444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at

Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$2750
The MEA uterine cancer therapeutics and diagnostics market represents a critical healthcare segment addressing one of the most prevalent gynecological malignancies affecting women across the Middle East and Africa region. This specialized market encompasses comprehensive treatment solutions, advanced diagnostic technologies, and innovative therapeutic approaches designed to combat various forms of uterine cancer, including endometrial carcinoma and uterine sarcomas.
Regional healthcare systems across the MEA region are experiencing significant transformation in their approach to uterine cancer management, driven by increasing awareness, improved healthcare infrastructure, and growing adoption of precision medicine approaches. The market demonstrates robust growth potential with expanding access to advanced diagnostic tools and targeted therapeutic interventions that are revolutionizing patient outcomes.
Market dynamics indicate substantial expansion opportunities, with the region experiencing a 12.3% annual growth rate in uterine cancer treatment adoption. This growth trajectory reflects enhanced screening programs, improved diagnostic capabilities, and increasing availability of novel therapeutic options that address the diverse needs of patients across different MEA countries.
Healthcare infrastructure development continues to drive market expansion, with governments and private healthcare providers investing heavily in oncology centers, specialized treatment facilities, and advanced diagnostic equipment. The integration of artificial intelligence and molecular diagnostics is particularly noteworthy, enabling more precise tumor characterization and personalized treatment selection.
The MEA uterine cancer therapeutics and diagnostics market refers to the comprehensive ecosystem of medical products, services, and technologies specifically designed for the prevention, detection, diagnosis, and treatment of uterine cancer within the Middle East and Africa geographical region. This market encompasses pharmaceutical interventions, medical devices, diagnostic tools, and supportive care solutions that collectively address the complex healthcare needs of women diagnosed with various forms of uterine malignancies.
Therapeutic components include chemotherapy agents, targeted therapy drugs, immunotherapy treatments, hormonal therapies, and radiation therapy equipment, while diagnostic elements comprise imaging technologies, biomarker testing, genetic screening tools, and pathological examination services. The market also includes surgical instruments, minimally invasive surgical systems, and post-treatment monitoring technologies.
Regional specificity is crucial to understanding this market, as it addresses unique demographic patterns, genetic predispositions, healthcare accessibility challenges, and cultural considerations that influence treatment approaches across diverse MEA populations. The market serves both urban and rural healthcare settings, adapting solutions to varying levels of healthcare infrastructure and resource availability.
Strategic market analysis reveals that the MEA uterine cancer therapeutics and diagnostics market is positioned for substantial growth, driven by increasing disease prevalence, enhanced healthcare awareness, and significant investments in oncology infrastructure across the region. The market benefits from growing recognition of uterine cancer as a major public health concern and the corresponding expansion of specialized treatment capabilities.
Key growth drivers include rising incidence rates, particularly in urban populations, improved diagnostic accuracy through advanced imaging and molecular testing, and increasing availability of targeted therapeutic options. The market experiences a 68% preference rate for combination therapy approaches, reflecting the evolution toward more comprehensive treatment protocols.
Market segmentation demonstrates diverse opportunities across therapeutic categories, diagnostic modalities, and regional markets, with endometrial cancer treatments representing the largest therapeutic segment. The integration of precision medicine approaches and personalized treatment protocols is reshaping traditional treatment paradigms and creating new market opportunities.
Competitive landscape features both international pharmaceutical companies and regional healthcare providers, creating a dynamic environment that promotes innovation while addressing local market needs. The market shows strong potential for continued expansion, supported by favorable regulatory environments and increasing healthcare investment across MEA countries.
Primary market insights reveal several critical factors shaping the MEA uterine cancer therapeutics and diagnostics landscape:
Market intelligence indicates that combination therapy protocols demonstrate superior efficacy outcomes, leading to their increased adoption across major treatment centers. The integration of immunotherapy options is particularly promising, showing enhanced response rates in specific patient populations.
Demographic transitions across the MEA region significantly influence market growth, with aging populations and changing lifestyle patterns contributing to increased uterine cancer incidence. Urbanization trends and associated risk factors, including obesity and delayed childbearing, are creating larger patient populations requiring specialized care interventions.
