444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at

Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$2750
The Europe pharmaceutical plastic bottles market represents a critical segment of the broader pharmaceutical packaging industry, experiencing robust growth driven by increasing healthcare demands and stringent regulatory requirements. Pharmaceutical plastic bottles serve as essential primary packaging solutions for liquid medications, tablets, capsules, and other pharmaceutical products across European markets. The market encompasses various plastic materials including polyethylene terephthalate (PET), high-density polyethylene (HDPE), polypropylene (PP), and specialized barrier materials designed to protect pharmaceutical contents from environmental factors.
Market dynamics indicate significant expansion opportunities, with the sector growing at a steady CAGR of approximately 6.2% throughout the forecast period. This growth trajectory reflects the increasing adoption of plastic packaging solutions over traditional glass alternatives, driven by advantages such as lightweight properties, cost-effectiveness, and enhanced safety profiles. European pharmaceutical manufacturers are increasingly recognizing the benefits of plastic bottle packaging, particularly for over-the-counter medications and prescription drugs requiring extended shelf life.
Regional distribution shows Germany, France, and the United Kingdom leading market adoption, collectively accounting for over 55% of regional demand. The market’s evolution is closely tied to pharmaceutical industry growth, aging population demographics, and evolving consumer preferences for convenient packaging formats. Sustainability initiatives are also shaping market development, with manufacturers increasingly focusing on recyclable and bio-based plastic materials to meet environmental regulations and corporate responsibility goals.
The Europe pharmaceutical plastic bottles market refers to the comprehensive ecosystem encompassing the production, distribution, and utilization of plastic bottle packaging specifically designed for pharmaceutical applications across European countries. These specialized containers are engineered to meet stringent pharmaceutical standards, ensuring product integrity, safety, and compliance with regulatory requirements established by the European Medicines Agency (EMA) and national health authorities.
Pharmaceutical plastic bottles differ significantly from conventional plastic containers through their advanced material compositions, barrier properties, and manufacturing processes. They incorporate features such as child-resistant closures, tamper-evident seals, light protection capabilities, and moisture barrier properties essential for maintaining pharmaceutical product efficacy. The market encompasses various bottle sizes, shapes, and configurations tailored to specific medication types and dosage requirements.
Market scope includes primary packaging solutions for liquid pharmaceuticals, solid dosage forms, nutraceuticals, and veterinary medicines. The definition extends beyond simple container manufacturing to include comprehensive packaging systems integrating bottles, closures, labels, and secondary packaging components that collectively ensure pharmaceutical product safety and regulatory compliance throughout the European Union.
Strategic market analysis reveals the Europe pharmaceutical plastic bottles market as a dynamic and rapidly evolving sector characterized by technological innovation, regulatory compliance, and sustainability initiatives. The market demonstrates resilient growth patterns driven by fundamental healthcare trends including population aging, chronic disease prevalence, and expanding pharmaceutical production capabilities across European manufacturing hubs.
Key growth drivers include the ongoing shift from glass to plastic packaging solutions, representing approximately 42% of packaging conversion activities among European pharmaceutical manufacturers. This transition is motivated by cost reduction opportunities, improved supply chain efficiency, and enhanced patient safety considerations. Regulatory harmonization across EU member states has facilitated market expansion by standardizing packaging requirements and enabling cross-border pharmaceutical distribution.
Market segmentation analysis indicates liquid pharmaceutical applications dominating demand, followed by solid dosage forms and specialized therapeutic categories. The prescription medication segment maintains the largest market share, while over-the-counter products show the highest growth rates. Innovation trends focus on smart packaging technologies, sustainable materials, and enhanced barrier properties to meet evolving pharmaceutical industry requirements and environmental sustainability goals.
Market intelligence reveals several critical insights shaping the Europe pharmaceutical plastic bottles landscape. The following key insights provide strategic understanding of market dynamics and growth opportunities:
Primary market drivers propelling the Europe pharmaceutical plastic bottles market encompass demographic, technological, and regulatory factors creating sustained demand growth. The aging European population represents a fundamental driver, with individuals over 65 years requiring increased pharmaceutical interventions and convenient packaging solutions that plastic bottles effectively provide.
