444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The Enteric-Coated and Controlled-Release Tablets and Capsules market is a rapidly growing sector within the pharmaceutical industry. These specialized forms of medication delivery offer unique benefits to both patients and healthcare providers. Enteric-coated tablets and capsules are designed to protect the drug from the acidic environment of the stomach, ensuring that it is released in the intestines where it can be absorbed more effectively. Controlled-release tablets and capsules, on the other hand, provide a slow and steady release of the drug over an extended period of time, allowing for reduced dosing frequency and improved patient compliance.
Meaning
Enteric-coated and controlled-release tablets and capsules are pharmaceutical formulations that have been specifically designed to optimize the delivery of medications to the target site in the body. Enteric coating refers to a protective layer that prevents the drug from being released in the stomach and instead allows it to dissolve and be absorbed in the intestines. Controlled-release formulations, on the other hand, release the drug at a predetermined rate, maintaining therapeutic levels in the bloodstream over an extended period. These technologies are employed to improve drug efficacy, reduce side effects, and enhance patient convenience.
Executive Summary
The Enteric-Coated and Controlled-Release Tablets and Capsules market is witnessing substantial growth due to the increasing demand for improved drug delivery systems. The rise in chronic diseases, such as cardiovascular disorders and diabetes, coupled with the need for patient-friendly medication regimens, has fueled the adoption of enteric-coated and controlled-release formulations. Furthermore, advancements in pharmaceutical manufacturing technologies and the expanding geriatric population have contributed to market expansion. The market is characterized by intense competition among key players, who are focusing on research and development activities to introduce innovative products.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
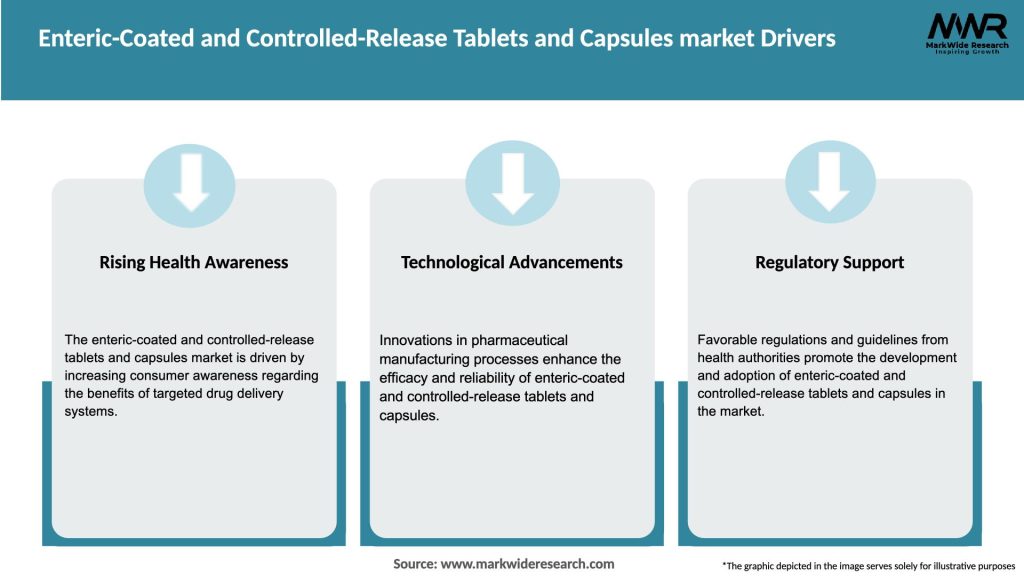
The Enteric-Coated and Controlled-Release Tablets and Capsules market is driven by several factors that contribute to its rapid growth. Firstly, the increasing prevalence of chronic diseases, such as cardiovascular disorders, diabetes, and gastrointestinal disorders, has created a demand for medications that can provide prolonged therapeutic effects. Enteric-coated and controlled-release formulations offer a solution by ensuring a steady and controlled release of the drug, reducing the need for frequent dosing and improving patient compliance. Secondly, advancements in pharmaceutical manufacturing technologies have made it possible to develop more efficient and effective drug delivery systems. These technological advancements have enabled the development of enteric-coated and controlled-release tablets and capsules that can deliver drugs at specific sites within the body, optimizing their therapeutic benefits.
Market Restraints
Despite the numerous advantages offered by enteric-coated and controlled-release tablets and capsules, there are certain challenges and restraints that the market faces. One of the primary restraints is the high cost associated with the development and production of these specialized formulations. The incorporation of advanced technologies and specialized coatings increases the manufacturing costs, which can make these products more expensive for patients and healthcare systems. Additionally, the regulatory requirements for the approval of enteric-coated and controlled-release formulations can be complex and time-consuming, leading to delays in product launches. Moreover, the lack of awareness among healthcare professionals and patients about the benefits and availability of these formulations can hinder market growth.
