444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The Epinephrine Injection market is a crucial segment within the pharmaceutical industry. Epinephrine, also known as adrenaline, is a hormone and neurotransmitter that plays a vital role in the body’s response to stress and emergency situations. Epinephrine injections are widely used in the treatment of severe allergic reactions, such as anaphylaxis, as well as in the management of certain cardiac conditions.
Meaning
Epinephrine injections, also referred to as adrenaline injections, are a life-saving medication administered via injection for the immediate treatment of severe allergic reactions. The injections work by constricting blood vessels, opening airways, and increasing heart rate, thereby counteracting the potentially life-threatening symptoms of anaphylaxis.
Executive Summary
The Epinephrine Injection market has witnessed significant growth in recent years, driven by the rising incidence of severe allergic reactions and the increasing awareness about the importance of immediate medical intervention in such cases. The market is characterized by the presence of several key players offering a wide range of epinephrine injection products, ensuring a competitive landscape. Additionally, technological advancements and the development of auto-injectors have further propelled market growth.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The Epinephrine Injection market is influenced by various dynamic factors that shape its growth and competitiveness. These dynamics include market trends, customer preferences, regulatory landscape, and technological advancements. Understanding and adapting to these dynamics are crucial for market players to sustain their position and capitalize on growth opportunities.
Regional Analysis
The Epinephrine Injection market exhibits regional variations due to differences in allergy prevalence, healthcare infrastructure, and regulatory frameworks. North America holds a significant market share, driven by a high incidence of allergies and well-established healthcare systems. Europe also accounts for a substantial market share, with countries like Germany and France contributing to market growth. Asia Pacific is anticipated to witness rapid growth due to the increasing awareness about anaphylaxis and improving healthcare facilities in countries such as China, India, and Japan.
Competitive Landscape
Leading Companies in the Epinephrine Injection Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
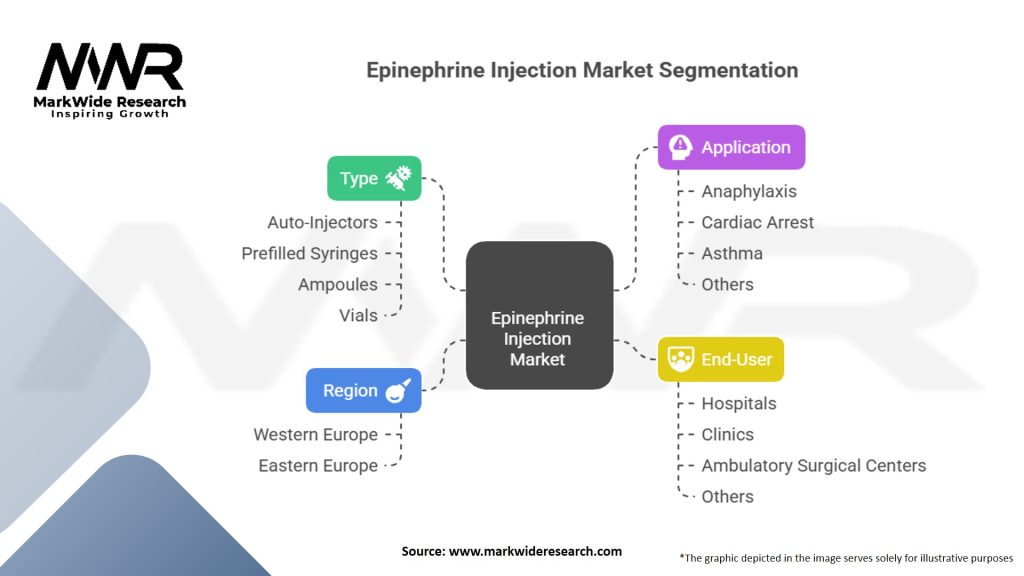
Segmentation
The Epinephrine Injection market can be segmented based on product type, distribution channel, and end-user.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Covid-19 Impact
The Covid-19 pandemic has had a mixed impact on the Epinephrine Injection market. While the overall demand for epinephrine injections remained stable, there were some challenges faced by market players. The pandemic led to disruptions in the supply chain, impacting the manufacturing and distribution of epinephrine injections. However, the heightened focus on healthcare preparedness and emergency response during the pandemic brought increased attention to the importance of immediate treatment for severe allergic reactions.
One significant impact of the pandemic was the delay in elective medical procedures and non-essential healthcare visits, which temporarily reduced the demand for epinephrine injections in certain healthcare settings. However, as the healthcare system gradually resumed normal operations, the demand for these life-saving injections started to recover.
