444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The non-invasive brain trauma monitoring devices market refers to the market for medical devices that are used to monitor brain function and detect any abnormalities or injuries without the need for invasive procedures. These devices play a crucial role in the diagnosis and treatment of brain trauma, allowing healthcare professionals to assess the severity of injuries, monitor patients’ progress, and make informed decisions about their care. The market for non-invasive brain trauma monitoring devices has witnessed significant growth in recent years, driven by advancements in technology, increasing awareness about brain trauma, and the growing prevalence of traumatic brain injuries (TBIs) worldwide.
Meaning
Non-invasive brain trauma monitoring devices are medical instruments designed to assess brain function and detect any signs of trauma or injury without the need for invasive procedures such as surgery or the insertion of probes into the brain. These devices utilize various techniques, including electroencephalography (EEG), near-infrared spectroscopy (NIRS), transcranial Doppler (TCD), and magnetic resonance imaging (MRI), to measure and analyze brain activity, blood flow, and oxygen levels. By providing real-time data on brain function, these devices enable healthcare professionals to monitor patients’ neurological status and detect any abnormalities or changes that may indicate brain trauma or injury.
Executive Summary
The non-invasive brain trauma monitoring devices market is experiencing significant growth due to several factors, including technological advancements, increasing awareness about brain trauma, and the rising incidence of traumatic brain injuries. The market is characterized by the presence of both established players and emerging companies that are focusing on developing innovative devices with enhanced features and capabilities. Key market players are also investing in research and development activities to introduce advanced monitoring solutions, thereby driving market growth. The market is expected to continue its upward trajectory in the coming years, presenting lucrative opportunities for industry participants and stakeholders.
