444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
In the world of pharmaceuticals, biotechnology, and food and beverage industries, the concept of aseptic sampling has emerged as a critical practice to ensure product quality, safety, and regulatory compliance. Aseptic sampling plays a pivotal role in maintaining the integrity of samples by preventing contamination during the collection process. This comprehensive guide dives into the Aseptic Sampling market, exploring its nuances, trends, challenges, and opportunities.
Meaning
Aseptic sampling refers to the meticulous process of collecting samples from a controlled environment without introducing contaminants. It’s a fundamental practice in industries where even the slightest contamination can lead to product recalls, compromised quality, and potential harm to consumers. The goal is to ensure that samples accurately represent the condition of the entire batch or product without altering its composition.
Executive Summary
The Aseptic Sampling market has witnessed significant growth over the years, driven by increasing quality standards, stringent regulations, and the need for reliable and representative samples. This report delves into key market insights, drivers, restraints, opportunities, and emerging trends, providing stakeholders with a holistic view of the market landscape.

Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Adoption of single-use bioreactors and closed-system processing has fueled the need for compatible aseptic samplers and sterile connectors.
Regulatory guidelines (e.g., FDA’s PAT, EMA’s annexes on sterility) emphasize routine, representative sampling, boosting demand for automated, validated sampling solutions.
Integration of in-line sensors (e.g., Raman, FTIR, pH, turbidity) with aseptic sampling ports enables real-time quality monitoring and process control.
The market’s leading end-users—biopharmaceutical and injectable drug manufacturers—account for over 60% of global aseptic sampling equipment revenues.
Emerging applications in cell therapy and viral vector production require ultra-low-volume sampling, creating niche demand for micro-sampling technologies.
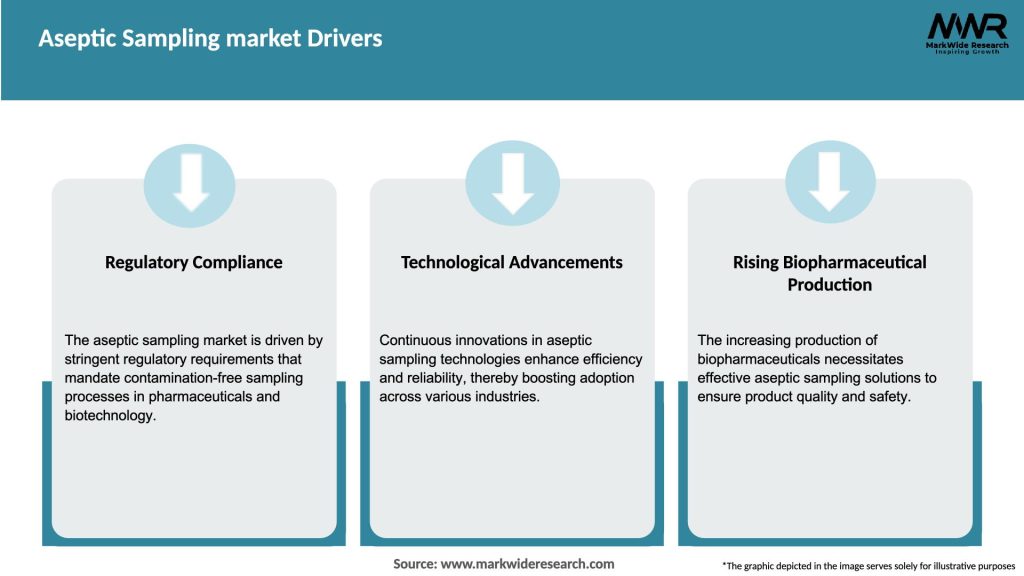
Market Drivers
Single-Use Technology Growth: The rise of disposable bioprocessing components necessitates aseptic sampling devices compatible with single-use assemblies.
Regulatory Pressure: Stricter sterility and validation requirements from global regulatory bodies compel manufacturers to adopt closed, validated sampling methods.
Continuous Manufacturing & PAT: Real-time analytics and continuous processing frameworks demand frequent, sterile sampling without process interruption.
Miniaturization & Small-Batch Production: Customized therapies and cell-based products drive the need for precise, low-volume aseptic sampling solutions.
Focus on Risk Mitigation: Reducing manual handling lowers the risk of contamination events, batch rejections, and costly recalls.
Market Restraints
High Validation Costs: Qualification and validation of aseptic sampling systems under GMP can be time-consuming and costly.
Complex Integration: Retrofitting existing stainless-steel facilities with single-use aseptic sampling may require significant process redesign.
Supply Chain Constraints: Dependence on specialized sterile components can lead to lead-time challenges and inventory complexities.
User Training Requirements: Effective use of closed-loop samplers demands staff training on sterile technique and connector handling.