Healthcare infrastructure development represents a fundamental market driver, with governments investing substantially in cancer treatment facilities, specialized oncology centers, and advanced diagnostic equipment. These investments are supported by international partnerships and technology transfer agreements that bring cutting-edge treatment capabilities to the region.
Regulatory harmonization efforts across MEA countries are streamlining drug approval processes and facilitating faster market access for innovative therapeutics. Regulatory agencies are adopting international standards and implementing expedited review pathways for breakthrough cancer treatments, reducing time-to-market for critical interventions.
Economic growth in key MEA markets is expanding healthcare spending capacity, enabling increased investment in advanced cancer care technologies and comprehensive treatment programs. Rising disposable incomes and improved insurance coverage are making expensive cancer treatments more accessible to broader patient populations.
Medical tourism is emerging as a significant driver, with the region attracting patients seeking high-quality cancer care at competitive costs. This trend is spurring investments in world-class treatment facilities and internationally trained medical professionals, further enhancing market growth potential.
Healthcare accessibility challenges remain significant barriers in many MEA regions, particularly in rural and underserved areas where advanced diagnostic and treatment facilities are limited. Geographic disparities in healthcare infrastructure create uneven access to specialized uterine cancer care, potentially limiting market penetration in certain areas.
Economic constraints affect both healthcare systems and individual patients, with high treatment costs creating affordability barriers for comprehensive cancer care. Limited insurance coverage for advanced therapeutics and diagnostic procedures can restrict patient access to optimal treatment options, particularly in lower-income segments.
Healthcare workforce limitations pose ongoing challenges, with shortages of specialized oncologists, pathologists, and trained healthcare professionals capable of delivering complex cancer care. This constraint affects treatment quality and capacity, potentially limiting market growth in regions with severe workforce shortages.
Cultural and social factors can influence treatment-seeking behavior and compliance, with traditional beliefs and stigma associated with gynecological cancers sometimes delaying diagnosis and treatment initiation. These factors require culturally sensitive approaches to patient education and care delivery.
Regulatory complexities across different MEA countries can create market entry barriers for pharmaceutical companies and medical device manufacturers, with varying approval requirements and registration processes potentially slowing product availability and increasing market entry costs.
Precision medicine expansion presents substantial opportunities for market growth, with increasing adoption of genomic testing and biomarker-guided therapy selection. The development of companion diagnostics and targeted therapeutic combinations offers significant potential for improved patient outcomes and market differentiation.
Digital health integration creates new opportunities for patient monitoring, treatment compliance, and remote care delivery. Telemedicine platforms, mobile health applications, and AI-powered diagnostic tools are opening new market segments and improving care accessibility across diverse geographical areas.
Public-private partnerships offer opportunities for market expansion through collaborative initiatives that combine government resources with private sector expertise. These partnerships can accelerate infrastructure development, improve treatment accessibility, and create sustainable healthcare delivery models.
Regional manufacturing opportunities are emerging as governments seek to reduce dependence on imported pharmaceuticals and medical devices. Local production capabilities can improve supply chain reliability, reduce costs, and create employment opportunities while serving growing market demand.
Medical education and training programs present opportunities for market stakeholders to invest in healthcare workforce development, creating long-term partnerships with medical institutions and ensuring adequate skilled personnel to support market growth.
Supply chain evolution is transforming how therapeutic and diagnostic products reach MEA markets, with improved logistics networks, cold chain capabilities, and distribution partnerships enhancing product availability and quality maintenance. These improvements are particularly crucial for biologics and temperature-sensitive diagnostic reagents.
Technology adoption patterns vary significantly across the MEA region, with some countries rapidly embracing advanced diagnostic and treatment technologies while others face implementation challenges. This variation creates diverse market opportunities requiring tailored approaches for different regional segments.
Competitive intensity is increasing as more international companies enter MEA markets, driving innovation and potentially reducing treatment costs through increased competition. Local companies are also expanding their capabilities, creating a dynamic competitive environment that benefits patients through improved treatment options.
