444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$2750
Market Overview
The Asia Pacific region has witnessed significant advancements in pharmacogenetics testing, particularly in the field of psychiatry and depression. Pharmacogenetics testing involves analyzing an individual’s genetic makeup to determine how they may respond to specific medications. This personalized approach to treatment has gained popularity in recent years, as it offers a way to optimize medication selection and dosage, leading to improved patient outcomes.
Meaning
Pharmacogenetics testing in psychiatry and depression involves studying genetic variations that impact drug metabolism, response, and adverse reactions. By identifying these genetic markers, healthcare professionals can tailor medication choices and dosages to individual patients, enhancing the efficacy and safety of psychiatric treatments. This approach moves away from the traditional trial-and-error method, where patients may undergo several medication changes before finding the most suitable one.
Executive Summary
The Asia Pacific pharmacogenetics testing in psychiatry/depression market is experiencing rapid growth due to the increasing demand for personalized medicine and advancements in genetic testing technologies. This market offers a promising future as it addresses the challenges of psychiatric treatment and enhances patient care. The growing awareness among healthcare professionals and patients about the benefits of pharmacogenetics testing is driving market expansion.

Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
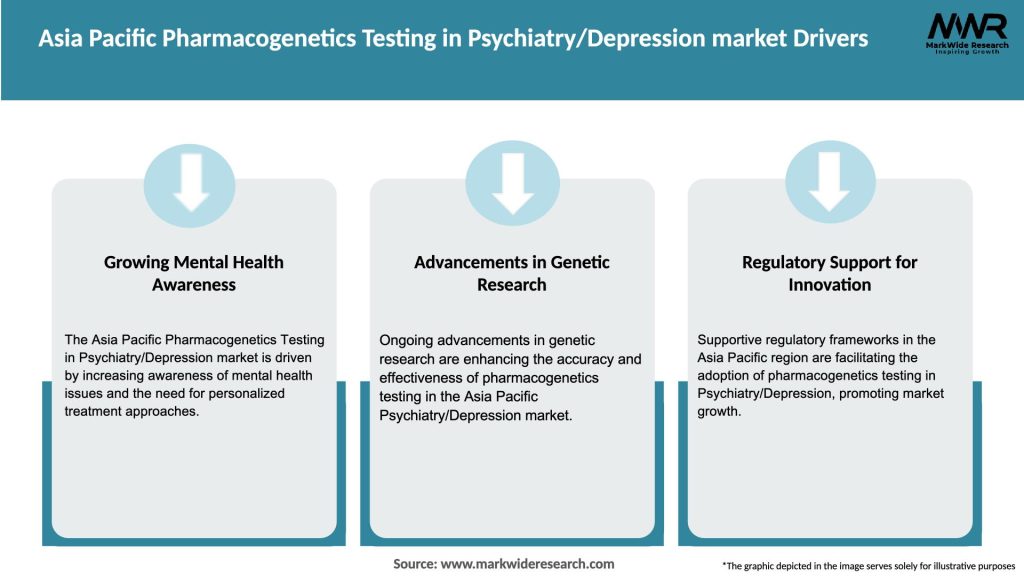
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The Asia Pacific pharmacogenetics testing in psychiatry/depression market is dynamic and influenced by various factors. The market is primarily driven by the growing demand for personalized medicine and advancements in genetic testing technologies. Rising awareness among healthcare professionals and patients, coupled with the increasing prevalence of psychiatric disorders, further fuels market expansion. However, limited awareness, cost and infrastructure challenges, and ethical considerations act as restraints. Collaborations, technological advancements, and research and development efforts present significant opportunities for market players.
Regional Analysis
The Asia Pacific region encompasses diverse countries with varying healthcare systems, regulatory frameworks, and levels of economic development. Japan, China, India, and Australia are key players in the pharmacogenetics testing market for psychiatry and depression in the region. Japan, in particular, has made significant strides in implementing pharmacogenetics testing in routine clinical practice. China and India are witnessing increasing adoption due to the rising prevalence of psychiatric disorders and improving healthcare infrastructure. Australia has a well-established genetic testing industry and offers a conducive environment for market growth.