Upfront Investment: Automated sampling platforms often involve higher initial capital expenditure than manual, open methods.
Market Opportunities
Disposable Micro-Sampling Kits: Development of aseptic micro-sampling devices for cell and gene therapy intermediates.
Integrated Sampling-Sensor Cartridges: Combined sampling and in-line analytical sensors for seamless process monitoring.
Modular Sampling Manifolds: Flexible manifold systems that adapt to various vessel sizes and tubing configurations.
Digital Validation Tools: Software-driven qualification and traceability platforms to streamline regulatory submissions.
Emerging Regions Expansion: Growth in Asia-Pacific and Latin America as bioprocessing capacity expands in India, China, and Brazil.

Market Dynamics
Innovation in Connector Design: A shift toward luer-lock-free, aseptic-disconnect connectors reduces contamination risk and simplifies handling.
Consolidation Among Suppliers: Partnerships between bioprocess-equipment OEMs and sterile component manufacturers drive bundled solution offerings.
Digital Transformation: Adoption of cloud-based monitoring and IoT connectivity for aseptic sampling systems enhances process oversight.
Customization Demand: Producers of niche biologics require tailored sampling assemblies, leading suppliers to offer configurable kits.
Emphasis on Sustainability: Development of recyclable or resource-efficient single-use samplers aligns with green manufacturing initiatives.
Regional Analysis
North America: Largest market share, fueled by established biopharma infrastructure and rapid adoption of single-use technologies in the U.S.
Europe: Strong growth driven by robust regulatory frameworks, established biotech clusters in Germany, the UK, and Switzerland.
Asia-Pacific: Fastest regional CAGR, underpinned by rising domestic biologics production in China, India, and South Korea.
Latin America: Emerging demand as local biopharma capacity grows; Brazil leads with increased government investment in vaccine manufacturing.
Middle East & Africa: Nascent market with opportunities in contract development & manufacturing organizations (CDMOs) and vaccine fill-finish operations.
Competitive Landscape
Leading Companies in the Aseptic Sampling Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Segmentation
By Technology: Manual Swab Sampling, Closed-Loop Aseptic Sampling, Automated Sampling Platforms
By Application: Bioreactor Sampling, Fill-Finish Sampling, Filtrate Sampling, In-Process Analytics
By End-User: Biopharmaceutical Manufacturers, CDMOs, Vaccine Producers, Food & Beverage Sterile Processors
By Region: North America, Europe, Asia-Pacific, Latin America, Middle East & Africa
Category-wise Insights
Manual Swab Sampling: Cost-effective, but higher risk of operator-dependent variability; suited for smaller labs and early-stage R&D.
Closed-Loop Aseptic Sampling: Provides sterility assurance and compatibility with single-use systems; widely adopted in GMP manufacturing.
Automated Sampling Platforms: Minimal manual intervention, high reproducibility, and integration with PAT frameworks; favored by large-scale producers.
Bioreactor Sampling: Requires specialized sterile ports and low-volume sampling probes to preserve cell culture integrity.
Fill-Finish Sampling: Emphasis on particulate testing and container closure integrity; often integrated with fill-finish lines for real-time quality checks.
Key Benefits for Industry Participants and Stakeholders
Enhanced Sterility Assurance: Closed-system sampling virtually eliminates contamination risk, reducing batch failures and re-work costs.
Regulatory Compliance: Validated sampling processes support submission dossiers and GMP audits, ensuring alignment with cGMP regulations.
Process Efficiency: Automated and single-use samplers shorten cycle times and minimize manual interventions, boosting throughput.
Data Integrity: Integration with digital validation and traceability systems ensures complete audit trails and sample provenance.
Flexibility & Scalability: Modular sampling solutions adapt to multiple vessel sizes, batch scales, and evolving process requirements.
SWOT Analysis
Strengths:
High sterility assurance and closed-loop operation
Alignment with single-use bioprocessing trends
Compatibility with advanced PAT and continuous manufacturing
Weaknesses:
High initial validation and qualification costs
Dependence on specialized sterile components
Operator training requirements for complex systems
Opportunities:
Rising adoption in cell & gene therapy and personalized medicine
Development of low-dead-volume micro-sampling devices
Expansion of cloud-based monitoring and AI-driven analytics
Threats:
Supply chain disruptions for single-use assemblies
Regulatory variations across regions complicating global rollouts
Competition from traditional open sampling in legacy facilities
Market Key Trends
Micro-Sampling Innovations: Ultra-low-volume sampling probes (<1 mL) for precious biologics and high-value therapeutics.
Connector Standardization: Industry-wide move toward luer-free, color-coded aseptic connectors to reduce errors and training complexity.
Digital Validation Platforms: Cloud-based tools for real-time qualification of sampling systems and complete audit record maintenance.