Cost optimization pressures within the pharmaceutical industry drive adoption of plastic bottle packaging as manufacturers seek to reduce packaging costs while maintaining product quality and safety standards. Plastic bottles offer significant cost advantages over glass alternatives, with manufacturing cost reductions of approximately 35% compared to equivalent glass packaging solutions. This economic benefit becomes particularly important as pharmaceutical companies face pricing pressures from healthcare systems and regulatory authorities.
Technological advancement in plastic manufacturing enables development of specialized pharmaceutical-grade materials with enhanced properties including improved barrier characteristics, chemical resistance, and compatibility with various drug formulations. These innovations expand application possibilities and drive market adoption across diverse pharmaceutical categories. Regulatory support for plastic packaging solutions, evidenced by updated guidelines and approval processes, facilitates market growth by providing clear pathways for product development and commercialization.
Sustainability mandates from European Union environmental policies encourage adoption of recyclable plastic packaging solutions, creating opportunities for manufacturers developing eco-friendly pharmaceutical bottle alternatives. The circular economy framework promotes plastic bottle recycling and reuse, supporting long-term market sustainability and growth prospects.
Significant market restraints challenge the Europe pharmaceutical plastic bottles market, requiring strategic navigation by industry participants. Regulatory complexity represents a primary constraint, with varying requirements across EU member states creating compliance challenges and increased operational costs for manufacturers serving multiple markets simultaneously.
Material compatibility concerns limit plastic bottle adoption for certain pharmaceutical formulations, particularly those containing aggressive solvents or requiring absolute barrier protection that only glass packaging can provide. These technical limitations restrict market penetration in specialized therapeutic areas and high-value pharmaceutical segments where product integrity requirements exceed plastic material capabilities.
Environmental scrutiny of plastic packaging creates reputational and regulatory risks for pharmaceutical companies, despite recycling initiatives and sustainable material development. Public perception challenges regarding plastic waste impact healthcare industry packaging decisions, potentially slowing adoption rates in environmentally conscious markets. Quality control complexity associated with plastic bottle manufacturing requires sophisticated testing and validation processes, increasing production costs and time-to-market for new packaging solutions.
Supply chain vulnerabilities exposed during recent global disruptions highlight dependencies on raw material availability and manufacturing capacity, creating potential constraints on market growth. Competition from alternative packaging formats including glass, aluminum, and innovative biodegradable materials presents ongoing challenges to plastic bottle market share expansion.
Emerging opportunities within the Europe pharmaceutical plastic bottles market present significant growth potential for innovative manufacturers and strategic investors. The expanding biologics and biosimilar pharmaceutical segments create demand for specialized packaging solutions with enhanced stability and protection characteristics that advanced plastic bottle technologies can address effectively.
Digital health integration opportunities enable pharmaceutical plastic bottles to incorporate smart technologies including sensors, indicators, and connectivity features that enhance patient adherence monitoring and medication management. These value-added capabilities command premium pricing and differentiate products in competitive markets. Personalized medicine trends drive demand for smaller batch sizes and customized packaging solutions, favoring flexible plastic bottle manufacturing processes over traditional glass packaging alternatives.
Sustainability innovation presents opportunities for developing bio-based and biodegradable plastic bottle materials that meet pharmaceutical quality requirements while addressing environmental concerns. Companies successfully developing these solutions can capture market share from traditional materials and benefit from regulatory incentives supporting sustainable packaging adoption. Market expansion into Eastern European countries offers growth opportunities as healthcare infrastructure development and pharmaceutical market maturation create increased demand for quality packaging solutions.
Contract manufacturing growth enables specialized plastic bottle manufacturers to serve pharmaceutical companies lacking internal packaging capabilities, creating service-based revenue opportunities beyond traditional product sales. This trend particularly benefits companies with advanced technical capabilities and regulatory expertise.
Complex market dynamics shape the Europe pharmaceutical plastic bottles market through interconnected forces influencing supply, demand, and competitive positioning. The relationship between pharmaceutical industry growth and packaging demand creates direct correlation patterns, with pharmaceutical production increases driving 78% of packaging demand growth across European markets.
Competitive dynamics reflect industry consolidation trends as larger packaging companies acquire specialized pharmaceutical bottle manufacturers to expand capabilities and market reach. This consolidation creates economies of scale while potentially reducing supplier options for pharmaceutical companies. Innovation cycles driven by regulatory changes and technological advancement create periodic market disruptions requiring continuous adaptation by industry participants.