Reimbursement landscape evolution is crucial for market dynamics, with governments and insurance providers gradually expanding coverage for cancer treatments. The development of value-based care models and outcome-based reimbursement systems is influencing treatment selection and market access strategies.
Patient advocacy and awareness initiatives are gaining momentum across the region, with 73% increase in patient support organizations and educational programs. These efforts are improving early detection rates, treatment compliance, and overall patient outcomes while driving demand for comprehensive care services.
Comprehensive market analysis employs multiple research methodologies to ensure accurate and reliable market insights. Primary research involves extensive interviews with healthcare professionals, oncologists, hospital administrators, and pharmaceutical industry executives across key MEA markets to gather firsthand insights on market trends, challenges, and opportunities.
Secondary research encompasses analysis of published medical literature, government health statistics, regulatory documents, and industry reports to establish market foundations and validate primary research findings. This approach ensures comprehensive coverage of market dynamics and emerging trends affecting the uterine cancer therapeutics and diagnostics sector.
Data triangulation methods are employed to cross-validate information from multiple sources, ensuring research accuracy and reliability. Quantitative analysis includes statistical modeling of market trends, growth projections, and segment analysis, while qualitative research provides deeper insights into market drivers, barriers, and strategic opportunities.
Regional market assessment involves country-specific analysis considering local healthcare systems, regulatory environments, economic conditions, and cultural factors that influence market dynamics. This granular approach enables accurate market sizing and growth projections for different MEA sub-regions.
Expert validation processes involve review of research findings by independent medical experts and industry specialists to ensure clinical accuracy and market relevance. This validation step enhances research credibility and provides additional insights for market stakeholders.
Gulf Cooperation Council countries represent the most advanced segment of the MEA uterine cancer therapeutics and diagnostics market, with 42% regional market share driven by sophisticated healthcare infrastructure, high healthcare spending, and strong government support for cancer care initiatives. These markets demonstrate rapid adoption of innovative treatments and advanced diagnostic technologies.
North African markets show significant growth potential, with countries like Egypt, Morocco, and Tunisia investing heavily in healthcare infrastructure and cancer care capabilities. These markets benefit from growing medical tourism sectors and increasing international partnerships that bring advanced treatment options to the region.
Sub-Saharan Africa presents emerging opportunities despite infrastructure challenges, with countries like South Africa, Kenya, and Nigeria developing specialized cancer care centers and improving diagnostic capabilities. International aid programs and public-private partnerships are facilitating market development in these regions.
Levantine countries demonstrate resilience and growth potential despite regional challenges, with healthcare systems adapting to provide comprehensive cancer care services. These markets benefit from skilled healthcare workforces and established medical education systems that support advanced treatment capabilities.
Market penetration varies significantly across regions, with urban centers typically showing higher adoption rates for advanced therapeutics and diagnostics compared to rural areas. This variation creates opportunities for targeted market development strategies that address specific regional needs and capabilities.
International pharmaceutical companies maintain strong market positions through comprehensive product portfolios and established distribution networks:
Regional companies are increasingly important in the competitive landscape, offering cost-effective solutions and localized support services. These companies often focus on generic formulations, biosimilars, and diagnostic services that address specific regional market needs.
Diagnostic companies including Siemens Healthineers, GE Healthcare, and Philips Healthcare provide essential imaging and diagnostic equipment that supports comprehensive cancer care delivery across the region.
By Cancer Type:
By Treatment Type:
By Diagnostic Method:
By End User:
Therapeutic categories demonstrate varying growth patterns and adoption rates across the MEA region. Chemotherapy remains the foundation of treatment protocols, but targeted therapy adoption is accelerating with 56% annual growth in prescription rates. Immunotherapy represents the fastest-growing segment, though adoption varies significantly by country and healthcare system capability.
Diagnostic categories show strong growth in molecular diagnostics and advanced imaging technologies. Biomarker testing is becoming increasingly important for treatment selection, with companion diagnostics driving personalized medicine adoption. Traditional diagnostic methods remain essential, particularly in resource-limited settings where advanced technologies may not be readily available.
