444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$2750
Market Overview
The Middle East and Africa (MEA) region is witnessing a significant transformation in its healthcare landscape, with the In-Vitro Diagnostics (IVD) market playing a crucial role in this evolution. IVD refers to medical devices and technologies used to perform tests on samples such as blood, urine, and tissues in a controlled environment outside of a living organism. This market encompasses a wide array of diagnostic tools that aid in the accurate detection and monitoring of diseases, enabling healthcare professionals to make informed decisions about patient care.
Meaning
In-Vitro Diagnostics are pivotal tools that provide invaluable insights into a patient’s health condition without the need for invasive procedures. They encompass a diverse range of products, including reagents, instruments, and systems that aid in disease diagnosis and management. IVD has revolutionized modern medicine, enabling early disease detection, personalized treatment plans, and efficient healthcare delivery.
Executive Summary
The MEA IVD market is on a steady trajectory of growth, driven by factors such as increasing healthcare awareness, rising chronic diseases, and technological advancements. The region’s diverse population, coupled with a growing demand for quality healthcare services, presents a fertile ground for IVD market expansion. This executive summary delves into the key facets of the MEA IVD market, shedding light on its drivers, restraints, opportunities, and future prospects.
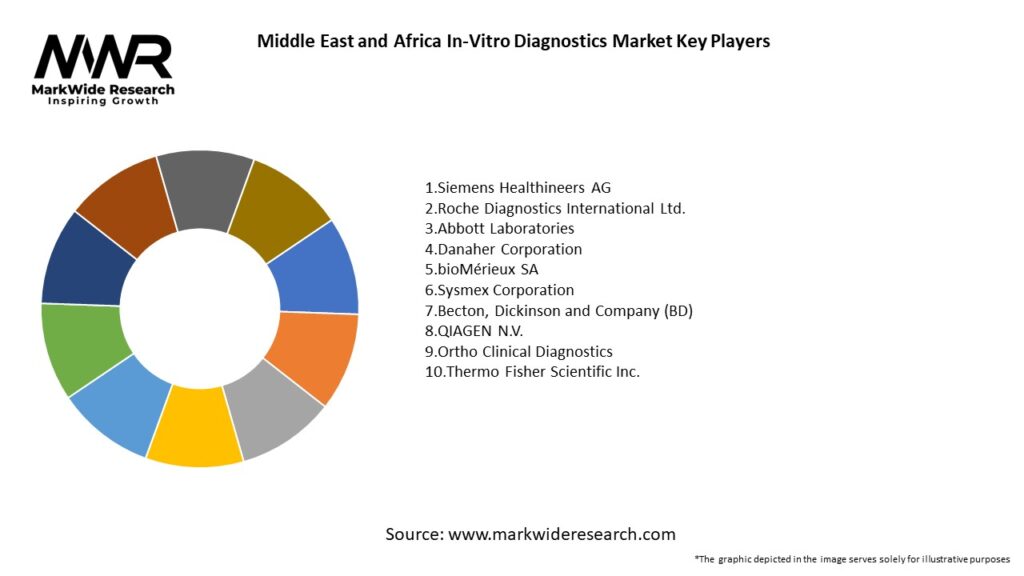
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
