444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview: The CD CHO media market serves the biopharmaceutical industry by providing a specialized cell culture medium optimized for the growth and productivity of Chinese Hamster Ovary (CHO) cells. CHO cells are widely used in the production of therapeutic proteins, monoclonal antibodies, vaccines, and viral vectors due to their ability to express complex proteins with human-like glycosylation patterns. CD CHO media, formulated with specific nutrients, growth factors, and supplements, supports the robust growth and high-yield production of CHO cells in bioreactor systems, facilitating the scalable manufacturing of biologics for clinical and commercial applications. The market for CD CHO media is driven by factors such as the increasing demand for biopharmaceuticals, advancements in cell culture technology, and the growing trend of bioprocessing optimization for cost-effective production.
Meaning: CD CHO media refers to a chemically defined cell culture medium designed to support the growth, viability, and productivity of CHO cells in biopharmaceutical manufacturing processes. Unlike complex serum-containing media, CD CHO media contains defined components, such as amino acids, vitamins, minerals, and growth factors, with known concentrations and compositions. This enables precise control over cell culture conditions, reduces batch-to-batch variability, and enhances product consistency and quality. CD CHO media plays a critical role in bioprocess optimization, enabling biopharmaceutical companies to achieve higher cell densities, faster cell growth rates, and increased protein yields, ultimately leading to more efficient and cost-effective biologics production.
Executive Summary: The CD CHO media market is experiencing robust growth driven by the expanding biopharmaceutical industry’s need for reliable and scalable cell culture solutions. Key market players are investing in research and development initiatives to innovate new formulations, improve media performance, and optimize manufacturing processes. With the increasing adoption of cell-based therapies, gene therapies, and personalized medicines, the demand for CD CHO media is expected to grow further, supporting the development and commercialization of novel biologics for various therapeutic applications.

Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights:
Market Drivers:
Market Restraints:
Market Opportunities:
Market Dynamics: The CD CHO media market is characterized by dynamic interactions between technological advancements, regulatory requirements, market competition, and customer preferences. Market players need to continuously innovate, adapt, and differentiate their product offerings to meet the evolving needs of biopharmaceutical developers and manufacturers worldwide. Strategic collaborations, acquisitions, and partnerships play a key role in shaping the competitive landscape and driving market growth and innovation in the CD CHO media sector.
Regional Analysis: The CD CHO media market exhibits regional variations influenced by factors such as biopharmaceutical R&D investments, manufacturing infrastructure, regulatory frameworks, and market dynamics. North America dominates the market, owing to the presence of leading biopharmaceutical companies, academic research institutions, and established biomanufacturing facilities. Europe and Asia Pacific regions are witnessing rapid growth driven by increasing outsourcing activities, technology transfer initiatives, and government support for biopharmaceutical innovation and manufacturing.
Competitive Landscape:
Leading Companies in CD CHO Media Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Segmentation: The CD CHO media market can be segmented based on product type, formulation, application, end-user, and geography. Product types include liquid media, powdered media, and ready-to-use media formats. Formulations may vary in nutrient composition, pH, osmolality, and supplements tailored to specific cell culture requirements. Applications encompass monoclonal antibody production, recombinant protein expression, vaccine manufacturing, and viral vector production. End-users include biopharmaceutical companies, contract manufacturing organizations (CMOs), academic research laboratories, and bioprocessing facilities.
Category-wise Insights:
Key Benefits for Industry Participants and Stakeholders:
SWOT Analysis:
Market Key Trends:
Covid-19 Impact: The Covid-19 pandemic has underscored the importance of biopharmaceutical innovation, vaccine development, and global health security, driving investment in biomanufacturing capacity, supply chain resilience, and pandemic preparedness. The pandemic has accelerated the adoption of CD CHO media and other bioprocessing technologies for the rapid development and production of Covid-19 vaccines, monoclonal antibodies, and other biologic therapeutics. While the pandemic has presented challenges such as supply chain disruptions, manufacturing constraints, and regulatory pressures, it has also catalyzed collaboration, innovation, and investment in the biopharmaceutical industry, positioning CD CHO media suppliers for long-term growth and value creation.