Pricing dynamics demonstrate sensitivity to raw material costs, particularly petroleum-based plastic resins, creating margin pressure during commodity price volatility periods. Manufacturers increasingly implement cost-plus pricing models and long-term supply agreements to manage these fluctuations. Quality dynamics emphasize continuous improvement in manufacturing processes and testing capabilities as regulatory requirements evolve and pharmaceutical companies demand higher performance standards.
Geographic dynamics reflect varying market maturity levels across European regions, with Western European markets showing stable growth while Eastern European markets demonstrate higher growth rates but smaller absolute volumes. These regional differences create opportunities for targeted market development strategies and localized manufacturing approaches.
Comprehensive research methodology employed in analyzing the Europe pharmaceutical plastic bottles market incorporates multiple data collection and analysis techniques to ensure accuracy and reliability of market insights. Primary research activities include structured interviews with industry executives, pharmaceutical packaging managers, and regulatory specialists across major European markets to gather firsthand market intelligence and trend observations.
Secondary research components encompass analysis of industry publications, regulatory filings, company financial reports, and trade association data to establish market baseline information and validate primary research findings. MarkWide Research analytical frameworks integrate quantitative and qualitative research methodologies to provide balanced market perspectives addressing both statistical trends and strategic implications.
Market sizing methodology utilizes bottom-up and top-down approaches, analyzing pharmaceutical production volumes, packaging consumption ratios, and regional market penetration rates to develop accurate market assessments. Data triangulation processes compare multiple information sources to ensure consistency and identify potential discrepancies requiring additional investigation.
Forecasting methodology incorporates econometric modeling, trend analysis, and scenario planning to project future market development under various assumption sets. Expert validation processes involve industry specialists reviewing research findings and providing feedback on market projections and strategic recommendations to enhance analytical accuracy and practical relevance.
Regional market analysis reveals distinct patterns and opportunities across European pharmaceutical plastic bottles markets, with Western European countries maintaining market leadership while Eastern European regions demonstrate accelerating growth trajectories. Germany represents the largest individual market, accounting for approximately 22% of regional demand, driven by robust pharmaceutical manufacturing capabilities and advanced packaging technology adoption.
France and United Kingdom collectively contribute 28% of market volume, benefiting from established pharmaceutical industries and favorable regulatory environments supporting plastic packaging adoption. These markets demonstrate mature characteristics with steady growth rates and emphasis on premium packaging solutions for specialized pharmaceutical applications. Italy and Spain show increasing market participation, particularly in generic pharmaceutical packaging where cost optimization drives plastic bottle adoption over traditional glass alternatives.
Nordic countries including Sweden, Denmark, and Norway exhibit high per-capita pharmaceutical consumption and strong environmental consciousness, creating demand for sustainable plastic bottle solutions and innovative packaging technologies. These markets often serve as testing grounds for new packaging concepts before broader European deployment.
Eastern European markets including Poland, Czech Republic, and Hungary demonstrate the highest growth rates, with annual expansion exceeding 8.5% as healthcare infrastructure development and pharmaceutical market liberalization drive increased packaging demand. These regions offer significant opportunities for market expansion and manufacturing investment, supported by competitive labor costs and improving regulatory frameworks.
Competitive landscape analysis reveals a dynamic market structure characterized by both global packaging giants and specialized pharmaceutical bottle manufacturers competing across various market segments. The market demonstrates moderate concentration with leading companies maintaining significant market shares through technological innovation, regulatory expertise, and comprehensive service offerings.
Competitive strategies emphasize technological differentiation, regulatory compliance excellence, and customer service capabilities. Leading companies invest heavily in research and development to create innovative packaging solutions addressing evolving pharmaceutical industry requirements and regulatory standards.