The Middle East and Africa In-Vitro Diagnostics Market is influenced by several key trends:
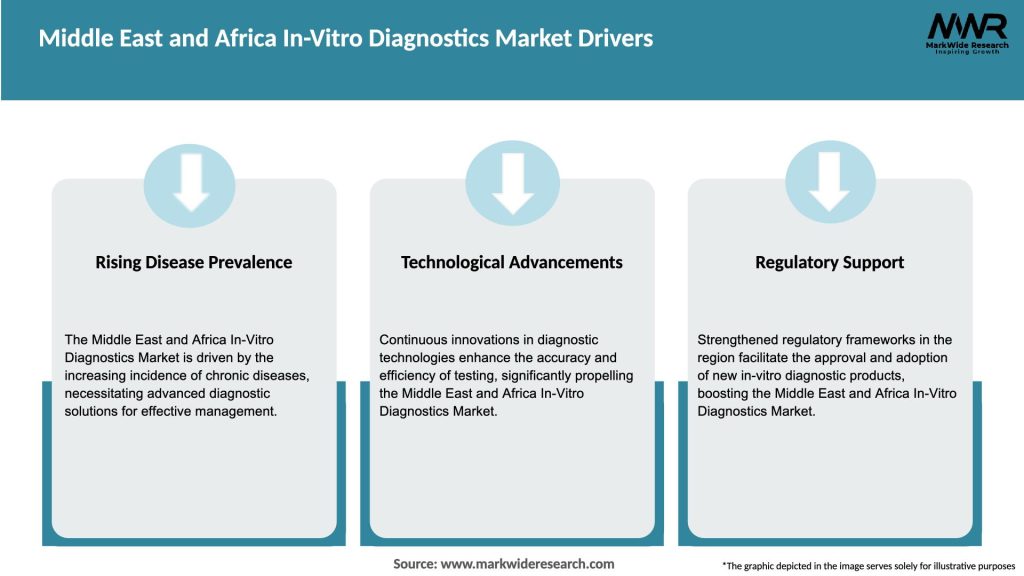
Market Drivers
The Middle East and Africa In-Vitro Diagnostics Market is being driven by several factors:
Market Restraints
Despite the growth potential, the Middle East and Africa In-Vitro Diagnostics Market faces several challenges:
Market Opportunities
The Middle East and Africa In-Vitro Diagnostics Market presents several opportunities for growth:
Market Dynamics
The dynamics of the Middle East and Africa In-Vitro Diagnostics Market are shaped by both supply-side and demand-side factors:
Regional Analysis
The Middle East and Africa In-Vitro Diagnostics Market exhibits diverse trends across different regions:
Competitive Landscape
Leading Companies in the Middle East and Africa In-Vitro Diagnostics Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
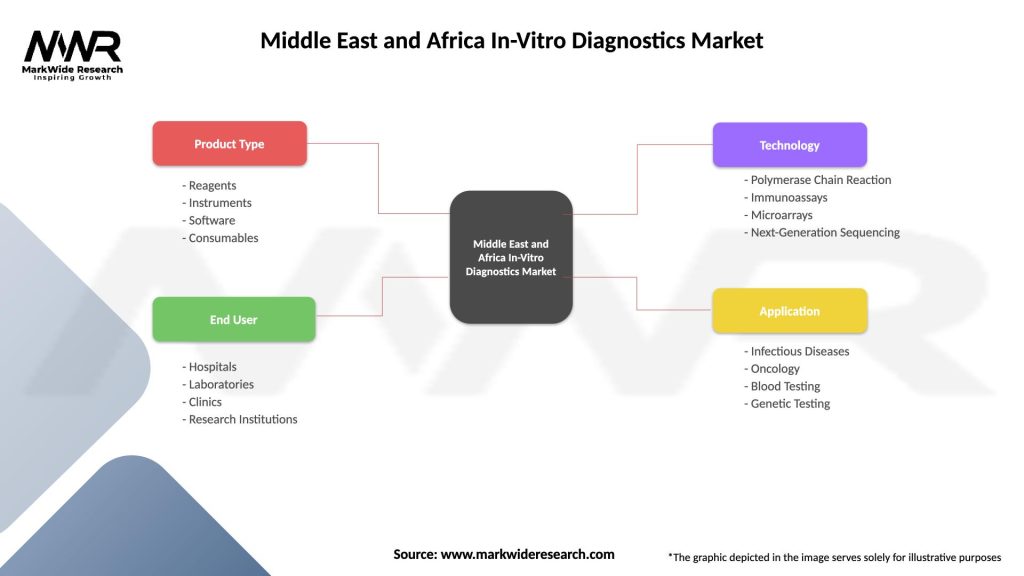
Segmentation
The Middle East and Africa In-Vitro Diagnostics Market can be segmented based on various factors to provide a detailed understanding of its structure:
Category-wise Insights
Each segment in the Middle East and Africa In-Vitro Diagnostics Market offers distinct advantages and growth opportunities:
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Covid-19 Impact
The Covid-19 pandemic has had a profound impact on the MEA IVD market. The urgent need for diagnostic tests during the pandemic accelerated the adoption of rapid testing and molecular diagnostics. The crisis highlighted the importance of robust diagnostic infrastructure and paved the way for future preparedness against infectious diseases.