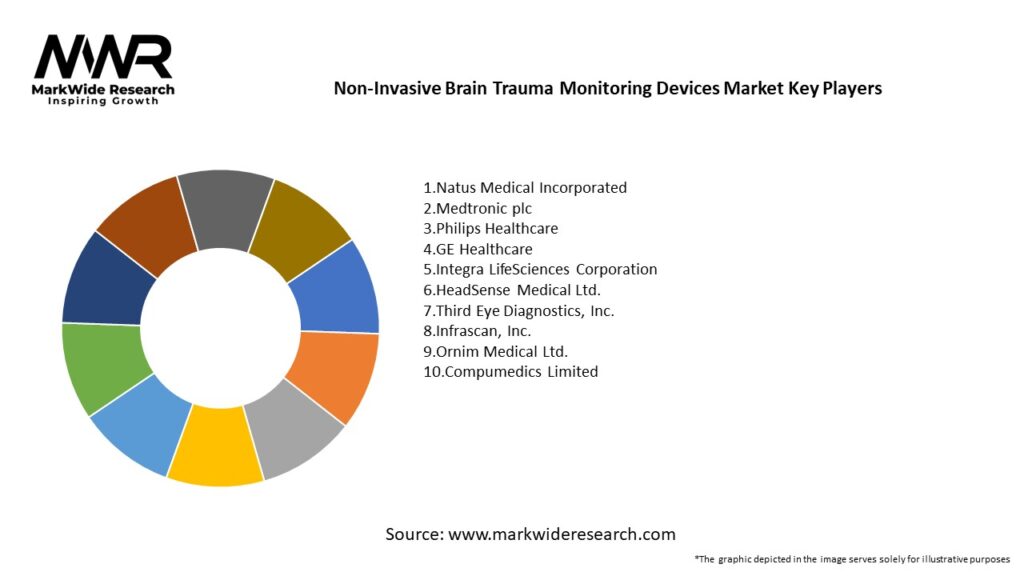
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
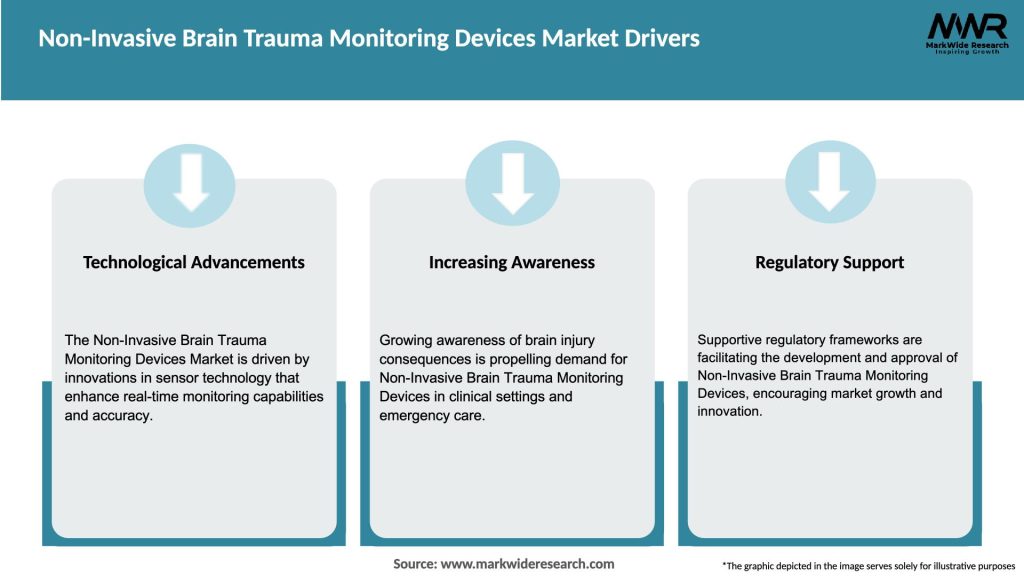
Market Drivers
The non-invasive brain trauma monitoring devices market is driven by several key factors:
Market Restraints
Despite the positive market drivers, certain factors act as restraints for the non-invasive brain trauma monitoring devices market:
Market Opportunities
The non-invasive brain trauma monitoring devices market presents several opportunities for growth and expansion:
Market Dynamics
The non-invasive brain trauma monitoring devices market is influenced by various dynamic factors:
Regional Analysis
The non-invasive brain trauma monitoring devices market can be analyzed based on regional segmentation:
Competitive Landscape
Leading companies in the Non-Invasive Brain Trauma Monitoring Devices Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
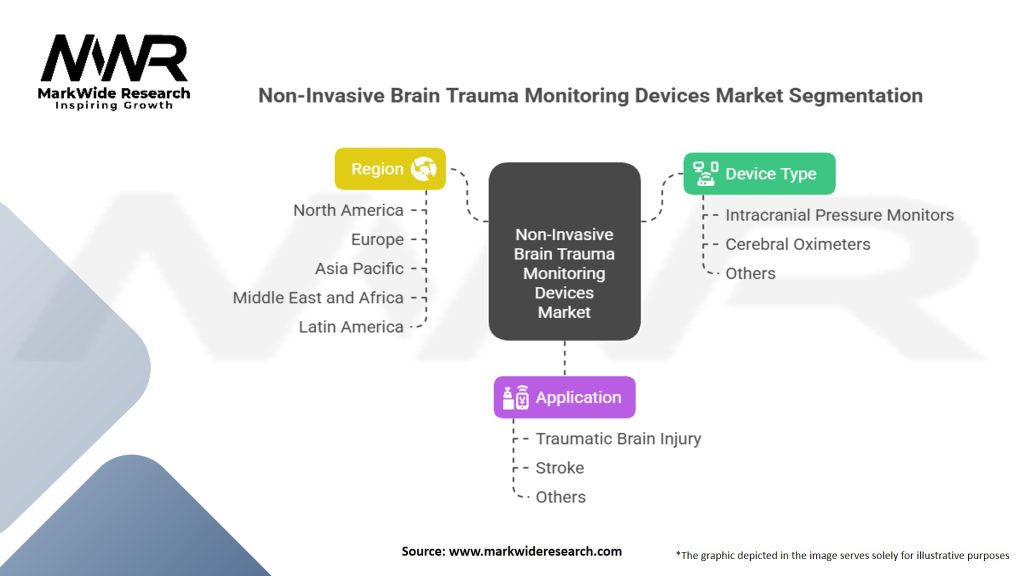
Segmentation
The non-invasive brain trauma monitoring devices market can be segmented based on various factors, including:
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
The non-invasive brain trauma monitoring devices market offers several benefits for industry participants and stakeholders:
SWOT Analysis
A SWOT analysis of the non-invasive brain trauma monitoring devices market reveals the following:
Market Key Trends
The non-invasive brain trauma monitoring devices market is influenced by several key trends:
Covid-19 Impact
The COVID-19 pandemic has had a significant impact on the non-invasive brain trauma monitoring devices market. The pandemic led to an increased focus on critical care and monitoring, including the monitoring of brain function in severe cases. Healthcare providers recognized the importance of early detection and monitoring of neurological complications associated with COVID-19, which resulted in a surge in demand for non-invasive brain trauma monitoring devices.