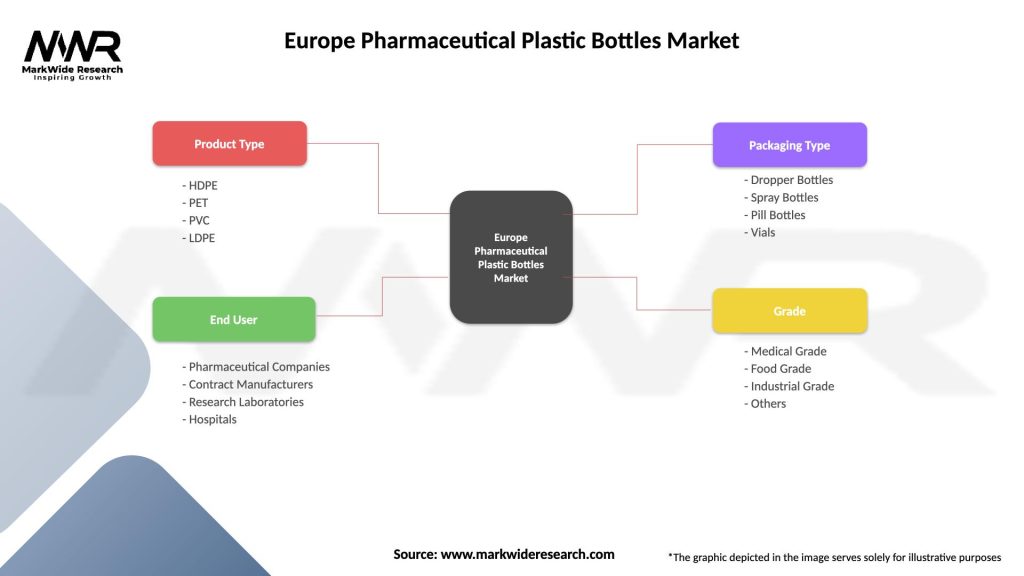
Market segmentation analysis provides detailed insights into various market categories and their respective growth patterns, enabling strategic decision-making for industry participants. The Europe pharmaceutical plastic bottles market demonstrates clear segmentation across multiple dimensions including material type, application, end-user, and bottle capacity.
By Material Type:
By Application:
Detailed category analysis reveals specific trends and opportunities within major pharmaceutical plastic bottle segments, providing actionable insights for market participants seeking to optimize their strategic positioning and product development initiatives.
Prescription Medication Category represents the most stringent segment requiring comprehensive regulatory compliance and advanced packaging technologies. This category demonstrates steady growth with annual expansion of approximately 5.8%, driven by aging population demographics and increasing chronic disease prevalence. Manufacturers serving this segment must maintain extensive quality systems and regulatory documentation to meet pharmaceutical industry standards.
Over-the-Counter (OTC) Medication Category shows the highest growth potential, benefiting from consumer preference for convenient packaging and self-medication trends. This segment emphasizes cost optimization and consumer appeal, creating opportunities for innovative packaging designs and sustainable material adoption. Market penetration rates in OTC applications continue expanding as plastic bottles replace traditional glass packaging for safety and convenience benefits.
Specialty Pharmaceutical Category including biologics and high-value medications requires advanced packaging solutions with enhanced barrier properties and stability characteristics. This premium segment commands higher pricing and margins while demanding significant technical expertise and regulatory compliance capabilities from packaging suppliers.
Generic Pharmaceutical Category focuses primarily on cost optimization while maintaining quality standards, creating opportunities for efficient manufacturing processes and standardized packaging solutions. This segment drives volume growth and supports market expansion in price-sensitive applications.
Industry participants across the Europe pharmaceutical plastic bottles market value chain realize significant benefits through strategic market participation and technological innovation. Pharmaceutical manufacturers benefit from cost reduction opportunities, with plastic bottle adoption delivering average packaging cost savings of 30-40% compared to glass alternatives while maintaining product quality and regulatory compliance.
Packaging manufacturers benefit from stable demand patterns and long-term customer relationships characteristic of pharmaceutical industry partnerships. The regulatory complexity and quality requirements create barriers to entry that protect established suppliers while enabling premium pricing for specialized solutions. Innovation opportunities in smart packaging and sustainable materials provide differentiation possibilities and competitive advantages.
Healthcare providers benefit from improved medication safety and patient compliance enabled by plastic bottle packaging features including child-resistant closures, easy-open designs, and clear labeling capabilities. These benefits translate into reduced healthcare costs and improved patient outcomes. Patients and consumers benefit from enhanced medication accessibility, safety features, and convenience factors that plastic bottle packaging provides compared to traditional alternatives.
Environmental stakeholders benefit from increasing adoption of recyclable and sustainable plastic bottle materials, supporting circular economy objectives and reducing packaging waste. The lightweight properties of plastic bottles also reduce transportation emissions and environmental impact throughout the supply chain.