Market Opportunities
The Enteric-Coated and Controlled-Release Tablets and Capsules market offers significant opportunities for growth and expansion. With the increasing focus on personalized medicine and targeted drug deliverysystems, there is a growing demand for customized formulations that can deliver drugs to specific sites within the body. Enteric-coated and controlled-release tablets and capsules have the potential to meet this demand by providing targeted drug delivery and optimizing therapeutic outcomes. Additionally, the expanding geriatric population, which is more susceptible to chronic diseases and requires long-term medication regimens, presents a substantial market opportunity. Furthermore, the rising investments in research and development activities by key market players indicate a strong commitment to innovation and the introduction of new and improved formulations. These developments can lead to the discovery of novel drug delivery technologies and expand the market further.
Market Dynamics
The Enteric-Coated and Controlled-Release Tablets and Capsules market is characterized by dynamic factors that influence its growth and evolution. Technological advancements play a crucial role in shaping the market landscape. The development of innovative coatings and manufacturing processes has enabled the production of enteric-coated and controlled-release formulations with improved drug release profiles and enhanced therapeutic efficacy. Additionally, the market is driven by changing patient preferences and the increasing demand for patient-friendly medication options. Patients are seeking convenient and easy-to-administer dosage forms that can improve treatment outcomes and overall quality of life. Furthermore, the market dynamics are influenced by regulatory policies and guidelines that govern the approval and commercialization of these specialized formulations. Compliance with regulatory standards and obtaining necessary approvals are critical for market players to bring their products to market successfully.
Regional Analysis
The Enteric-Coated and Controlled-Release Tablets and Capsules market exhibits regional variations in terms of market size, growth rate, and adoption. North America holds a significant share of the market due to the presence of well-established pharmaceutical companies and a high prevalence of chronic diseases in the region. The demand for enteric-coated and controlled-release formulations is also driven by the advanced healthcare infrastructure and the availability of reimbursement policies. Europe is another key market for enteric-coated and controlled-release products, fueled by the increasing geriatric population and the focus on patient-centric healthcare delivery. Asia Pacific is expected to witness substantial growth in the market due to the rising healthcare expenditure, expanding pharmaceutical industry, and improving healthcare infrastructure in countries such as China and India. Latin America and the Middle East & Africa are also emerging markets with untapped growth potential, driven by the increasing awareness about advanced drug delivery systems and the growing prevalence of chronic diseases.
Competitive Landscape
Leading Companies in the Enteric-Coated and Controlled-Release Tablets and Capsules Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Segmentation
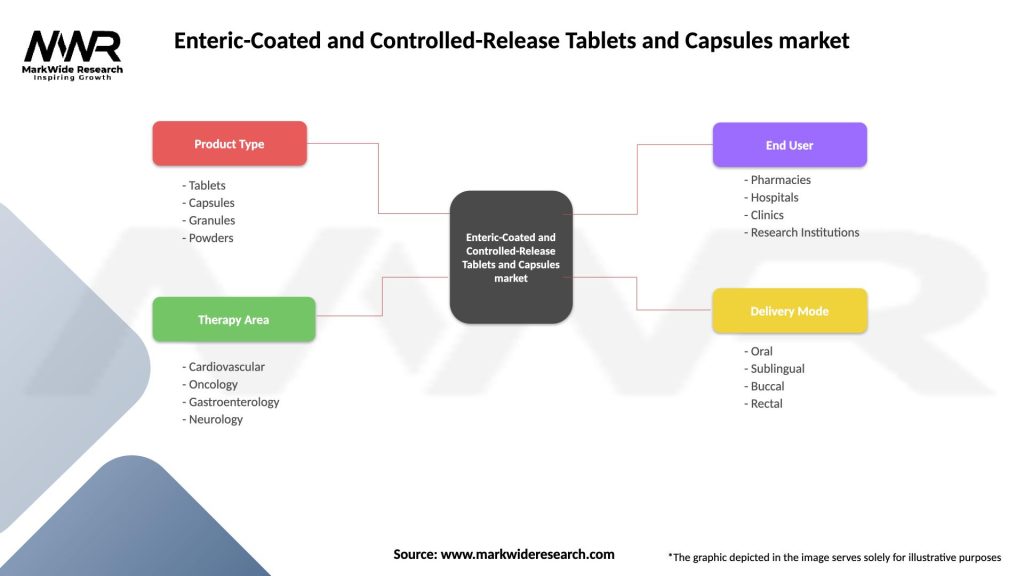
The Enteric-Coated and Controlled-Release Tablets and Capsules market can be segmented based on various factors, including drug class, therapeutic area, and distribution channel. Drug classes include analgesics, anti-inflammatory drugs, cardiovascular drugs, gastrointestinal drugs, and others. Therapeutic areas encompass cardiovascular disorders, gastrointestinal disorders, neurology, oncology, and others. Distribution channels comprise hospitals pharmacies, retail pharmacies, online pharmacies, and drug stores. This segmentation allows for a comprehensive understanding of the market dynamics and aids in targeted marketing strategies and product development.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Market Key Trends
Covid-19 Impact
The Covid-19 pandemic has had both positive and negative impacts on the Enteric-Coated and Controlled-Release Tablets and Capsules market. On one hand, the increased focus on healthcare and the urgency to find effective treatments and vaccines have driven investments in pharmaceutical research and development, including the development of advanced drug delivery systems. This has created opportunities for market growth and innovation.