Hybrid Sampling Stations: Integrated benches combining swab, closed-loop, and sensor-based sampling in a single, ergonomic workstation.
Sustainability Initiatives: Development of recyclable or reduced-footprint single-use kits to align with corporate environmental goals.
Covid-19 Impact
The COVID-19 pandemic underscored the critical need for rapid, sterile sampling in vaccine and antiviral production. Scaling up global vaccine manufacture led to accelerated implementation of closed-loop aseptic samplers in fill-finish lines and upstream bioreactors. Supply chain pressures prompted manufacturers to qualify multiple sterile connector suppliers and invest in micro-sampling capabilities for viral vector and mRNA feedstock monitoring. Lessons learned during the pandemic continue to drive investments in resilient, flexible aseptic sampling platforms for agile response to future biologic emergencies.
Key Industry Developments
Sartorius launched a modular automated sampler with integrated sensor analytics for continuous bioprocess monitoring in 2024.
Pall Corporation introduced color-coded, luer-free sterile connectors for simplified, error-proof aseptic sampling operations.
Thermo Fisher expanded its FlexAct™ portfolio with disposable micro-sampling probes optimized for ATMP (Advanced Therapy Medicinal Products) applications.
Merck’s Mobius® team collaborated with a major CDMO to co-develop pre-qualified sampling manifolds for large-scale mRNA vaccine production.
Analyst Suggestions
Invest in Micro-Sampling R&D: Prioritize development of ultra-low-volume, dead-leg-free samplers for next-generation biotherapeutics.
Standardize Aseptic Connectors: Advocate for industry standards on connectors to reduce training overhead and contamination risk.
Leverage AI-Enhanced Validation: Deploy machine-learning platforms to predict maintenance needs and flag deviations in sampling performance.
Expand Cloud Connectivity: Integrate sampling systems with digital monitoring platforms to provide real-time sterility and process data to remote stakeholders.
Forge Regional Partnerships: Partner with local suppliers in high-growth regions to ensure supply chain resilience and reduce lead times.
Future Outlook
The Aseptic Sampling market is poised for continued expansion as bioprocessing evolves toward smaller batch sizes, personalized therapies, and continuous manufacturing. Innovations in micro-sampling, sensor integration, and digital validation will further enhance aseptic sample quality and process transparency. As regulatory bodies emphasize in-process controls and real-time release testing, closed-loop aseptic sampling solutions will become indispensable across all stages of sterile production, solidifying their role in the future of biologics manufacturing.
Conclusion
In conclusion, aseptic sampling represents a cornerstone of quality assurance in sterile manufacturing. By enabling contamination-free, representative sampling in both upstream and downstream processes, these systems help manufacturers meet rigorous regulatory standards, optimize operations, and accelerate product development. Stakeholders who invest in flexible, validated sampling platforms—and embrace digital and micro-sampling innovations—will lead the transformation toward more agile, efficient, and safe biopharmaceutical and sterile production ecosystems.
What is Aseptic Sampling?
Aseptic sampling refers to the process of collecting samples in a manner that prevents contamination. This technique is crucial in industries such as pharmaceuticals and food and beverage, where maintaining product integrity is essential.
What are the key players in the Aseptic Sampling market?
Key players in the Aseptic Sampling market include Merck KGaA, Sartorius AG, and Thermo Fisher Scientific, among others. These companies are known for their innovative solutions and technologies that enhance aseptic sampling processes.
What are the main drivers of the Aseptic Sampling market?
The main drivers of the Aseptic Sampling market include the increasing demand for biopharmaceuticals, the need for stringent quality control in manufacturing, and the growing focus on food safety. These factors are pushing industries to adopt aseptic sampling methods.
What challenges does the Aseptic Sampling market face?
Challenges in the Aseptic Sampling market include the high costs associated with advanced sampling technologies and the need for skilled personnel to perform aseptic techniques. Additionally, regulatory compliance can be complex and demanding.
What opportunities exist in the Aseptic Sampling market?
Opportunities in the Aseptic Sampling market include the development of new technologies that improve sampling efficiency and the expansion of applications in emerging markets. There is also potential for growth in the food and beverage sector as safety regulations tighten.
What trends are shaping the Aseptic Sampling market?
Trends in the Aseptic Sampling market include the increasing use of automated sampling systems and the integration of digital technologies for better data management. Additionally, there is a growing emphasis on sustainability and eco-friendly practices in sampling methods.
Aseptic Sampling Market:
| Segmentation Details | Information |
|---|---|
| Technique | Manual Aseptic Sampling, Automated Aseptic Sampling |
| Application | Food & Beverages, Pharmaceuticals, Others |
| Region | ASEAN countries (Indonesia, Malaysia, Thailand, Philippines, Vietnam, Singapore, Myanmar, Cambodia, Laos, Brunei) |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Aseptic Sampling Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at