444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$2750
Market Overview
The North America and Europe PEGylated Drugs Market is a thriving sector within the pharmaceutical industry. PEGylation refers to the process of attaching polyethylene glycol (PEG) chains to drugs or therapeutic proteins. This technique has gained significant attention due to its ability to enhance the pharmacokinetic properties and therapeutic efficacy of drugs. PEGylation helps to increase the half-life of drugs, improve their solubility, reduce immunogenicity, and enhance their stability.
Meaning
PEGylation is a technique that involves the attachment of PEG chains to drugs or therapeutic proteins. This process can modify the properties of drugs and improve their performance in terms of pharmacokinetics, efficacy, and safety.
Executive Summary
The North America and Europe PEGylated Drugs Market is experiencing substantial growth due to the increasing demand for innovative and effective drug delivery systems. The market is driven by factors such as the rising prevalence of chronic diseases, advancements in biotechnology, and the need for targeted drug delivery. Additionally, the market is benefiting from the increasing investment in research and development activities by pharmaceutical companies.
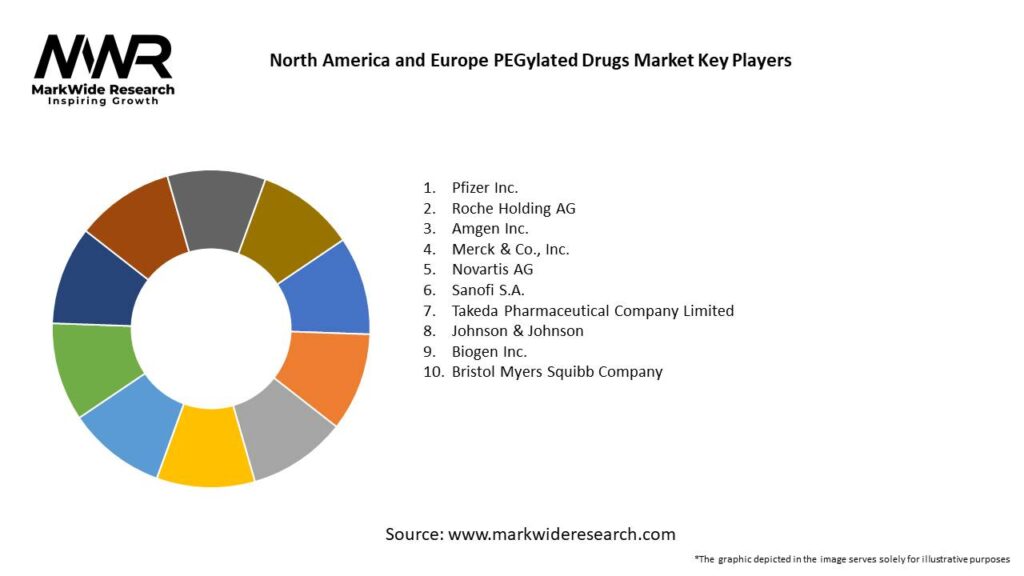
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
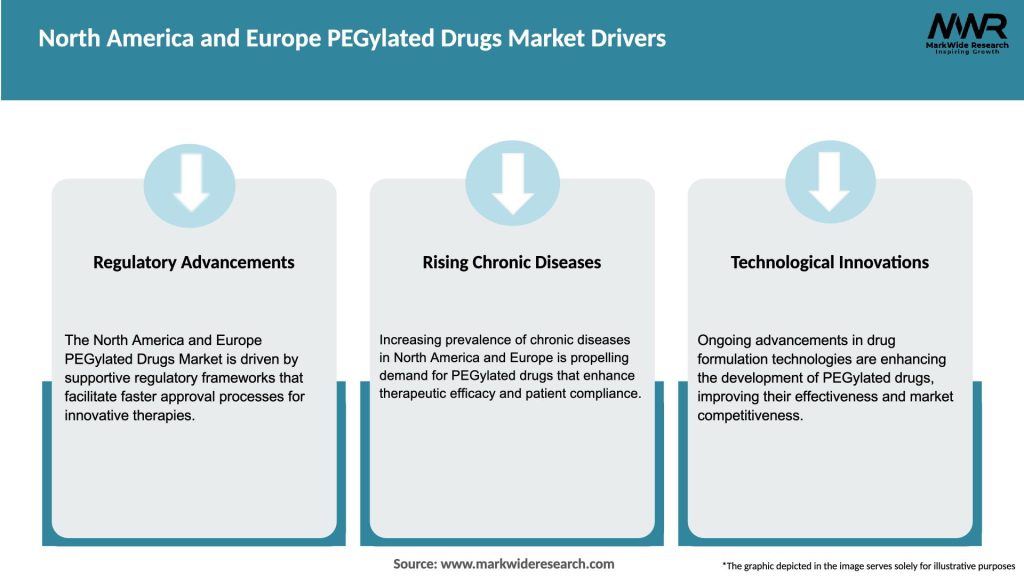
Market Drivers
The North America and Europe PEGylated Drugs Market is propelled by various drivers:
Market Restraints
Despite the positive market outlook, the North America and Europe PEGylated Drugs Market also faces certain challenges:
Market Opportunities
The North America and Europe PEGylated Drugs Market presents several opportunities for growth:
Market Dynamics
The North America and Europe PEGylated Drugs Market is characterized by dynamic factors that influence its growth:
Regional Analysis
The North America and Europe PEGylated Drugs Market is segmented into two major regions:
Competitive Landscape
Leading Companies in North America and Europe PEGylated Drugs Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
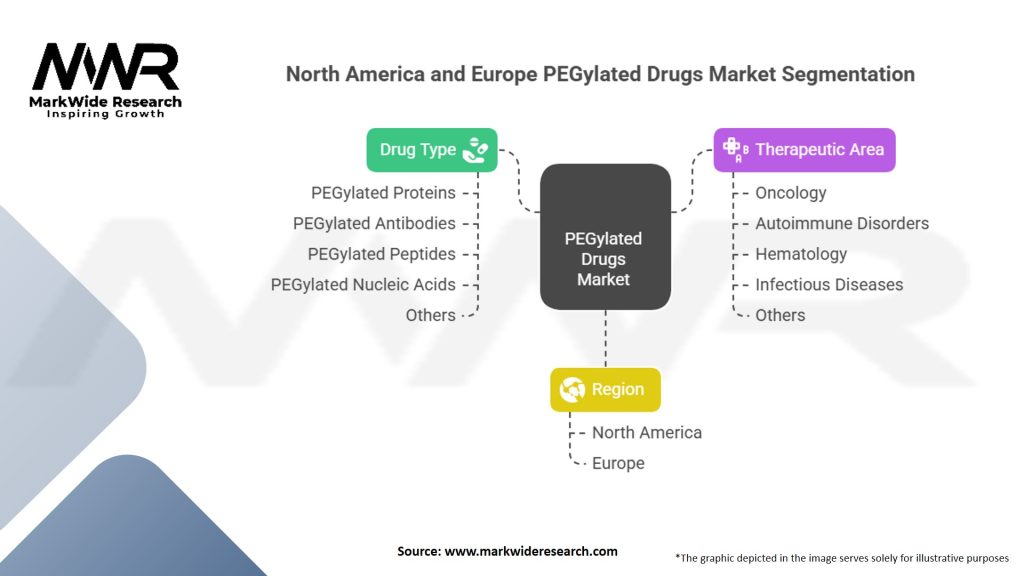
Segmentation
The North America and Europe PEGylated Drugs Market can be segmented based on:
These segmentation criteria enable a more targeted analysis of the market and help stakeholders identify specific growth opportunities.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
The North America and Europe PEGylated Drugs Market offers several benefits for industry participants and stakeholders:
SWOT Analysis
A SWOT analysis provides an overview of the strengths, weaknesses, opportunities, and threats in the North America and Europe PEGylated Drugs Market:
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Several key trends are shaping the North America and Europe PEGylated Drugs Market:
Covid-19 Impact
The Covid-19 pandemic has had a significant impact on the pharmaceutical industry, including the North America and Europe PEGylated Drugs Market. The pandemic has highlighted the importance of drug development and innovation in addressing global health challenges. Some specific impacts include:
Key Industry Developments
The North America and Europe PEGylated Drugs Market has witnessed several key industry developments:
Analyst Suggestions
Based on the analysis of the North America and Europe PEGylated Drugs Market, analysts make the following suggestions:
Future Outlook
The North America and Europe PEGylated Drugs Market is poised for significant growth in the coming years. Factors such as the increasing prevalence of chronic diseases, advancements in biotechnology, and the need for targeted drug delivery are expected to drive market expansion.
