444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview: The Celecoxib API Market encompasses the global trade and production of Celecoxib Active Pharmaceutical Ingredients (APIs), which are key components in the manufacturing of medications used to treat pain, inflammation, and arthritis. Celecoxib, a nonsteroidal anti-inflammatory drug (NSAID), is widely prescribed for conditions such as osteoarthritis, rheumatoid arthritis, and acute pain. The market for Celecoxib APIs is driven by factors such as the increasing prevalence of chronic pain conditions, growing demand for generic medications, and ongoing research into the therapeutic applications of Celecoxib across various medical specialties.
Meaning: Celecoxib API refers to the active pharmaceutical ingredient, Celecoxib, which is the chemical compound responsible for the therapeutic effects of medications such as Celebrex. Celecoxib belongs to the class of NSAIDs known as selective COX-2 inhibitors, which work by inhibiting the enzyme cyclooxygenase-2 (COX-2) involved in the inflammatory response. Celecoxib APIs are synthesized through chemical processes and serve as the foundation for formulating oral tablets, capsules, and other dosage forms used in pain management and anti-inflammatory therapies.
Executive Summary: The Celecoxib API Market is experiencing steady growth driven by the rising demand for pain relief medications, expanding applications of Celecoxib in various medical conditions, and increasing investment in pharmaceutical research and development. Key market players are focused on expanding their manufacturing capacities, improving production efficiency, and ensuring regulatory compliance to meet the growing demand for Celecoxib APIs worldwide. With the patent expiration of branded formulations and the emergence of generic alternatives, the market for Celecoxib APIs is poised for continued expansion in the foreseeable future.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights:
Market Drivers:
Market Restraints:
Market Opportunities:
Market Dynamics: The Celecoxib API Market is influenced by dynamic factors such as regulatory changes, patent expirations, healthcare policies, and market competition. Market players need to adapt to these dynamics by investing in research and development, regulatory compliance, and market intelligence to capitalize on emerging opportunities and address evolving challenges in the global pharmaceutical landscape.
Regional Analysis: The Celecoxib API Market exhibits regional variations shaped by factors such as population demographics, disease prevalence, healthcare expenditure, and regulatory frameworks. Developed regions such as North America and Europe dominate the market due to established pharmaceutical industries, robust research infrastructure, and high healthcare spending. Emerging markets in Asia Pacific, Latin America, and the Middle East offer growth opportunities driven by rising disposable incomes, urbanization, and expanding access to healthcare services.
Competitive Landscape:
Leading Companies in Celecoxib API Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
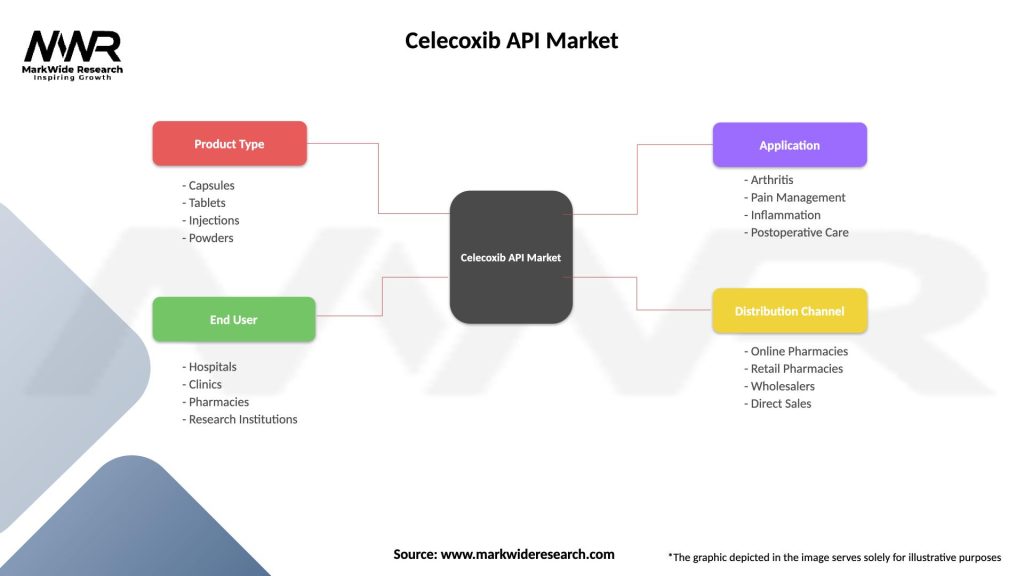
Segmentation: The Celecoxib API Market can be segmented based on synthesis method, purity level, formulation type, end-user, and geography. Synthesis methods include chemical synthesis, biotechnological synthesis, and semisynthetic approaches. Purity levels range from standard-grade APIs to high-purity, pharmaceutical-grade materials suitable for injectable formulations and sterile dosage forms. Formulation types encompass oral tablets, capsules, suspensions, and injectable solutions used in hospital pharmacies, compounding facilities, and retail pharmacies.
