444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The Cytotoxic Safety Cabinets Market is experiencing substantial growth attributed to the increasing prevalence of cancer and the rising demand for safe handling of cytotoxic drugs in healthcare facilities. Cytotoxic safety cabinets are essential equipment designed to provide a controlled environment for the preparation, handling, and administration of cytotoxic drugs, ensuring operator safety, environmental protection, and product integrity. With advancements in technology and stringent regulatory guidelines, the cytotoxic safety cabinets market is poised for further expansion.
Meaning
Cytotoxic safety cabinets, also known as cytotoxic drug safety cabinets or biological safety cabinets (BSCs) class II type B3, are specialized containment devices designed to protect laboratory personnel, patients, and the environment from exposure to hazardous drugs such as cytotoxic agents, antineoplastic drugs, and chemotherapy medications. These cabinets employ high-efficiency particulate air (HEPA) filtration and airflow systems to maintain a clean, sterile, and controlled work environment, minimizing the risk of contamination and airborne exposure during drug preparation and handling procedures.
Executive Summary
The Cytotoxic Safety Cabinets Market is witnessing rapid growth driven by factors such as the increasing incidence of cancer, growing adoption of chemotherapy drugs, and rising awareness of occupational hazards associated with cytotoxic drug exposure. Key players in the market are investing in product innovation, quality assurance, and regulatory compliance to meet the evolving needs of healthcare providers and ensure safe handling practices in oncology and pharmacy settings. The market is poised for continued expansion as stakeholders prioritize patient safety, regulatory compliance, and quality assurance in cytotoxic drug handling.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The Cytotoxic Safety Cabinets Market is characterized by dynamic growth driven by evolving healthcare needs, technological innovations, and regulatory trends. Market dynamics are influenced by factors such as cancer epidemiology, chemotherapy utilization, healthcare policies, and occupational safety regulations, shaping the competitive landscape, innovation pipeline, and market expansion strategies of key players in the oncology and pharmacy equipment industry.
Regional Analysis
Competitive Landscape
Leading Companies in Cytotoxic Safety Cabinets Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
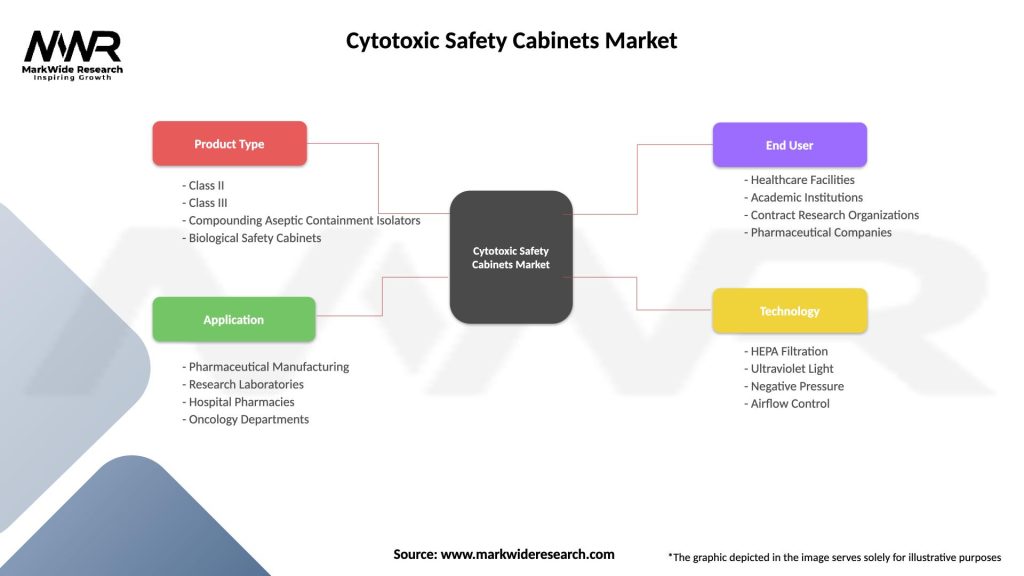
Segmentation
The Cytotoxic Safety Cabinets Market can be segmented based on cabinet type, airflow pattern, application, end-user, and region.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Market Key Trends
COVID-19 Impact
The COVID-19 pandemic has influenced the Cytotoxic Safety Cabinets Market by increasing demand for safety equipment, infection control measures, and regulatory compliance in healthcare facilities. Cytotoxic safety cabinets play a role in protecting healthcare workers, patients, and the environment from infectious diseases, aerosolized contaminants, and airborne pathogens, supporting infection prevention and control initiatives in pharmacy, laboratory, and clinical settings.
Key Industry Developments
Analyst Suggestions
Future Outlook
The Cytotoxic Safety Cabinets Market is poised for continued growth, driven by increasing demand for safe handling practices, regulatory compliance requirements, and technological advancements in safety cabinet design and performance. The market is expected to witness further innovation, market expansion, and adoption of safety cabinet solutions as stakeholders prioritize user safety, patient care, and environmental protection in cytotoxic drug handling operations.
Conclusion
In conclusion, the Cytotoxic Safety Cabinets Market offers significant growth opportunities for industry participants, driven by increasing demand for safe handling practices, regulatory compliance requirements, and technological advancements in safety cabinet design and performance. By leveraging innovation, quality assurance, and regulatory compliance, stakeholders can contribute to advancing safety practices, enhancing user safety, and improving patient care in cytotoxic drug handling operations.
What is Cytotoxic Safety Cabinets?
Cytotoxic safety cabinets are specialized enclosures designed to protect users from exposure to hazardous drugs and materials, ensuring a sterile environment for handling cytotoxic substances. They are commonly used in laboratories, hospitals, and pharmaceutical settings.
What are the key players in the Cytotoxic Safety Cabinets Market?
Key players in the Cytotoxic Safety Cabinets Market include companies like Thermo Fisher Scientific, Esco Micro Pte Ltd, and Baker Company, which are known for their innovative designs and safety features in laboratory equipment, among others.
What are the growth factors driving the Cytotoxic Safety Cabinets Market?
The growth of the Cytotoxic Safety Cabinets Market is driven by the increasing demand for safe handling of hazardous drugs in healthcare settings, rising awareness of occupational safety, and advancements in cabinet technology that enhance user protection and efficiency.
What challenges does the Cytotoxic Safety Cabinets Market face?
Challenges in the Cytotoxic Safety Cabinets Market include high initial costs of advanced cabinets, the need for regular maintenance and certification, and regulatory compliance that can vary by region, impacting market accessibility.
What opportunities exist in the Cytotoxic Safety Cabinets Market?
Opportunities in the Cytotoxic Safety Cabinets Market include the development of more energy-efficient models, integration of smart technology for monitoring safety conditions, and expansion into emerging markets where healthcare infrastructure is improving.
What trends are shaping the Cytotoxic Safety Cabinets Market?
Trends in the Cytotoxic Safety Cabinets Market include a growing emphasis on sustainability, with manufacturers focusing on eco-friendly materials, and the increasing adoption of modular designs that allow for customization based on specific laboratory needs.
Cytotoxic Safety Cabinets Market
| Segmentation Details | Description |
|---|---|
| Product Type | Class II, Class III, Compounding Aseptic Containment Isolators, Biological Safety Cabinets |
| Application | Pharmaceutical Manufacturing, Research Laboratories, Hospital Pharmacies, Oncology Departments |
| End User | Healthcare Facilities, Academic Institutions, Contract Research Organizations, Pharmaceutical Companies |
| Technology | HEPA Filtration, Ultraviolet Light, Negative Pressure, Airflow Control |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in Cytotoxic Safety Cabinets Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at