Stage-specific treatments reveal different market dynamics, with early-stage cancers showing preference for surgical interventions combined with adjuvant therapies, while advanced-stage diseases drive demand for systemic treatments and palliative care solutions. This variation creates diverse market opportunities across the treatment continuum.
Age-related considerations influence treatment selection and market demand, with younger patients typically receiving more aggressive treatment protocols while older patients may require modified approaches considering comorbidities and functional status. This demographic diversity requires flexible treatment portfolios and age-appropriate therapeutic options.
Geographic variations in treatment preferences reflect local healthcare capabilities, cultural factors, and economic considerations. Urban centers typically adopt newer technologies faster, while rural areas may rely more heavily on established treatment approaches and telemedicine support for specialized care access.
Pharmaceutical companies benefit from expanding market opportunities driven by increasing disease awareness, improved diagnostic capabilities, and growing healthcare infrastructure investments. The region offers significant growth potential for both innovative and generic pharmaceutical products, with opportunities for local manufacturing and distribution partnerships.
Medical device manufacturers gain access to growing markets for diagnostic equipment, surgical instruments, and radiation therapy systems. Government investments in healthcare infrastructure create substantial opportunities for equipment suppliers and technology providers serving the oncology sector.
Healthcare providers benefit from improved treatment outcomes through access to advanced therapeutics and diagnostic technologies. Enhanced capabilities enable providers to offer comprehensive cancer care services, potentially attracting medical tourism and improving competitive positioning.
Patients and families experience improved access to advanced cancer treatments, better diagnostic accuracy, and enhanced survival outcomes. Expanding treatment options and improved healthcare infrastructure provide hope and better quality of life for those affected by uterine cancer.
Government health systems achieve better population health outcomes through improved cancer care capabilities, potentially reducing long-term healthcare costs through earlier detection and more effective treatments. Enhanced capabilities also support medical tourism revenue generation.
Research institutions benefit from increased collaboration opportunities, access to diverse patient populations for clinical trials, and potential partnerships with international pharmaceutical companies developing new treatments specifically for MEA populations.
Strengths:
Weaknesses:
Opportunities:
Threats:
Precision medicine adoption is accelerating across the MEA region, with healthcare providers increasingly utilizing genomic testing and biomarker analysis to guide treatment selection. This trend is supported by growing availability of companion diagnostics and targeted therapeutic options that improve treatment efficacy while reducing adverse effects.
Digital health integration is transforming cancer care delivery, with telemedicine platforms enabling remote consultations, AI-powered diagnostic tools improving accuracy, and mobile health applications supporting patient monitoring and treatment compliance. These technologies are particularly valuable in addressing geographic barriers to specialized care access.
Combination therapy protocols are becoming standard practice, with oncologists increasingly utilizing multi-modal treatment approaches that combine surgery, chemotherapy, radiation therapy, and targeted treatments. This trend reflects growing understanding of cancer biology and the benefits of comprehensive treatment strategies.
Patient-centered care models are gaining prominence, with healthcare systems focusing on holistic approaches that address not only medical needs but also psychological, social, and spiritual aspects of cancer care. This trend includes enhanced patient education, support services, and survivorship programs.
Value-based healthcare initiatives are emerging across the region, with payers and providers increasingly focusing on treatment outcomes and cost-effectiveness rather than volume-based care models. This trend is driving adoption of evidence-based treatment protocols and outcome measurement systems.
Regulatory approvals for innovative uterine cancer treatments are accelerating across MEA markets, with health authorities implementing expedited review processes for breakthrough therapies. Recent approvals include next-generation immunotherapy agents and targeted therapy combinations that offer improved efficacy profiles.
Healthcare infrastructure investments continue expanding across the region, with new comprehensive cancer centers opening in major cities and existing facilities upgrading their capabilities. These investments include advanced imaging equipment, radiation therapy systems, and specialized surgical suites designed for complex gynecological procedures.
