444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at

Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$2750
The MEA insulin syringe market represents a critical component of diabetes management infrastructure across the Middle East and Africa region. This specialized medical device market encompasses disposable and reusable insulin delivery systems designed specifically for subcutaneous insulin administration. Market dynamics in the MEA region are characterized by increasing diabetes prevalence, growing healthcare awareness, and expanding access to diabetes care solutions.
Regional healthcare systems across the Middle East and Africa are experiencing significant transformation, with governments investing heavily in diabetes prevention and management programs. The insulin syringe market benefits from this healthcare infrastructure development, particularly in countries like Saudi Arabia, UAE, South Africa, and Egypt. Growth trajectories indicate robust expansion driven by demographic shifts, lifestyle changes, and improved diagnostic capabilities.
Market penetration varies considerably across different MEA countries, with Gulf Cooperation Council nations demonstrating higher adoption rates of advanced insulin delivery systems compared to sub-Saharan African markets. The region’s unique healthcare challenges, including limited cold chain infrastructure and varying regulatory frameworks, create both opportunities and constraints for market development. Healthcare accessibility improvements and diabetes education initiatives are driving increased demand for reliable insulin delivery solutions.
The MEA insulin syringe market refers to the comprehensive ecosystem of insulin delivery devices, distribution networks, and supporting services operating across Middle Eastern and African healthcare systems. This market encompasses various syringe types, including disposable insulin syringes, safety syringes, and specialized delivery systems designed for different insulin formulations and patient needs.
Market scope includes manufacturing, importation, distribution, and retail of insulin syringes through hospitals, clinics, pharmacies, and diabetes care centers. The definition extends beyond physical devices to include associated services such as patient education, healthcare provider training, and diabetes management support programs. Regulatory compliance requirements vary across MEA countries, influencing market structure and competitive dynamics.
Strategic analysis reveals the MEA insulin syringe market as a rapidly evolving healthcare segment driven by increasing diabetes prevalence and improving healthcare infrastructure. The market demonstrates strong growth potential, with diabetes incidence rates rising approximately 12% annually across key MEA countries. This growth creates substantial opportunities for insulin syringe manufacturers and healthcare providers.
Key market characteristics include fragmented distribution networks, varying regulatory environments, and significant price sensitivity among end users. The market benefits from government healthcare initiatives, international development programs, and growing private healthcare investment. Competitive landscape features both international medical device companies and regional distributors, creating diverse market dynamics.
Future prospects indicate continued market expansion supported by demographic trends, urbanization, and healthcare system modernization. The integration of digital health solutions and improved diabetes care protocols presents additional growth opportunities for innovative insulin delivery systems.
Market intelligence reveals several critical insights shaping the MEA insulin syringe landscape:
Primary growth drivers propelling the MEA insulin syringe market include escalating diabetes prevalence across the region. Lifestyle changes associated with urbanization, dietary shifts, and reduced physical activity are contributing to increased Type 2 diabetes incidence. The growing elderly population in several MEA countries further amplifies demand for diabetes management solutions.
Healthcare system improvements represent another significant driver, with governments investing in diabetes care infrastructure and prevention programs. The establishment of specialized diabetes centers, improved healthcare provider training, and enhanced patient education initiatives create favorable market conditions. International health organizations and development programs are also supporting diabetes care improvements across the region.
Economic development in key MEA countries is improving healthcare accessibility and affordability. Rising disposable incomes, expanding health insurance coverage, and government healthcare subsidies are making insulin syringes more accessible to broader patient populations. Regulatory improvements and streamlined import processes are facilitating market entry for international manufacturers.
Technological advancement in insulin syringe design is driving market growth through improved safety features, ease of use, and patient comfort. The development of safety-engineered syringes addresses healthcare worker protection concerns while reducing needlestick injuries. Innovation in syringe materials and manufacturing processes is also improving product quality and reducing costs.
Significant challenges constraining MEA insulin syringe market growth include limited healthcare infrastructure in rural and remote areas. Cold chain limitations affect insulin storage and distribution, particularly in regions with unreliable electricity supply and extreme temperatures. These infrastructure constraints limit market penetration and increase distribution costs.
Economic barriers present substantial challenges, with high poverty rates and limited healthcare spending capacity affecting market accessibility. The lack of comprehensive health insurance coverage in many MEA countries creates affordability constraints for diabetes patients. Currency fluctuations and import dependency also impact product pricing and availability.