Key Industry Developments:
Analyst Suggestions:
Future Outlook: The CD CHO media market is poised for significant growth driven by the increasing demand for biopharmaceuticals, cell-based therapies, and personalized medicines worldwide. As biopharmaceutical companies continue to invest in biomanufacturing capacity, process optimization, and technology innovation, the demand for high-performance CD CHO media formulations and related cell culture products is expected to grow, creating new opportunities for suppliers, manufacturers, and service providers in the biopharmaceutical value chain. By embracing innovation, collaboration, and regulatory compliance, stakeholders can navigate the evolving landscape of biopharmaceutical manufacturing and contribute to the advancement of healthcare through the development and commercialization of safe, effective, and affordable biologic therapies for patients worldwide.
Conclusion: The CD CHO media market plays a critical role in supporting the growth, productivity, and scalability of CHO cell-based bioproduction processes, enabling the efficient manufacturing of biologics for diverse therapeutic applications. With their defined composition, high performance, and regulatory compliance, CD CHO media formulations offer biopharmaceutical developers and manufacturers a reliable and cost-effective solution for optimizing cell culture processes and maximizing productivity, yield, and product quality. By leveraging innovation, collaboration, and market insights, stakeholders can capitalize on the growing demand for biopharmaceuticals and biologics, driving value creation and positive impact in the global healthcare ecosystem.
What is CD CHO Media?
CD CHO Media refers to a segment of the media landscape that focuses on the production and distribution of content related to CD CHO, which includes various forms of media such as digital content, advertising, and promotional materials.
What are the key players in the CD CHO Media Market?
Key players in the CD CHO Media Market include companies like CD CHO Media Group, Creative Media Solutions, and Digital Content Innovations, among others.
What are the main drivers of growth in the CD CHO Media Market?
The growth of the CD CHO Media Market is driven by increasing digital content consumption, the rise of social media platforms, and the demand for targeted advertising solutions.
What challenges does the CD CHO Media Market face?
Challenges in the CD CHO Media Market include intense competition, rapid technological changes, and the need for continuous content innovation to meet consumer expectations.
What opportunities exist in the CD CHO Media Market?
Opportunities in the CD CHO Media Market include the expansion of mobile media consumption, the integration of augmented reality in advertising, and the potential for personalized content delivery.
What trends are shaping the CD CHO Media Market?
Trends in the CD CHO Media Market include the increasing use of data analytics for audience targeting, the growth of influencer marketing, and the shift towards subscription-based content models.
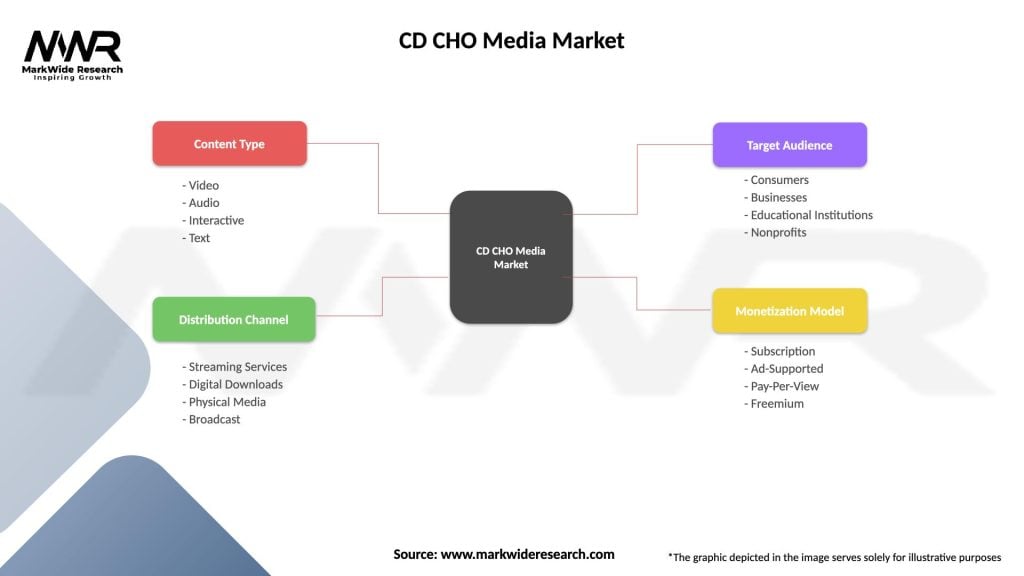
CD CHO Media Market
| Segmentation Details | Description |
|---|---|
| Content Type | Video, Audio, Interactive, Text |
| Distribution Channel | Streaming Services, Digital Downloads, Physical Media, Broadcast |
| Target Audience | Consumers, Businesses, Educational Institutions, Nonprofits |
| Monetization Model | Subscription, Ad-Supported, Pay-Per-View, Freemium |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in CD CHO Media Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at