The market will witness ongoing research and development activities, leading to the development of novel PEGylated drugs with improved therapeutic efficacy and safety profiles. The adoption of PEGylation technology in various therapeutic areas will continue to grow, presenting opportunities for industry participants.
However, challenges such as high development costs, regulatory hurdles, and limited market awareness need to be addressed for sustained market growth. Strategic collaborations, increased market education, and regulatory support will play crucial roles in overcoming these challenges and unlocking the full potential of the North America and Europe PEGylated Drugs Market.
Conclusion
The North America and Europe PEGylated Drugs Market is a rapidly growing sector within the pharmaceutical industry. PEGylation technology offers significant advantages, including improved drug stability, enhanced therapeutic efficacy, and reduced immunogenicity.
The market is driven by factors such as the growing prevalence of chronic diseases, advancements in biotechnology, and the need for targeted drug delivery. Industry participants and stakeholders can benefit from revenue generation, enhanced therapeutic efficacy, market expansion, and competitive advantage in the PEGylated Drugs Market.
Despite challenges such as high development costs and regulatory hurdles, the market presents opportunities for growth through expanding therapeutic applications, collaborations, and partnerships, as well as emerging markets.
What is PEGylated Drugs?
PEGylated drugs are pharmaceutical compounds that have been modified by the attachment of polyethylene glycol (PEG) chains. This modification enhances the solubility, stability, and bioavailability of the drugs, making them more effective in treating various medical conditions.
What are the key players in the North America and Europe PEGylated Drugs Market?
Key players in the North America and Europe PEGylated Drugs Market include Amgen, Merck & Co., and Pfizer, among others. These companies are involved in the development and commercialization of PEGylated therapeutics for various diseases.
What are the growth factors driving the North America and Europe PEGylated Drugs Market?
The growth of the North America and Europe PEGylated Drugs Market is driven by factors such as the increasing prevalence of chronic diseases, advancements in drug delivery systems, and the rising demand for targeted therapies.
What challenges does the North America and Europe PEGylated Drugs Market face?
The North America and Europe PEGylated Drugs Market faces challenges such as high production costs, regulatory hurdles, and potential immunogenicity issues associated with PEGylated products.
What opportunities exist in the North America and Europe PEGylated Drugs Market?
Opportunities in the North America and Europe PEGylated Drugs Market include the development of novel PEGylated formulations and the expansion of applications in oncology and rare diseases, which can enhance treatment options for patients.
What trends are shaping the North America and Europe PEGylated Drugs Market?
Trends shaping the North America and Europe PEGylated Drugs Market include the increasing focus on personalized medicine, the integration of nanotechnology in drug formulation, and the growing interest in biosimilars and biobetters.
North America and Europe PEGylated Drugs Market
| Segmentation Details | Description |
|---|---|
| Drug Type | PEGylated Proteins, PEGylated Antibodies, PEGylated Peptides, PEGylated Nucleic Acids, Others |
| Therapeutic Area | Oncology, Autoimmune Disorders, Hematology, Infectious Diseases, Others |
| Region | North America, Europe |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in North America and Europe PEGylated Drugs Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at