Key Industry Developments
Recent industry developments underscore the market’s evolution. Collaborations between diagnostic companies and telehealth providers are enabling remote diagnostics. The launch of novel diagnostic platforms that offer faster results and higher accuracy is gaining traction. Furthermore, regulatory reforms are streamlining market access for innovative IVD products.
Analyst Suggestions
Analysts suggest that market players should prioritize innovation to cater to the region’s unique healthcare challenges. Collaborative partnerships with local healthcare providers can facilitate market entry. Additionally, investment in training and education about advanced diagnostic technologies will enhance adoption.
Future Outlook
The future of the MEA IVD market is promising, with a projected upward trajectory. The integration of AI, data analytics, and telehealth solutions will drive market growth. As healthcare access improves, the demand for accurate and rapid diagnostics will escalate, leading to increased market demand. Looking ahead, the future of the MEA IVD market holds promise. Continued investments in research and development, capacity building, and education will drive innovation and address existing challenges. The integration of digital health solutions, data analytics, and artificial intelligence will further enhance diagnostic accuracy and patient outcomes.
Conclusion
The Middle East and Africa In-Vitro Diagnostics market is a vital component of the region’s healthcare transformation. With a focus on early disease detection, personalized treatment, and efficient healthcare delivery, the market holds immense potential for growth. By navigating the dynamics of the market, embracing technological advancements, and addressing regional healthcare needs, industry participants can shape a healthier future for the MEA region.
In conclusion, the Middle East and Africa (MEA) In-Vitro Diagnostics (IVD) market represents a dynamic and evolving sector that plays a pivotal role in shaping the region’s healthcare landscape. The journey through this market overview has revealed a multifaceted landscape characterized by innovation, challenges, and immense potential.
What is In-Vitro Diagnostics?
In-Vitro Diagnostics refers to medical devices and tests that analyze biological samples outside the body, such as blood or tissue, to diagnose diseases, monitor health, and guide treatment decisions.
What are the key players in the Middle East and Africa In-Vitro Diagnostics Market?
Key players in the Middle East and Africa In-Vitro Diagnostics Market include Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, and Bio-Rad Laboratories, among others.
What are the growth factors driving the Middle East and Africa In-Vitro Diagnostics Market?
The growth of the Middle East and Africa In-Vitro Diagnostics Market is driven by increasing prevalence of chronic diseases, rising demand for early diagnosis, and advancements in diagnostic technologies.
What challenges does the Middle East and Africa In-Vitro Diagnostics Market face?
Challenges in the Middle East and Africa In-Vitro Diagnostics Market include regulatory hurdles, high costs of advanced diagnostic equipment, and limited access to healthcare in certain regions.
What opportunities exist in the Middle East and Africa In-Vitro Diagnostics Market?
Opportunities in the Middle East and Africa In-Vitro Diagnostics Market include the expansion of healthcare infrastructure, increasing investments in research and development, and the growing trend of personalized medicine.
What trends are shaping the Middle East and Africa In-Vitro Diagnostics Market?
Trends shaping the Middle East and Africa In-Vitro Diagnostics Market include the rise of point-of-care testing, integration of artificial intelligence in diagnostics, and the shift towards home-based testing solutions.
Middle East and Africa In-Vitro Diagnostics Market
| Segmentation Details | Description |
|---|---|
| Product Type | Reagents, Instruments, Software, Consumables |
| End User | Hospitals, Laboratories, Clinics, Research Institutions |
| Technology | Polymerase Chain Reaction, Immunoassays, Microarrays, Next-Generation Sequencing |
| Application | Infectious Diseases, Oncology, Blood Testing, Genetic Testing |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Middle East and Africa In-Vitro Diagnostics Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at