Moreover, the increased emphasis on personal hygiene and safety measures during the pandemic highlighted the importance of epinephrine auto-injectors for individuals with known allergies and the need for them to carry their medication at all times. This awareness contributed to sustained demand for auto-injectors, as people became more conscious of the potential risks associated with severe allergic reactions.
Additionally, the Covid-19 pandemic prompted several manufacturers to adapt their production facilities to meet the increased demand for medical supplies and equipment. Some companies expanded their manufacturing capacities to ensure a steady supply of epinephrine injections and mitigate any potential shortages.
Furthermore, the pandemic underscored the significance of telemedicine and remote healthcare services. Virtual consultations and telehealth platforms became more prevalent during the pandemic, allowing healthcare professionals to remotely assess and advise patients regarding severe allergic reactions and the use of epinephrine injections. This shift in healthcare delivery opened up new avenues for reaching patients in remote areas and improved accessibility to medical advice.
Overall, while the Covid-19 pandemic presented challenges to the Epinephrine Injection market, it also highlighted the critical role of these medications in emergency medical care. As the world recovers from the pandemic and healthcare systems adapt to the new normal, the market is expected to regain momentum and continue its growth trajectory.
Key Industry Developments
Analyst Suggestions
Future Outlook
The future of the Epinephrine Injection market looks promising, with sustained growth expected in the coming years. Factors such as the increasing prevalence of severe allergic reactions, growing awareness about anaphylaxis, and technological advancements in delivery systems will drive market expansion. The market is likely to witness further product innovations, such as extended-release formulations, needle-free injection systems, and advancements in auto-injector design.
Emerging markets in Asia Pacific and Latin America present significant growth opportunities, as improving healthcare infrastructure and rising awareness contribute to increased demand for epinephrine injections. Market players should focus on expanding their presence in these regions through strategic partnerships and localized marketing strategies.
However, challenges such as the high cost of epinephrine injections and stringent regulatory requirements need to be addressed. Market players should strive to develop cost-effective alternatives and ensure compliance with regulatory standards to maintain product quality and safety.
Overall, with a combination of innovation, market expansion, patient education, and strategic collaborations, the Epinephrine Injection market is expected to witness steady growth and offer substantial opportunities for industry participants and stakeholders.
Conclusion
The Epinephrine Injection market plays a crucial role in the treatment of severe allergic reactions, providing immediate medical intervention to counteract life-threatening symptoms. The market is driven by factors such as the increasing prevalence of allergies, rising awareness about anaphylaxis, and technological advancements in delivery systems. The demand for epinephrine injections, especially auto-injectors, continues to grow, fueled by the need for convenient and user-friendly treatment options.
While the market offers significant growth opportunities, challenges such as the high cost of epinephrine injections, stringent regulations, and adverse effects associated with certain brands need to be addressed. Market players should focus on continuous innovation, market expansion in emerging economies, patient education, and compliance with regulatory standards to maintain product quality and safety.
What is Epinephrine Injection?
Epinephrine Injection is a medication used primarily to treat severe allergic reactions, known as anaphylaxis. It works by constricting blood vessels, relaxing airway muscles, and increasing heart rate, making it essential in emergency medical situations.
What are the key companies in the Epinephrine Injection market?
Key companies in the Epinephrine Injection market include Mylan N.V., Teva Pharmaceutical Industries Ltd., and Amgen Inc., among others.
What are the growth factors driving the Epinephrine Injection market?
The growth of the Epinephrine Injection market is driven by the increasing prevalence of allergies and anaphylaxis, rising awareness about emergency treatment options, and advancements in drug delivery systems.
What challenges does the Epinephrine Injection market face?
Challenges in the Epinephrine Injection market include the high cost of auto-injectors, potential side effects, and the need for proper training in their use to ensure effective administration during emergencies.
What opportunities exist in the Epinephrine Injection market?
Opportunities in the Epinephrine Injection market include the development of new formulations, expansion into emerging markets, and increasing partnerships between pharmaceutical companies and healthcare providers to improve access.
What trends are shaping the Epinephrine Injection market?
Trends in the Epinephrine Injection market include the rise of personalized medicine, innovations in auto-injector technology, and a growing emphasis on patient education regarding allergy management.
Epinephrine Injection Market Segmentation
| Segmentation Details | Information |
|---|---|
| Type | Auto-Injectors, Prefilled Syringes, Ampoules, Vials |
| Application | Anaphylaxis, Cardiac Arrest, Asthma, Others |
| End-User | Hospitals, Clinics, Ambulatory Surgical Centers, Others |
| Region | Western Europe, Eastern Europe |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Epinephrine Injection Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at