444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The Calcitonin Receptor (CTR) ELISA Kit market is experiencing significant growth owing to the increasing demand for precise and reliable assays for the detection and quantification of calcitonin receptor levels in various biological samples. ELISA (Enzyme-Linked Immunosorbent Assay) kits offer a robust and cost-effective method for researchers and clinicians to study the role of CTR in various physiological and pathological processes.
Meaning
The Calcitonin Receptor (CTR) ELISA Kit enables the quantitative measurement of CTR levels in biological samples such as serum, plasma, and tissue lysates. This assay relies on the specific binding of CTR molecules to capture and detection antibodies conjugated with enzymes, allowing for the quantification of CTR concentration based on colorimetric or chemiluminescent signals.
Executive Summary
The Calcitonin Receptor (CTR) ELISA Kit market is characterized by technological advancements, expanding research applications, and strategic collaborations among key players to develop innovative and high-performance assay platforms. Market leaders are investing in R&D to enhance assay sensitivity, specificity, and automation, catering to the diverse needs of researchers and clinicians.

Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The Calcitonin Receptor (CTR) ELISA Kit market is influenced by various factors, including technological innovations, regulatory requirements, market competition, and shifting research priorities. Key dynamics shaping the market include:
Regional Analysis
The Calcitonin Receptor (CTR) ELISA Kit market is segmented into regions, including North America, Europe, Asia-Pacific, Latin America, and the Middle East & Africa. North America dominates the market, followed by Europe, owing to the presence of leading research institutions, academic centers, and pharmaceutical companies engaged in biomarker discovery and validation studies.
Competitive Landscape
Leading Companies in Calcitonin Receptor (CTR) ELISA Kit Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Segmentation
The market can be segmented based on assay type, detection method, sample type, and end-user. Assay types include quantitative ELISA kits for CTR detection and semi-quantitative kits for relative expression analysis. Detection methods encompass colorimetric, chemiluminescent, and fluorescence-based detection systems, while sample types include serum, plasma, tissue lysates, and cell culture supernatants. End-users range from academic research laboratories and biotechnology companies to clinical diagnostic laboratories and pharmaceutical manufacturers.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Market Key Trends
Covid-19 Impact
The Covid-19 pandemic has underscored the importance of biomarker-based diagnostics for disease surveillance, patient monitoring, and therapeutic development. While the immediate focus has been on SARS-CoV-2 diagnostics, the pandemic has accelerated broader trends towards personalized medicine and precision diagnostics, driving demand for CTR ELISA kits and other biomarker assays.
Key Industry Developments
Analyst Suggestions
Future Outlook
The future outlook for the Calcitonin Receptor (CTR) ELISA Kit market is promising, with sustained growth expected driven by increasing research funding, expanding applications in clinical diagnostics and drug development, and technological innovations in assay design and automation. As the importance of biomarker-based diagnostics continues to grow, CTR ELISA kits will remain indispensable tools for researchers and clinicians seeking to unravel the role of CTR in health and disease.
Conclusion
In conclusion, the Calcitonin Receptor (CTR) ELISA Kit market is poised for significant expansion and innovation, driven by the growing demand for biomarker-based diagnostics, personalized medicine approaches, and translational research applications. Despite challenges such as assay variability and competition from alternative technologies, ELISA remains a cornerstone of biomarker analysis, offering reliability, versatility, and cost-effectiveness for CTR detection and quantification. As the market evolves, collaboration, standardization, and regulatory compliance will be crucial in unlocking the full potential of CTR ELISA kits for advancing biomedical research and improving patient care.
What is Calcitonin Receptor (CTR) ELISA Kit?
The Calcitonin Receptor (CTR) ELISA Kit is a laboratory tool used to measure the levels of calcitonin receptors in various biological samples. It is commonly utilized in research and clinical settings to study bone metabolism and related disorders.
What are the key players in the Calcitonin Receptor (CTR) ELISA Kit Market?