Additionally, the pandemic accelerated the adoption of remote patient monitoring solutions, including non-invasive brain trauma monitoring devices. These devices enabled healthcare professionals to remotely monitor and assess patients’ neurological status, reducing the need for in-person consultations and minimizing the risk of viral transmission.
However, the pandemic also posed challenges for the market, including disruptions in the global supply chain, temporary closure of manufacturing facilities, and reduced healthcare budgets in some regions. These factors impacted the production and distribution of non-invasive brain trauma monitoring devices and slowed down market growth temporarily.
Key Industry Developments
The non-invasive brain trauma monitoring devices market has witnessed several key industry developments:
Analyst Suggestions
Based on the analysis of the non-invasive brain trauma monitoring devices market, analysts suggest the following:
Future Outlook
The future of the non-invasive brain trauma monitoring devices market appears promising, with sustained growth expected. Technological advancements, increasing awareness about brain trauma, and a rising incidence of traumatic brain injuries are key drivers of market expansion. The market is likely to witness the introduction of more advanced monitoring devices with enhanced features and capabilities.
The adoption of non-invasive brain trauma monitoring devices is expected to increase, driven by the growing demand for personalized medicine, the focus on home healthcare, and the integration of AI and machine learning technologies. Emerging markets, including Asia Pacific and Latin America, present significant opportunities for market players to expand their market presence.
However, challenges such as the high cost of devices and the lack of skilled professionals to interpret data may impede market growth. Addressing these challenges and ensuring affordability, accessibility, and user-friendliness of non-invasive brain trauma monitoring devices will be crucial for market success.
Conclusion
The non-invasive brain trauma monitoring devices market is experiencing significant growth driven by technological advancements, increasing awareness about brain trauma, and a rising incidence of traumatic brain injuries. These devices play a vital role in the diagnosis, monitoring, and treatment of brain trauma without the need for invasive procedures. The market presents opportunities for industry participants to develop innovative solutions, expand market presence in emerging economies, and focus on personalized medicine and home healthcare.
While challenges such as high costs and the lack of skilled professionals exist, collaborations, research and development investments, and regulatory approvals contribute to market growth. The future outlook for the non-invasive brain trauma monitoring devices market is promising, with continued advancements in technology and increasing adoption of these devices expected to drive market expansion.
What are Non-Invasive Brain Trauma Monitoring Devices?
Non-Invasive Brain Trauma Monitoring Devices are medical tools designed to assess and monitor brain injuries without the need for surgical procedures. These devices utilize various technologies to provide real-time data on brain activity and health, making them essential in emergency and critical care settings.
Which companies are leading the Non-Invasive Brain Trauma Monitoring Devices Market?
Leading companies in the Non-Invasive Brain Trauma Monitoring Devices Market include Medtronic, Natus Medical Incorporated, and NeuroWave Systems, among others. These companies are known for their innovative solutions and contributions to brain monitoring technologies.
What are the key drivers of the Non-Invasive Brain Trauma Monitoring Devices Market?
Key drivers of the Non-Invasive Brain Trauma Monitoring Devices Market include the increasing prevalence of traumatic brain injuries, advancements in monitoring technologies, and the growing demand for non-invasive diagnostic methods in healthcare settings.
What challenges does the Non-Invasive Brain Trauma Monitoring Devices Market face?
The Non-Invasive Brain Trauma Monitoring Devices Market faces challenges such as high costs of advanced monitoring technologies, regulatory hurdles for new devices, and the need for extensive clinical validation to ensure accuracy and reliability.
What opportunities exist in the Non-Invasive Brain Trauma Monitoring Devices Market?
Opportunities in the Non-Invasive Brain Trauma Monitoring Devices Market include the development of portable monitoring solutions, integration of artificial intelligence for enhanced data analysis, and expansion into emerging markets with increasing healthcare investments.
What trends are shaping the Non-Invasive Brain Trauma Monitoring Devices Market?
Trends shaping the Non-Invasive Brain Trauma Monitoring Devices Market include the rise of telemedicine, increased focus on patient-centered care, and the adoption of wearable technologies that allow for continuous monitoring of brain health.
Non-Invasive Brain Trauma Monitoring Devices Market
| Segmentation | Details |
|---|---|
| Device Type | Intracranial Pressure Monitors, Cerebral Oximeters, Others |
| Application | Traumatic Brain Injury, Stroke, Others |
| Region | North America, Europe, Asia Pacific, Middle East and Africa, Latin America |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading companies in the Non-Invasive Brain Trauma Monitoring Devices Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at