444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
The CEA (Carcinoembryonic Antigen) testing market is witnessing significant growth due to the rising prevalence of cancer worldwide. CEA testing plays a crucial role in the early detection, diagnosis, and monitoring of various types of cancers, particularly colorectal cancer. This market analysis provides insights into the key factors driving the CEA testing market, along with an evaluation of the market’s current and future prospects.
CEA testing involves the measurement of carcinoembryonic antigen levels in the blood. Carcinoembryonic antigens are proteins that are produced during fetal development, but their levels decrease significantly after birth. Elevated levels of CEA in the blood can indicate the presence of certain cancers, especially colorectal, pancreatic, and lung cancers. CEA testing is a valuable diagnostic tool for oncologists, helping them assess disease progression and monitor treatment response.
Executive Summary
The executive summary of the CEA testing market analysis provides a concise overview of the market’s key findings, including market size, growth rate, and major trends. It highlights the significant market drivers, restraints, and opportunities that are shaping the growth of the CEA testing market. This section also summarizes the regional analysis, competitive landscape, and segmentation of the market.
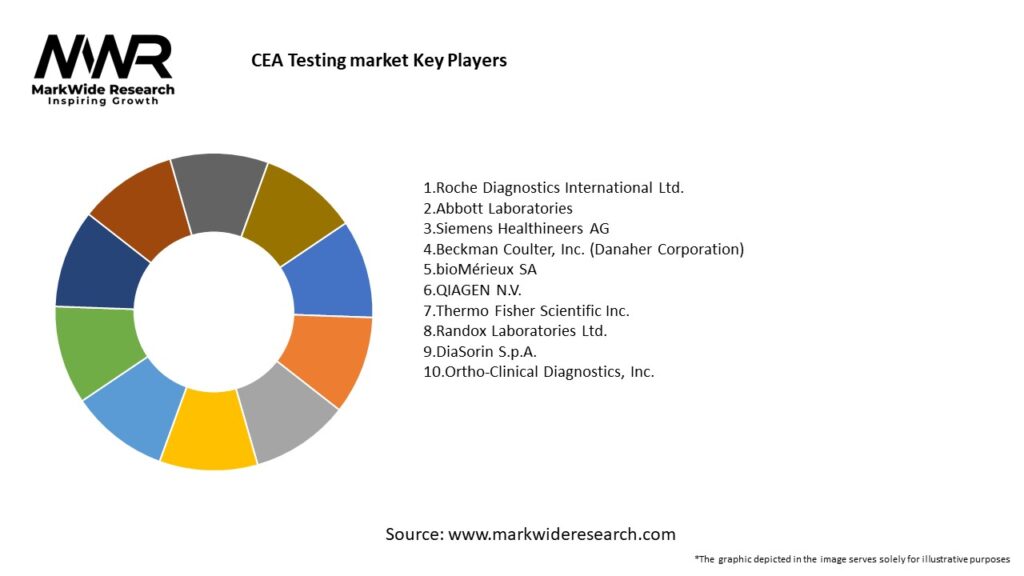
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
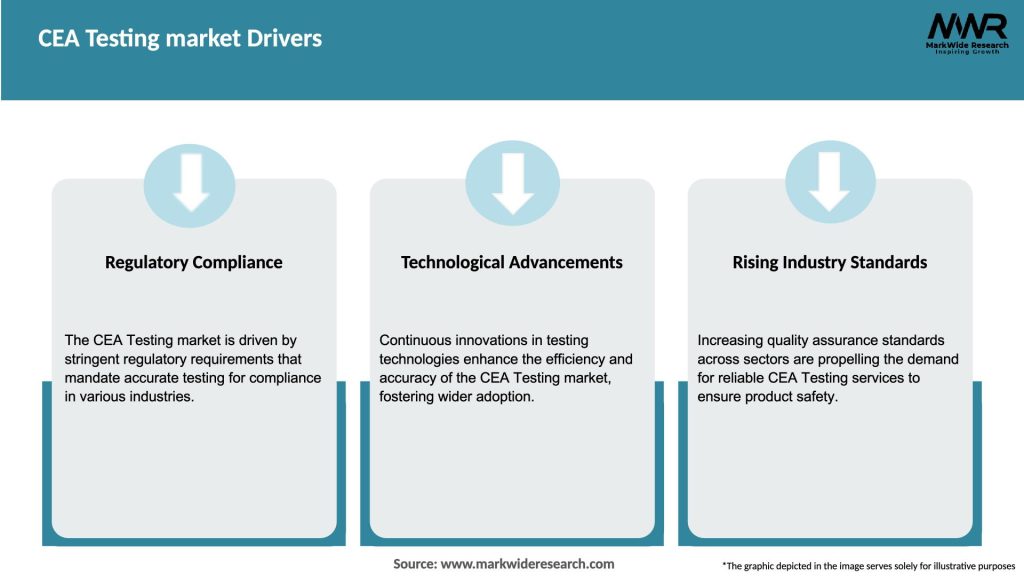
Market Drivers
The growth of the CEA Testing Market is being driven by several factors:
Market Restraints
Despite the positive growth, there are several factors restraining the CEA Testing Market:
Market Opportunities
The CEA Testing Market presents several opportunities for growth and development:
Market Dynamics
The CEA Testing Market is shaped by various dynamic trends and forces:
Regional Analysis
The CEA Testing Market shows diverse regional adoption rates, with varying growth trajectories:
Competitive Landscape
Leading Companies in the CEA Testing Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
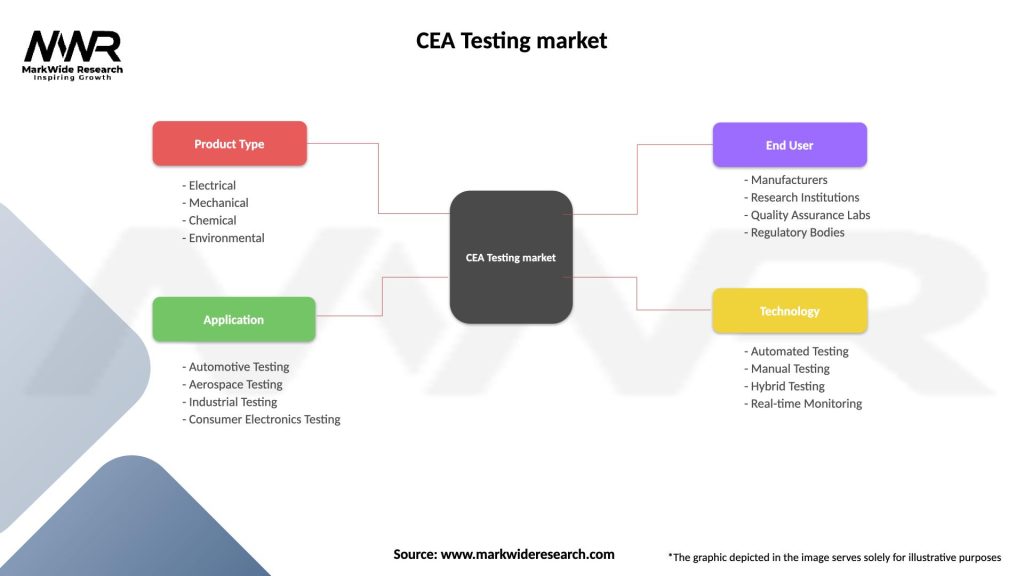
Segmentation
The CEA Testing Market can be segmented based on several factors:
Category-wise Insights
Each category of CEA testing solutions offers unique benefits:
Key Benefits for Industry Participants and Stakeholders
CEA testing provides several key benefits to industry participants and stakeholders:
SWOT Analysis
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Key trends influencing the CEA Testing Market include:
Covid-19 Impact
The Covid-19 pandemic has had a significant impact on the healthcare industry, including the CEA testing market. The section discusses the pandemic’s effects on market dynamics, supply chains, and customer behavior. It also explores the short-term and long-term implications of the pandemic on the CEA testing market and provides insights into the industry’s response and recovery strategies.
Key Industry Developments
This section highlights recent key developments in the CEA testing market, such as mergers and acquisitions, product launches, partnerships, and collaborations. These developments indicate the market’s growth trajectory, technological advancements, and strategic initiatives undertaken by key players.
Analyst Suggestions
Based on the comprehensive market analysis, the analyst suggestions section provides actionable recommendations for industry participants and stakeholders. These suggestions cover areas such as product development, market penetration, strategic partnerships, and investment opportunities. Implementing these suggestions can help stakeholders gain a competitive advantage and capitalize on market opportunities.