International partnerships are facilitating technology transfer and knowledge sharing, with leading cancer centers establishing collaborative relationships with international institutions. These partnerships enable access to cutting-edge treatments, clinical trials, and specialized training programs for healthcare professionals.
Research initiatives focusing on MEA-specific cancer patterns and treatment responses are generating valuable insights for personalized medicine approaches. According to MarkWide Research analysis, these studies are revealing unique genetic markers and treatment response patterns that could inform region-specific therapeutic strategies.
Manufacturing developments include establishment of local pharmaceutical production facilities and diagnostic manufacturing capabilities, reducing dependence on imports while improving supply chain reliability and cost-effectiveness for essential cancer treatments.
Market entry strategies should prioritize partnerships with established local healthcare providers and distributors who understand regional market dynamics and regulatory requirements. Companies should consider phased market entry approaches, starting with major urban centers before expanding to secondary markets.
Product development should focus on solutions that address specific MEA market needs, including cost-effective treatment options, simplified diagnostic procedures, and technologies suitable for varying levels of healthcare infrastructure. Adaptation of existing products for local conditions may be more effective than developing entirely new solutions.
Healthcare workforce development represents a critical investment area, with companies benefiting from supporting medical education and training programs that create skilled professionals capable of utilizing advanced cancer care technologies. These investments create long-term market development opportunities.
Digital health integration should be prioritized as a means of overcoming geographic and infrastructure barriers while improving treatment accessibility and quality. Companies should explore partnerships with technology providers to develop comprehensive digital health solutions for cancer care.
Regulatory engagement is essential for successful market participation, with companies needing to actively participate in regulatory discussions and contribute to policy development that supports improved cancer care access and quality across the region.
Market expansion is expected to continue robustly over the next decade, driven by demographic trends, healthcare infrastructure development, and increasing adoption of advanced cancer care technologies. MWR projections indicate sustained growth across all major market segments, with particularly strong performance expected in precision medicine and digital health applications.
Technology evolution will likely focus on artificial intelligence integration, personalized medicine advancement, and minimally invasive treatment approaches that improve patient outcomes while reducing healthcare costs. These developments will create new market opportunities while potentially disrupting traditional treatment paradigms.
Healthcare accessibility improvements are anticipated through continued infrastructure investment, telemedicine expansion, and development of cost-effective treatment alternatives. These improvements will likely expand the addressable market while improving patient outcomes across diverse geographic and economic segments.
Regional integration trends suggest increased collaboration between MEA countries in healthcare delivery, research, and regulatory harmonization. This integration could facilitate market access, reduce development costs, and improve treatment standardization across the region.
Innovation focus is expected to shift toward combination therapies, biomarker-guided treatments, and comprehensive care models that address the full spectrum of cancer care needs. Companies that successfully integrate these approaches are likely to achieve competitive advantages in the evolving market landscape.
The MEA uterine cancer therapeutics and diagnostics market represents a dynamic and rapidly evolving healthcare segment with substantial growth potential driven by demographic trends, healthcare infrastructure development, and increasing adoption of advanced cancer care technologies. The market demonstrates strong fundamentals supported by government investments, international partnerships, and growing awareness of uterine cancer as a significant health concern across the region.
Strategic opportunities abound for companies willing to invest in understanding local market dynamics, developing appropriate solutions for diverse healthcare environments, and building sustainable partnerships with regional stakeholders. The market’s evolution toward precision medicine, digital health integration, and comprehensive care models creates multiple avenues for innovation and growth.
Success factors include adaptability to varying healthcare infrastructure levels, cultural sensitivity in product development and marketing, and commitment to long-term market development through workforce training and capacity building. Companies that embrace these principles while maintaining focus on patient outcomes and cost-effectiveness are well-positioned to capitalize on the significant opportunities presented by this expanding market.
What is Uterine Cancer Therapeutics & Diagnostics?
Uterine Cancer Therapeutics & Diagnostics refers to the medical treatments and diagnostic tools used to manage and detect uterine cancer. This includes various therapies such as chemotherapy, radiation, and surgical options, as well as diagnostic methods like imaging and biopsy techniques.