Regulatory complexities across different MEA countries create market entry barriers and compliance challenges. Varying registration requirements, quality standards, and import procedures increase operational costs for manufacturers and distributors. Bureaucratic processes can delay product launches and market expansion initiatives.
Cultural and educational factors sometimes impede market growth, including limited diabetes awareness, traditional medicine preferences, and resistance to self-injection. Healthcare provider shortages in many regions limit patient education and proper diabetes management support, affecting insulin syringe utilization rates.
Substantial opportunities exist for MEA insulin syringe market expansion through government healthcare initiatives and international development programs. Public-private partnerships are creating new avenues for market development, particularly in diabetes prevention and management programs. These collaborations offer opportunities for sustainable market growth and improved patient outcomes.
Digital health integration presents significant opportunities for innovative insulin delivery solutions. The development of smart syringes with dose tracking capabilities, mobile health applications, and telemedicine integration can differentiate products and improve patient compliance. Technology adoption in healthcare is accelerating across the MEA region, creating favorable conditions for advanced solutions.
Market expansion opportunities exist in underserved regions and patient segments. The growing middle class in several MEA countries represents an expanding market for premium insulin delivery systems. Rural healthcare initiatives and mobile health programs create opportunities for innovative distribution models and product adaptations.
Manufacturing localization opportunities are emerging as governments promote domestic medical device production. Local manufacturing can reduce costs, improve supply chain reliability, and create employment opportunities while meeting growing market demand. Regional manufacturing hubs could serve multiple MEA markets efficiently.
Complex market dynamics characterize the MEA insulin syringe landscape, with supply and demand factors varying significantly across different countries and regions. Demand patterns are influenced by diabetes prevalence rates, healthcare accessibility, economic conditions, and cultural factors. Supply dynamics depend on manufacturing capacity, distribution networks, regulatory approvals, and import capabilities.
Competitive dynamics involve both international medical device manufacturers and regional distributors, creating diverse market structures. Price competition is intense, particularly in price-sensitive markets, while quality and safety features differentiate premium products. Market consolidation trends are emerging as larger companies acquire regional distributors and smaller manufacturers.
Regulatory dynamics are evolving as MEA countries work to harmonize medical device standards and improve market access procedures. Quality requirements are becoming more stringent, favoring established manufacturers with robust quality systems. Regulatory changes can significantly impact market dynamics and competitive positioning.
Technology dynamics are driving innovation in insulin syringe design and functionality. Safety features, ease of use improvements, and digital integration capabilities are becoming increasingly important market differentiators. The pace of technological advancement varies across different market segments and price categories.
Comprehensive research methodology employed for MEA insulin syringe market analysis incorporates multiple data collection and validation techniques. Primary research includes extensive interviews with healthcare providers, diabetes specialists, hospital administrators, and pharmacy managers across key MEA countries. Patient surveys and focus groups provide insights into usage patterns, preferences, and unmet needs.
Secondary research encompasses analysis of government health statistics, medical device registration databases, import/export data, and healthcare industry reports. Market intelligence is gathered from medical conferences, trade publications, and industry associations operating in the MEA region. Regulatory filing analysis provides insights into market entry trends and competitive activities.
Data validation processes include cross-referencing multiple sources, statistical analysis, and expert review panels. Market modeling incorporates demographic trends, healthcare spending patterns, and diabetes prevalence projections to ensure accuracy and reliability. Regional variations are carefully analyzed to provide country-specific insights and recommendations.
Analytical frameworks include Porter’s Five Forces analysis, SWOT assessment, and competitive benchmarking studies. Quantitative analysis focuses on market sizing, growth rate calculations, and trend identification, while qualitative analysis explores market dynamics, challenges, and opportunities in depth.
Gulf Cooperation Council countries represent the most developed segment of the MEA insulin syringe market, with Saudi Arabia and UAE leading in terms of market sophistication and adoption rates. These markets benefit from advanced healthcare infrastructure, higher disposable incomes, and comprehensive health insurance systems. Market penetration of premium insulin delivery systems is highest in this sub-region, with approximately 65% market share for safety-engineered syringes.
North African markets including Egypt, Morocco, and Algeria demonstrate significant growth potential driven by large populations and improving healthcare systems. Egypt represents the largest market by volume in this sub-region, with government healthcare initiatives supporting diabetes care expansion. Market dynamics are characterized by price sensitivity and growing demand for affordable insulin delivery solutions.