Strengths:
Weaknesses:
Opportunities:
Threats:
Transformative trends shaping the Europe pharmaceutical plastic bottles market reflect broader industry evolution toward sustainability, digitalization, and patient-centric packaging solutions. Sustainability integration represents the most significant trend, with manufacturers increasingly adopting recycled content and developing bio-based plastic materials to address environmental concerns and regulatory requirements.
Smart packaging adoption accelerates as pharmaceutical companies seek to enhance patient engagement and medication adherence through digital integration. These technologies include NFC tags, QR codes, and sensor integration that provide medication information, dosing reminders, and adherence tracking capabilities. Implementation rates for smart packaging features are increasing by approximately 25% annually across European pharmaceutical markets.
Customization demand grows as pharmaceutical companies seek differentiated packaging solutions tailored to specific therapeutic areas and patient demographics. This trend drives development of specialized bottle designs, closure systems, and labeling approaches that enhance brand recognition and patient experience. Personalized medicine advancement creates additional customization requirements for smaller batch sizes and specialized packaging configurations.
Regulatory harmonization across European Union member states simplifies compliance requirements and enables more efficient market access for pharmaceutical plastic bottle manufacturers. This trend reduces operational complexity and supports market expansion initiatives. Quality system integration incorporating advanced testing and validation technologies ensures consistent product performance and regulatory compliance across diverse applications.
Recent industry developments demonstrate accelerating innovation and strategic positioning within the Europe pharmaceutical plastic bottles market. Major packaging manufacturers have announced significant investments in sustainable material development, with several companies launching bio-based plastic bottle solutions that meet pharmaceutical quality standards while addressing environmental sustainability requirements.
Technological advancement in barrier coating technologies enables plastic bottles to achieve performance characteristics previously available only in glass packaging. These developments expand application possibilities for plastic bottles in sensitive pharmaceutical formulations and high-value medications. Manufacturing automation investments improve production efficiency and quality consistency while reducing operational costs for pharmaceutical plastic bottle manufacturers.
Strategic partnerships between packaging manufacturers and pharmaceutical companies create collaborative development programs focused on innovative packaging solutions and supply chain optimization. These partnerships enable customized product development and long-term supply agreements that benefit both parties. MWR analysis indicates that such collaborative approaches are becoming increasingly important for market success and competitive differentiation.
Regulatory approval of new plastic materials and manufacturing processes expands market opportunities while ensuring patient safety and product quality. Recent approvals include advanced barrier materials and recycled content formulations that meet pharmaceutical industry requirements. Market consolidation activities including acquisitions and joint ventures reshape competitive dynamics and create opportunities for enhanced market coverage and technological capabilities.
Strategic recommendations for Europe pharmaceutical plastic bottles market participants emphasize the importance of sustainability integration, technological innovation, and regulatory compliance excellence. Companies should prioritize development of environmentally sustainable packaging solutions that meet both pharmaceutical quality requirements and evolving environmental regulations across European markets.
Investment priorities should focus on advanced manufacturing technologies that improve production efficiency, quality consistency, and customization capabilities. Automation and digital integration investments enable competitive advantages through cost reduction and enhanced service capabilities. Market expansion strategies should target high-growth Eastern European markets while maintaining strong positions in established Western European markets.
Partnership development with pharmaceutical companies creates opportunities for collaborative innovation and long-term supply relationships that provide market stability and growth potential. These partnerships should focus on developing specialized packaging solutions for emerging therapeutic areas and patient-centric packaging features. Regulatory expertise development remains critical for success in pharmaceutical packaging markets, requiring continuous investment in compliance capabilities and quality systems.
Innovation focus should emphasize smart packaging technologies, sustainable materials, and enhanced barrier properties that address evolving pharmaceutical industry requirements. Companies successfully developing these capabilities will capture market share and command premium pricing in competitive markets. Supply chain resilience improvements including diversified sourcing and regional manufacturing capabilities reduce operational risks and improve customer service levels.
Future market prospects for the Europe pharmaceutical plastic bottles market remain highly positive, supported by fundamental healthcare trends, technological advancement, and regulatory evolution. MarkWide Research projections indicate sustained growth momentum with annual expansion rates exceeding 6% throughout the forecast period, driven by pharmaceutical industry growth and continued plastic packaging adoption.