Future Outlook
The future outlook section provides insights into the CEA testing market’s expected growth trajectory over the forecast period. It analyzes market trends, technological advancements, regulatory changes, and evolving customer preferences to forecast the market’s potential. Understanding the future outlook helps stakeholders plan their strategies and make informed business decisions.
Conclusion
In conclusion, the CEA testing market is witnessing substantial growth due to the increasing prevalence of cancer and the demand for early detection and personalized treatment. Technological advancements and the aging population further contribute to market expansion. However, challenges related to specificity and regulatory requirements need to be addressed. The market presents significant opportunities in emerging economies and personalized medicine. By staying abreast of market dynamics, leveraging key trends, and investing in research and development, industry participants can capitalize on the growing demand for CEA testing and drive the market’s future growth.
What is CEA Testing?
CEA Testing refers to the assessment of carcinoembryonic antigen (CEA) levels in the blood, which is primarily used to monitor certain types of cancers, particularly colorectal cancer. This testing helps in evaluating treatment effectiveness and detecting recurrences.
What are the key players in the CEA Testing market?
Key players in the CEA Testing market include Abbott Laboratories, Roche Diagnostics, and Siemens Healthineers, among others. These companies are known for their innovative diagnostic solutions and extensive product portfolios in cancer testing.
What are the growth factors driving the CEA Testing market?
The growth of the CEA Testing market is driven by the increasing prevalence of cancer, advancements in diagnostic technologies, and a growing emphasis on early detection and personalized medicine. Additionally, rising healthcare expenditure contributes to market expansion.
What challenges does the CEA Testing market face?
The CEA Testing market faces challenges such as the variability in CEA levels among different individuals, which can lead to false positives or negatives. Furthermore, regulatory hurdles and the need for continuous innovation in testing methods pose significant challenges.
What opportunities exist in the CEA Testing market?
Opportunities in the CEA Testing market include the development of more sensitive and specific testing methods, integration of CEA testing with other biomarkers, and expansion into emerging markets where cancer incidence is rising. These factors can enhance diagnostic accuracy and patient outcomes.
What trends are shaping the CEA Testing market?
Trends in the CEA Testing market include the increasing adoption of liquid biopsy techniques, advancements in point-of-care testing, and the integration of artificial intelligence in diagnostic processes. These innovations aim to improve the efficiency and accuracy of cancer diagnostics.
CEA Testing market
| Segmentation Details | Description |
|---|---|
| Product Type | Electrical, Mechanical, Chemical, Environmental |
| Application | Automotive Testing, Aerospace Testing, Industrial Testing, Consumer Electronics Testing |
| End User | Manufacturers, Research Institutions, Quality Assurance Labs, Regulatory Bodies |
| Technology | Automated Testing, Manual Testing, Hybrid Testing, Real-time Monitoring |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the CEA Testing Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at