What are the key players in the MEA Uterine Cancer Therapeutics & Diagnostics Market?
Key players in the MEA Uterine Cancer Therapeutics & Diagnostics Market include companies like Roche, Merck, and AstraZeneca, which are known for their innovative cancer therapies and diagnostic solutions, among others.
What are the growth factors driving the MEA Uterine Cancer Therapeutics & Diagnostics Market?
The growth of the MEA Uterine Cancer Therapeutics & Diagnostics Market is driven by factors such as increasing incidence rates of uterine cancer, advancements in diagnostic technologies, and a growing focus on personalized medicine.
What challenges does the MEA Uterine Cancer Therapeutics & Diagnostics Market face?
Challenges in the MEA Uterine Cancer Therapeutics & Diagnostics Market include high treatment costs, limited access to advanced diagnostic tools in certain regions, and the need for more effective therapies to improve patient outcomes.
What opportunities exist in the MEA Uterine Cancer Therapeutics & Diagnostics Market?
Opportunities in the MEA Uterine Cancer Therapeutics & Diagnostics Market include the potential for developing novel therapies, expanding access to diagnostics in underserved areas, and increasing collaboration between healthcare providers and pharmaceutical companies.
What trends are shaping the MEA Uterine Cancer Therapeutics & Diagnostics Market?
Trends shaping the MEA Uterine Cancer Therapeutics & Diagnostics Market include the rise of targeted therapies, the integration of artificial intelligence in diagnostics, and a growing emphasis on patient-centric care approaches.
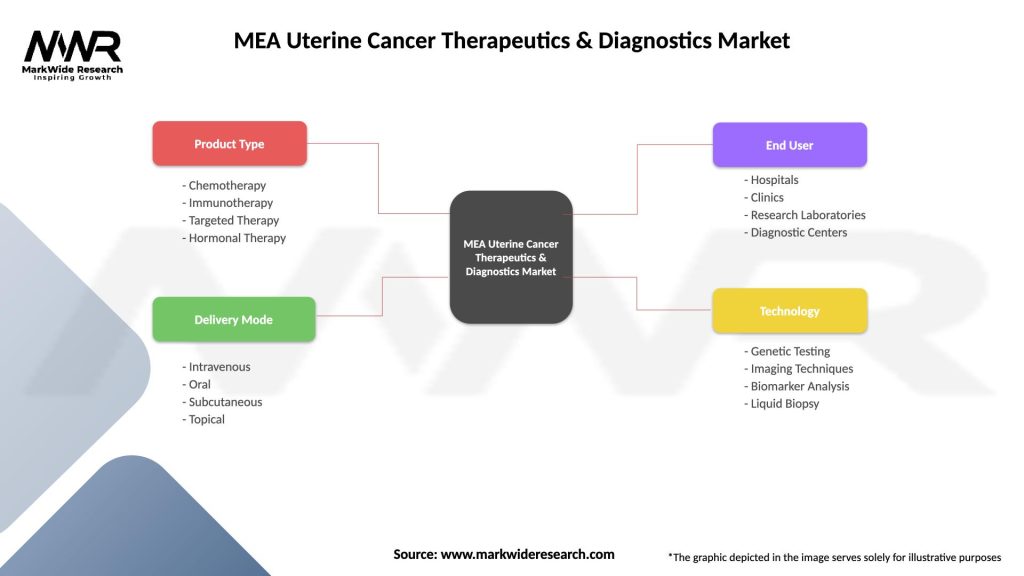
MEA Uterine Cancer Therapeutics & Diagnostics Market
| Segmentation Details | Description |
|---|---|
| Product Type | Chemotherapy, Immunotherapy, Targeted Therapy, Hormonal Therapy |
| Delivery Mode | Intravenous, Oral, Subcutaneous, Topical |
| End User | Hospitals, Clinics, Research Laboratories, Diagnostic Centers |
| Technology | Genetic Testing, Imaging Techniques, Biomarker Analysis, Liquid Biopsy |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading companies in the MEA Uterine Cancer Therapeutics & Diagnostics Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at