Sub-Saharan Africa presents both challenges and opportunities, with South Africa leading market development due to its advanced healthcare system and regulatory framework. Nigeria and Kenya show promising growth trajectories supported by healthcare infrastructure improvements and international development programs. Market penetration remains limited by economic constraints and healthcare accessibility challenges.
Regional market distribution indicates that GCC countries account for approximately 45% of total MEA insulin syringe demand, despite representing a smaller population base. North Africa contributes roughly 35% of market volume, while Sub-Saharan Africa represents about 20% of current market size but demonstrates the highest growth potential.
Market leadership in the MEA insulin syringe sector is characterized by a mix of international medical device manufacturers and regional distribution partners. The competitive landscape reflects varying market maturity levels across different MEA countries and price segments.
Competitive strategies include product differentiation through safety features, cost optimization for price-sensitive markets, and partnership development with healthcare providers and government agencies. Market consolidation trends are emerging as larger companies acquire regional distributors to strengthen market presence.
Product segmentation of the MEA insulin syringe market encompasses multiple categories based on design, functionality, and target applications:
By Product Type:
By Needle Length:
By End User:
Disposable insulin syringes dominate the MEA market due to safety considerations, convenience, and regulatory preferences. This category benefits from healthcare provider recommendations and patient safety protocols. Growth drivers include increasing diabetes prevalence and healthcare system modernization efforts across the region.
Safety-engineered syringes represent the fastest-growing category, driven by healthcare worker protection initiatives and hospital safety protocols. Adoption rates are highest in GCC countries and urban healthcare facilities, with annual growth exceeding 15% in key markets. Cost considerations remain a barrier in price-sensitive segments.
Prefilled insulin syringes show promising growth potential in premium market segments, particularly among affluent patients and private healthcare facilities. Market barriers include higher costs, cold chain requirements, and limited product availability. This category benefits from convenience advantages and improved dosing accuracy.
Hospital and clinic segments drive bulk purchasing patterns and influence product specifications. Procurement processes often emphasize cost-effectiveness, quality certifications, and supplier reliability. This segment benefits from government healthcare spending and international development programs supporting diabetes care infrastructure.
Home care applications represent the highest growth potential, driven by patient preference for self-administration and healthcare system efforts to reduce hospitalization costs. Patient education and support services are critical success factors in this segment.
Healthcare providers benefit from improved patient outcomes through reliable insulin delivery systems and enhanced safety features. Clinical advantages include reduced needlestick injuries, improved dosing accuracy, and better patient compliance with diabetes management protocols. Cost-effective solutions help healthcare facilities manage budget constraints while maintaining quality care standards.
Patients gain access to safer, more convenient insulin delivery options that improve quality of life and treatment adherence. Safety benefits include reduced infection risks, improved injection comfort, and better dose control. Educational support and product innovations help patients achieve better diabetes management outcomes.
Manufacturers and distributors benefit from growing market demand and opportunities for product differentiation. Market expansion potential exists through geographic diversification, product innovation, and partnership development. Regulatory harmonization efforts are reducing market entry barriers and operational complexities.
Government healthcare systems benefit from improved diabetes care outcomes and reduced long-term healthcare costs. Public health advantages include better disease management, reduced complications, and improved population health indicators. Economic benefits include job creation through local manufacturing and distribution activities.
Healthcare investors find attractive opportunities in the growing diabetes care market, with stable demand patterns and government support for healthcare infrastructure development. The market offers diversification opportunities across different countries, price segments, and product categories.
Strengths:
Weaknesses:
Opportunities:
Threats:
Safety enhancement trends are driving adoption of engineered safety features in insulin syringes across the MEA region. Healthcare facilities are increasingly prioritizing needlestick injury prevention, leading to growing demand for safety-engineered devices. This trend is particularly pronounced in hospital settings and urban healthcare facilities with advanced safety protocols.
Digital health integration is emerging as a significant trend, with development of smart syringes featuring dose tracking and mobile app connectivity. Technology adoption is accelerating among tech-savvy patients and progressive healthcare providers. This trend creates opportunities for product differentiation and improved patient outcomes through better diabetes management.
Sustainability initiatives are influencing product development and packaging design, with growing emphasis on environmentally friendly materials and waste reduction. Eco-conscious healthcare providers and patients are driving demand for sustainable insulin delivery solutions. This trend aligns with global environmental goals and corporate responsibility initiatives.