Competitive Landscape
Leading Companies in the Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
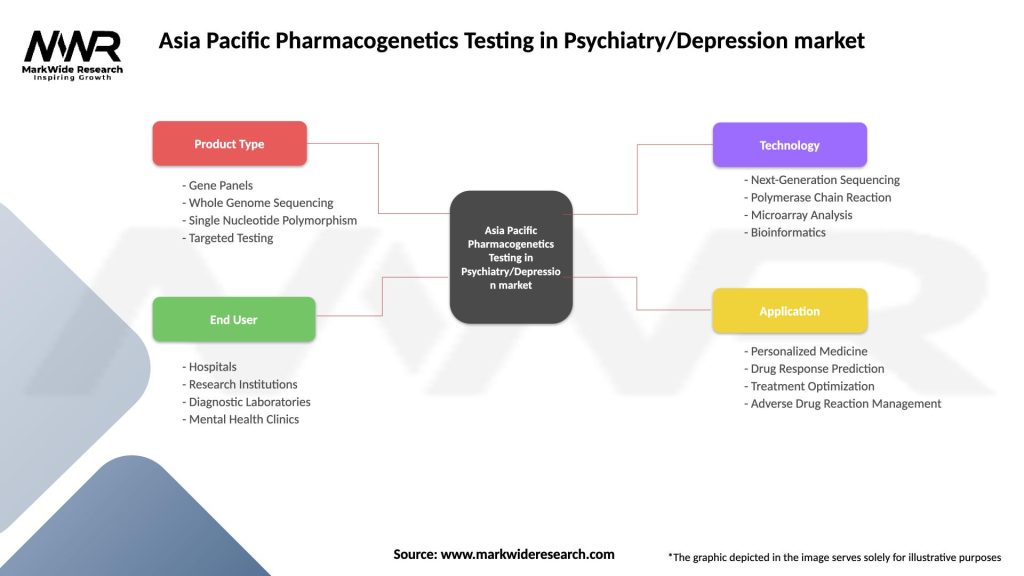
Segmentation
The pharmacogenetics testing market in Asia Pacific can be segmented based on test type, technology, end user, and geography. Test types may include drug metabolism testing, drug response testing, and adverse drug reaction testing. Technologies employed for genetic analysis include next-generation sequencing, polymerase chain reaction, and microarray. End users of pharmacogenetics testing services include hospitals, diagnostic laboratories, and academic research institutes.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Covid-19 Impact
The COVID-19 pandemic has had a profound impact on healthcare systems worldwide, including the pharmacogenetics testing market. While the pandemic initially disrupted healthcare services, including genetic testing, it also highlighted the need for personalized medicine and advanced diagnostic techniques. The crisis has emphasized the importance of optimizing treatment outcomes and minimizing adverse reactions, further driving the demand for pharmacogenetics testing in psychiatry and depression.
Key Industry Developments
Analyst Suggestions
Future Outlook
The Asia Pacific pharmacogenetics testing in psychiatry/depression market is poised for significant growth in the coming years. Factors such as the increasing prevalence of psychiatric disorders, rising awareness and adoption of pharmacogenetics testing, and advancements in genetic testing technologies will drive market expansion. Collaboration, technological advancements, and research and development activities will play a crucial role in shaping the future of the market. With supportive regulatory frameworks and growing industry initiatives, pharmacogenetics testing is expected to become an integral part of psychiatric care in the region.
Conclusion
Pharmacogenetics testing in psychiatry and depression is revolutionizing the field of personalized medicine in the Asia Pacific region. The market is driven by the growing demand for effective treatments, advancements in genetic testing technologies, and rising awareness among healthcare professionals and patients. Despite challenges such as limited awareness, cost considerations, and ethical concerns, the market presents significant opportunities for collaborations, technological advancements, and research and development activities. With regulatory support and increasing industry initiatives, the future of pharmacogenetics testing in psychiatry and depression looks promising, ultimately leading to improved patient outcomes and enhanced quality of care.
What is Pharmacogenetics Testing in Psychiatry/Depression?
Pharmacogenetics Testing in Psychiatry/Depression refers to the study of how an individual’s genetic makeup affects their response to psychiatric medications, particularly in the treatment of depression. This testing aims to optimize medication efficacy and minimize adverse effects by tailoring treatment to the patient’s genetic profile.
What are the key players in the Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression market?
Key players in the Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression market include companies like Myriad Genetics, GeneSight, and Invitae, which provide genetic testing services to help guide psychiatric treatment decisions, among others.
What are the growth factors driving the Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression market?
The growth of the Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression market is driven by increasing awareness of personalized medicine, advancements in genetic testing technologies, and a rising prevalence of mental health disorders, which necessitate more effective treatment options.
What challenges does the Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression market face?
Challenges in the Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression market include regulatory hurdles, the need for extensive clinical validation of tests, and potential ethical concerns regarding genetic data privacy and discrimination.
What opportunities exist in the Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression market?
Opportunities in the Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression market include the potential for expanding testing services to underserved populations, the integration of pharmacogenetic testing into routine psychiatric care, and collaborations between genetic testing companies and healthcare providers to enhance treatment outcomes.
What trends are shaping the Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression market?
Trends shaping the Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression market include the increasing adoption of digital health technologies, the rise of telemedicine for genetic counseling, and a growing emphasis on mental health awareness and personalized treatment approaches.
Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression market
| Segmentation Details | Description |
|---|---|
| Product Type | Gene Panels, Whole Genome Sequencing, Single Nucleotide Polymorphism, Targeted Testing |
| End User | Hospitals, Research Institutions, Diagnostic Laboratories, Mental Health Clinics |
| Technology | Next-Generation Sequencing, Polymerase Chain Reaction, Microarray Analysis, Bioinformatics |
| Application | Personalized Medicine, Drug Response Prediction, Treatment Optimization, Adverse Drug Reaction Management |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Asia Pacific Pharmacogenetics Testing in Psychiatry/Depression Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at