However, the pandemic has also posed challenges in terms of disrupted supply chains, manufacturing delays, and regulatory disruptions. Lockdown measures and restrictions on international trade have affected the production and distribution of pharmaceutical products, including enteric-coated and controlled-release formulations. Moreover, the diverted focus towards Covid-19 treatments and vaccines has led to delays in non-pandemic-related drug development and approvals.
Nevertheless, the long-term impact of the pandemic on the Enteric-Coated and Controlled-Release Tablets and Capsules market is expected to be positive, as the demand for improved drug delivery systems and patient-friendly medication options continues to rise.
Key Industry Developments
Analyst Suggestions
Future Outlook
The Enteric-Coated and Controlled-Release Tablets and Capsules market is poised for significant growth in the coming years. Factors such as the increasing prevalence of chronic diseases, advancements in coating technologies, and the demand for patient-friendly medication options are expected to drive market expansion. The ongoing focus on personalized medicine and targeted drug delivery will fuel the development of innovative formulations and technologies. However, challenges related to high manufacturing costs, regulatory requirements, and marketcompetition will need to be addressed to ensure sustained growth.
The market is anticipated to witness substantial opportunities in emerging economies, where improving healthcare infrastructure and increasing healthcare expenditure are creating a favorable environment for market growth. The expanding geriatric population in many regions will also contribute to the market’s expansion, as elderly individuals often require long-term medication regimens and can benefit from the convenience and improved therapeutic outcomes offered by enteric-coated and controlled-release formulations.
Technological advancements, particularly in coating technologies and nanotechnology applications, will continue to shape the market landscape. The development of advanced coatings, such as pH-sensitive and enzyme-sensitive coatings, will enable precise drug release and targeted delivery. Nanoparticles and nanocarriers will further enhance drug solubility and bioavailability, revolutionizing drug delivery optimization.
Strategic collaborations and partnerships will play a crucial role in the future of the market. Companies will seek to leverage the expertise of research institutions and contract manufacturing organizations to accelerate product development and enhance manufacturing capabilities. By combining resources and knowledge, industry participants can strengthen their market presence and introduce innovative formulations more efficiently.
Conclusion
In conclusion, the Enteric-Coated and Controlled-Release Tablets and Capsules market is poised for significant growth and innovation. The demand for enhanced drug delivery systems, personalized medicine, and patient-friendly medication options will continue to drive market expansion. Industry participants must focus on research and development, collaboration, market education, and regulatory compliance to capitalize on the market’s opportunities and address its challenges. With these strategies in place, the future outlook for the Enteric-Coated and Controlled-Release Tablets and Capsules market is promising.
What is Enteric-Coated and Controlled-Release Tablets and Capsules?
Enteric-Coated and Controlled-Release Tablets and Capsules are pharmaceutical formulations designed to release their active ingredients at specific sites in the gastrointestinal tract. These formulations enhance drug absorption and minimize side effects by protecting the active ingredients from the acidic environment of the stomach.
What are the key players in the Enteric-Coated and Controlled-Release Tablets and Capsules market?
Key players in the Enteric-Coated and Controlled-Release Tablets and Capsules market include companies like Pfizer, Johnson & Johnson, and Novartis, which are known for their innovative drug delivery systems and extensive product portfolios, among others.
What are the growth factors driving the Enteric-Coated and Controlled-Release Tablets and Capsules market?
The growth of the Enteric-Coated and Controlled-Release Tablets and Capsules market is driven by the increasing demand for advanced drug delivery systems, rising prevalence of chronic diseases, and the need for improved patient compliance. Additionally, technological advancements in formulation development are contributing to market expansion.
What challenges does the Enteric-Coated and Controlled-Release Tablets and Capsules market face?
The Enteric-Coated and Controlled-Release Tablets and Capsules market faces challenges such as high manufacturing costs, stringent regulatory requirements, and potential issues with drug stability and bioavailability. These factors can hinder the development and commercialization of new formulations.
What opportunities exist in the Enteric-Coated and Controlled-Release Tablets and Capsules market?
Opportunities in the Enteric-Coated and Controlled-Release Tablets and Capsules market include the growing focus on personalized medicine, the development of novel drug delivery technologies, and the increasing investment in research and development. These factors are expected to enhance the market landscape in the coming years.
What trends are shaping the Enteric-Coated and Controlled-Release Tablets and Capsules market?
Trends in the Enteric-Coated and Controlled-Release Tablets and Capsules market include the rise of biopharmaceuticals, advancements in nanotechnology for drug delivery, and the increasing use of digital health technologies to monitor patient adherence. These trends are influencing the future direction of the market.
Enteric-Coated and Controlled-Release Tablets and Capsules market
| Segmentation Details | Description |
|---|---|
| Product Type | Tablets, Capsules, Granules, Powders |
| Therapy Area | Cardiovascular, Oncology, Gastroenterology, Neurology |
| End User | Pharmacies, Hospitals, Clinics, Research Institutions |
| Delivery Mode | Oral, Sublingual, Buccal, Rectal |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Enteric-Coated and Controlled-Release Tablets and Capsules Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at