Technological evolution will continue transforming market dynamics through advanced material development, smart packaging integration, and manufacturing process innovation. These developments will expand application possibilities and create new value propositions for pharmaceutical plastic bottle solutions. Sustainability requirements will increasingly influence material selection and manufacturing processes, creating opportunities for companies developing environmentally responsible packaging solutions.
Market maturation in Western European countries will be balanced by accelerating growth in Eastern European markets as healthcare infrastructure development and pharmaceutical market expansion drive increased packaging demand. This geographic diversification provides stability and growth opportunities for market participants with appropriate regional strategies.
Regulatory evolution will continue supporting plastic packaging adoption through updated guidelines and approval processes that recognize technological advancement and safety benefits. The harmonization of European Union packaging regulations will facilitate market access and reduce compliance complexity for manufacturers serving multiple markets. Innovation acceleration in areas including personalized medicine, biologics, and digital health will create additional demand for specialized packaging solutions and drive market expansion beyond traditional pharmaceutical applications.
The Europe pharmaceutical plastic bottles market represents a dynamic and rapidly evolving sector with significant growth potential driven by healthcare industry expansion, technological innovation, and regulatory support. Market analysis reveals strong fundamentals including cost advantages, safety benefits, and manufacturing flexibility that position plastic bottles as preferred packaging solutions for diverse pharmaceutical applications across European markets.
Strategic opportunities abound for companies capable of navigating regulatory complexity, investing in sustainable innovation, and developing collaborative partnerships with pharmaceutical manufacturers. The market’s evolution toward smart packaging, sustainable materials, and customized solutions creates differentiation possibilities and competitive advantages for innovative market participants.
Future success in this market will depend on balancing traditional pharmaceutical packaging requirements with emerging trends including sustainability, digitalization, and patient-centricity. Companies that successfully integrate these elements while maintaining quality standards and regulatory compliance will capture market share and achieve sustainable growth in the expanding European pharmaceutical plastic bottles market.
What is Pharmaceutical Plastic Bottles?
Pharmaceutical plastic bottles are containers made from various types of plastics used to store and dispense medications and other pharmaceutical products. They are designed to ensure the safety, integrity, and efficacy of the contents while providing convenience for users.
What are the key players in the Europe Pharmaceutical Plastic Bottles Market?
Key players in the Europe Pharmaceutical Plastic Bottles Market include companies like Amcor plc, Gerresheimer AG, and Berry Global, Inc. These companies are known for their innovative packaging solutions and extensive product portfolios, among others.
What are the growth factors driving the Europe Pharmaceutical Plastic Bottles Market?
The growth of the Europe Pharmaceutical Plastic Bottles Market is driven by the increasing demand for pharmaceutical products, the rise in chronic diseases, and the need for convenient packaging solutions. Additionally, advancements in plastic materials and manufacturing processes contribute to market expansion.
What challenges does the Europe Pharmaceutical Plastic Bottles Market face?
The Europe Pharmaceutical Plastic Bottles Market faces challenges such as stringent regulations regarding plastic use and recycling, competition from alternative packaging materials, and the environmental impact of plastic waste. These factors can hinder market growth and innovation.
What opportunities exist in the Europe Pharmaceutical Plastic Bottles Market?
Opportunities in the Europe Pharmaceutical Plastic Bottles Market include the development of sustainable packaging solutions, the integration of smart packaging technologies, and the expansion of e-commerce in the pharmaceutical sector. These trends can enhance product offerings and market reach.
What trends are shaping the Europe Pharmaceutical Plastic Bottles Market?
Trends shaping the Europe Pharmaceutical Plastic Bottles Market include the increasing focus on sustainability, the adoption of child-resistant and tamper-evident designs, and the use of advanced materials for better barrier properties. These trends aim to improve safety and environmental impact.
Europe Pharmaceutical Plastic Bottles Market
| Segmentation Details | Description |
|---|---|
| Product Type | HDPE, PET, PVC, LDPE |
| End User | Pharmaceutical Companies, Contract Manufacturers, Research Laboratories, Hospitals |
| Packaging Type | Dropper Bottles, Spray Bottles, Pill Bottles, Vials |
| Grade | Medical Grade, Food Grade, Industrial Grade, Others |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading companies in the Europe Pharmaceutical Plastic Bottles Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at