy Devices Market?
Opportunities in the Europe Biopsy Devices Market include the development of novel biopsy techniques, the integration of artificial intelligence in diagnostic processes, and the expansion of telemedicine services for remote consultations.
What trends are shaping the Europe Biopsy Devices Market?
Trends in the Europe Biopsy Devices Market include the increasing adoption of image-guided biopsy procedures, the rise of liquid biopsy technologies, and a growing focus on patient-centric approaches in diagnostic practices.
Europe Biopsy Devices Market
| Segmentation Details | Description |
|---|---|
| Product Type | Needle Biopsy, Vacuum-Assisted Biopsy, Core Biopsy, Fine Needle Aspiration |
| End User | Hospitals, Diagnostic Laboratories, Outpatient Clinics, Research Institutions |
| Technology | Ultrasound-Guided, CT-Guided, MRI-Guided, Stereotactic |
| Application | Oncology, Infectious Diseases, Autoimmune Disorders, Others |
Leading Companies in the Europe Biopsy Devices Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at