Personalization trends are leading to development of patient-specific insulin syringe solutions, including different needle lengths, syringe sizes, and comfort features. Patient-centered care approaches are driving demand for customized diabetes management tools. This trend supports better treatment adherence and improved patient satisfaction.
Telemedicine integration is creating opportunities for remote diabetes management and insulin syringe usage monitoring. Digital health platforms are incorporating insulin delivery tracking and patient education components. This trend is particularly relevant in regions with limited healthcare provider access.
Regulatory harmonization initiatives across MEA countries are streamlining medical device approval processes and reducing market entry barriers. Regional cooperation efforts are creating more efficient regulatory pathways for insulin syringe manufacturers and distributors. These developments support market expansion and improve product availability.
Manufacturing capacity expansion is occurring through both international company investments and local production facility development. Supply chain localization efforts are improving product availability and reducing dependency on imports. Several MEA countries are developing medical device manufacturing capabilities to serve regional markets.
Partnership agreements between international manufacturers and regional distributors are expanding market reach and improving customer service capabilities. Strategic alliances are enabling better market penetration and local market knowledge utilization. These partnerships often include technology transfer and capacity building components.
Product innovation launches are introducing advanced safety features, improved ergonomics, and digital integration capabilities to MEA markets. Technology advancement is making insulin syringes safer, more user-friendly, and more effective for diabetes management. Innovation focus areas include needle technology, safety mechanisms, and dose accuracy improvements.
Healthcare infrastructure investments by governments and international organizations are improving diabetes care capabilities and creating new market opportunities. Facility development includes specialized diabetes centers, upgraded hospital equipment, and expanded pharmacy networks. These investments directly support insulin syringe market growth.
Market entry strategies should prioritize partnership development with established regional distributors and healthcare providers. MarkWide Research analysis indicates that successful market penetration requires deep understanding of local healthcare systems, regulatory requirements, and cultural factors. Companies should invest in relationship building and local market expertise development.
Product portfolio optimization should balance cost-effectiveness with safety and quality features to address diverse market segments. Tiered product strategies can serve both premium and price-sensitive market segments effectively. Focus on safety-engineered syringes for hospital markets while maintaining affordable options for individual patients and smaller healthcare facilities.
Distribution network development should emphasize cold chain capabilities and rural market access to maximize market penetration. Multi-channel approaches including hospitals, pharmacies, and online platforms can improve product availability and customer convenience. Investment in logistics infrastructure and distributor training is essential for market success.
Regulatory compliance strategies should anticipate harmonization trends while maintaining flexibility for country-specific requirements. Proactive engagement with regulatory authorities and industry associations can provide competitive advantages and market access benefits. Quality system investments are essential for meeting evolving regulatory standards.
Innovation focus areas should prioritize safety features, ease of use, and digital integration capabilities that address regional market needs. Patient-centered design approaches can create competitive differentiation and improve market acceptance. Collaboration with healthcare providers and patients can guide product development priorities.
Long-term market prospects for the MEA insulin syringe market remain highly positive, driven by demographic trends, healthcare infrastructure development, and increasing diabetes awareness. Market expansion is expected to continue at robust rates, with compound annual growth rates exceeding 8% in key regional markets over the next decade.
Technology evolution will drive product innovation and market differentiation, with smart syringes and digital health integration becoming increasingly important. Digital transformation in healthcare will create new opportunities for insulin delivery system manufacturers to provide comprehensive diabetes management solutions. Integration with telemedicine platforms and mobile health applications will become standard features.
Market maturation in developed MEA countries will drive demand for premium products and advanced features, while emerging markets will continue to prioritize cost-effective solutions. Segmentation strategies will become increasingly important as markets develop and patient needs diversify. The gap between premium and basic market segments is expected to widen.
Regulatory environment improvements will facilitate market access and reduce operational complexities for manufacturers and distributors. Harmonization efforts will create more efficient market entry processes and reduce compliance costs. Quality standards will continue to evolve, favoring established manufacturers with robust quality systems.
Sustainability considerations will increasingly influence product development and purchasing decisions, creating opportunities for environmentally friendly insulin delivery solutions. Circular economy principles may drive innovation in syringe design and waste management systems. Corporate responsibility initiatives will become important competitive factors.
The MEA insulin syringe market represents a dynamic and rapidly evolving healthcare segment with substantial growth potential driven by increasing diabetes prevalence and healthcare infrastructure development. Market dynamics reflect the region’s diverse economic conditions, healthcare systems, and regulatory environments, creating both opportunities and challenges for industry participants.