Category-wise Insights:
Key Benefits for Industry Participants and Stakeholders:
SWOT Analysis:
Market Key Trends:
Covid-19 Impact: The Covid-19 pandemic has underscored the importance of pharmaceutical innovation, supply chain resilience, and healthcare preparedness in addressing global health challenges. While the crisis has disrupted clinical trials, drug development timelines, and market access for new therapies, it has also accelerated digital transformation, telemedicine adoption, and regulatory flexibility in the pharmaceutical industry.
Key Industry Developments:
Analyst Suggestions:
Future Outlook: The Celecoxib API Market is poised for steady growth driven by the increasing demand for pain relief medications, expanding therapeutic applications of Celecoxib, and advancements in pharmaceutical manufacturing and drug delivery technologies. Continued investment in research, regulatory compliance, and market expansion strategies will shape the future of Celecoxib APIs as essential components of modern pharmacotherapy for pain and inflammation management.
Conclusion: The Celecoxib API Market represents a vital segment of the global pharmaceutical industry, supplying active ingredients for medications used in pain management, arthritis treatment, and anti-inflammatory therapy. With their proven efficacy, safety profile, and therapeutic versatility, Celecoxib APIs play a central role in addressing unmet medical needs, improving patient outcomes, and enhancing healthcare quality worldwide. By fostering innovation, collaboration, and regulatory compliance, stakeholders can contribute to advancing the Celecoxib API market and supporting the evolving needs of patients and healthcare providers in the 21st century.
What is Celecoxib API?
Celecoxib API refers to the active pharmaceutical ingredient used in the formulation of Celecoxib, a non-steroidal anti-inflammatory drug (NSAID) primarily used to treat pain and inflammation associated with conditions like arthritis and acute pain.
What are the key companies in the Celecoxib API Market?
Key companies in the Celecoxib API Market include Teva Pharmaceutical Industries, Aurobindo Pharma, and Mylan, among others.
What are the growth factors driving the Celecoxib API Market?
The growth of the Celecoxib API Market is driven by the increasing prevalence of chronic pain conditions, rising demand for effective pain management solutions, and advancements in pharmaceutical manufacturing technologies.
What challenges does the Celecoxib API Market face?
The Celecoxib API Market faces challenges such as stringent regulatory requirements, potential side effects associated with long-term use, and competition from alternative pain management therapies.
What opportunities exist in the Celecoxib API Market?
Opportunities in the Celecoxib API Market include the development of new formulations, expansion into emerging markets, and increasing collaborations between pharmaceutical companies for research and development.
What trends are shaping the Celecoxib API Market?
Trends in the Celecoxib API Market include a growing focus on personalized medicine, the rise of biosimilars, and increased investment in research to enhance drug efficacy and safety.
Celecoxib API Market
| Segmentation Details | Description |
|---|---|
| Product Type | Capsules, Tablets, Injections, Powders |
| End User | Hospitals, Clinics, Pharmacies, Research Institutions |
| Application | Arthritis, Pain Management, Inflammation, Postoperative Care |
| Distribution Channel | Online Pharmacies, Retail Pharmacies, Wholesalers, Direct Sales |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in Celecoxib API Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at