Key players in the Calcitonin Receptor (CTR) ELISA Kit Market include companies like Thermo Fisher Scientific, Abcam, and R&D Systems, which are known for their innovative diagnostic solutions and high-quality assay kits, among others.
What are the growth factors driving the Calcitonin Receptor (CTR) ELISA Kit Market?
The growth of the Calcitonin Receptor (CTR) ELISA Kit Market is driven by increasing research activities in endocrinology, rising prevalence of bone-related diseases, and advancements in diagnostic technologies that enhance assay sensitivity and specificity.
What challenges does the Calcitonin Receptor (CTR) ELISA Kit Market face?
Challenges in the Calcitonin Receptor (CTR) ELISA Kit Market include the high cost of advanced kits, competition from alternative testing methods, and the need for skilled personnel to perform complex assays accurately.
What opportunities exist in the Calcitonin Receptor (CTR) ELISA Kit Market?
Opportunities in the Calcitonin Receptor (CTR) ELISA Kit Market include the development of novel biomarkers for bone diseases, expansion into emerging markets, and increasing collaborations between research institutions and diagnostic companies.
What trends are shaping the Calcitonin Receptor (CTR) ELISA Kit Market?
Trends in the Calcitonin Receptor (CTR) ELISA Kit Market include the integration of automation in laboratory processes, the rise of personalized medicine approaches, and the growing emphasis on rapid and accurate diagnostic solutions.
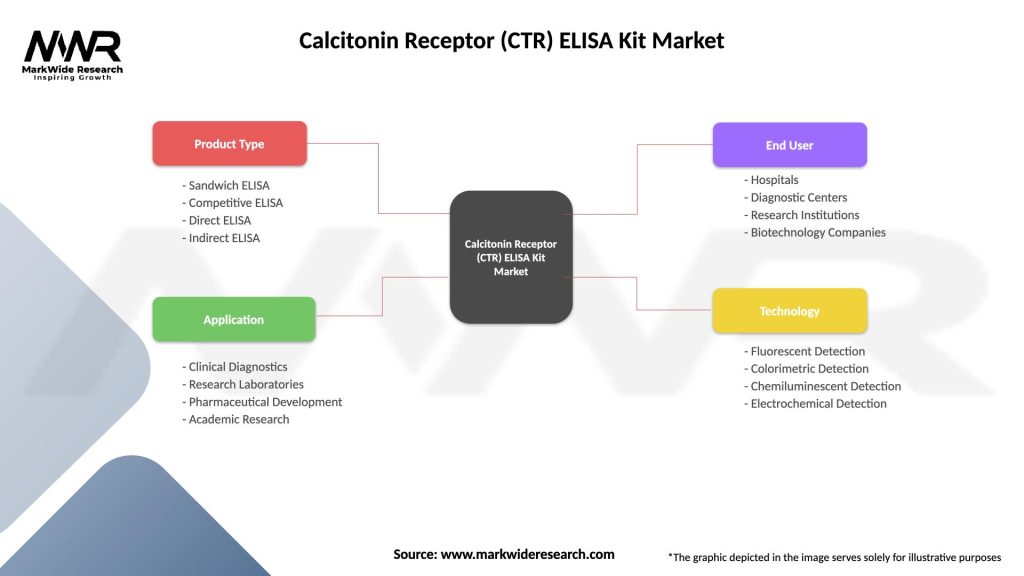
Calcitonin Receptor (CTR) ELISA Kit Market
| Segmentation Details | Description |
|---|---|
| Product Type | Sandwich ELISA, Competitive ELISA, Direct ELISA, Indirect ELISA |
| Application | Clinical Diagnostics, Research Laboratories, Pharmaceutical Development, Academic Research |
| End User | Hospitals, Diagnostic Centers, Research Institutions, Biotechnology Companies |
| Technology | Fluorescent Detection, Colorimetric Detection, Chemiluminescent Detection, Electrochemical Detection |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in Calcitonin Receptor (CTR) ELISA Kit Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at