Strategic success factors include understanding local market conditions, developing appropriate product portfolios, and building strong distribution networks. MWR analysis indicates that companies focusing on safety features, cost-effectiveness, and market access partnerships will be best positioned for long-term success in this growing market.
Future market development will be shaped by technology innovation, regulatory harmonization, and healthcare system modernization across the MEA region. The integration of digital health solutions and emphasis on patient-centered care will create new opportunities for product differentiation and market expansion. Sustainable growth requires balancing innovation with affordability while maintaining focus on patient safety and clinical outcomes.
What is Insulin Syringe?
An insulin syringe is a medical device used to inject insulin into the body for the management of diabetes. It typically consists of a barrel, plunger, and a needle, designed for precise dosage and ease of use.
What are the key players in the MEA Insulin Syringe Market?
Key players in the MEA Insulin Syringe Market include Becton, Dickinson and Company, Terumo Corporation, and Novo Nordisk, among others. These companies are known for their innovative products and extensive distribution networks.
What are the growth factors driving the MEA Insulin Syringe Market?
The growth of the MEA Insulin Syringe Market is driven by the increasing prevalence of diabetes, rising awareness about diabetes management, and advancements in syringe technology. Additionally, the growing demand for disposable syringes contributes to market expansion.
What challenges does the MEA Insulin Syringe Market face?
The MEA Insulin Syringe Market faces challenges such as regulatory hurdles, the high cost of advanced syringes, and competition from alternative drug delivery systems. These factors can impact market accessibility and growth.
What opportunities exist in the MEA Insulin Syringe Market?
Opportunities in the MEA Insulin Syringe Market include the development of smart syringes with integrated technology for better patient monitoring and the expansion of healthcare infrastructure in emerging markets. These advancements can enhance patient care and increase market penetration.
What trends are shaping the MEA Insulin Syringe Market?
Trends in the MEA Insulin Syringe Market include the shift towards user-friendly designs, the rise of prefilled syringes, and the integration of digital health solutions. These trends aim to improve patient adherence and overall diabetes management.
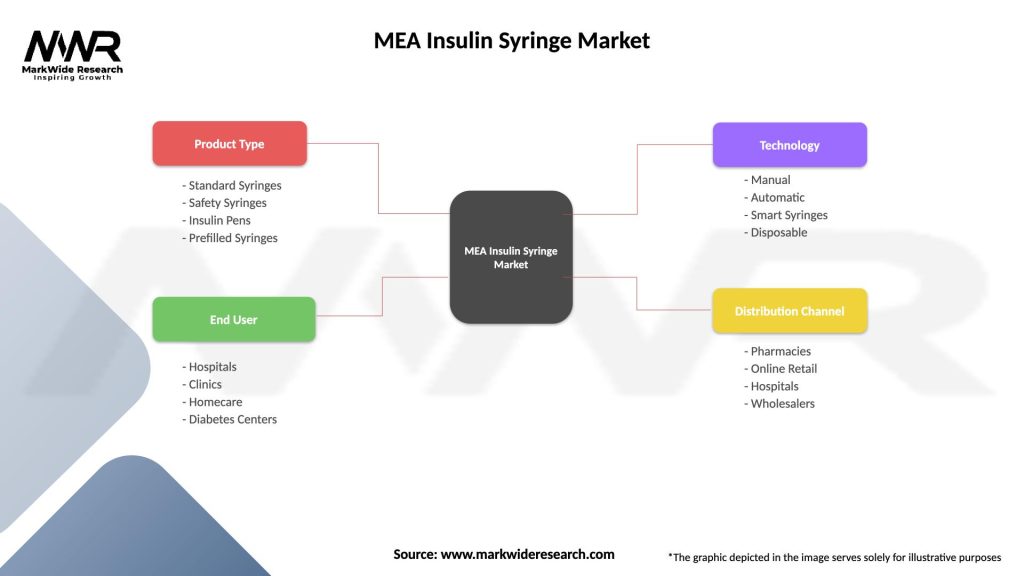
MEA Insulin Syringe Market
| Segmentation Details | Description |
|---|---|
| Product Type | Standard Syringes, Safety Syringes, Insulin Pens, Prefilled Syringes |
| End User | Hospitals, Clinics, Homecare, Diabetes Centers |
| Technology | Manual, Automatic, Smart Syringes, Disposable |
| Distribution Channel | Pharmacies, Online Retail, Hospitals, Wholesalers |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading companies in the MEA Insulin Syringe Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at