444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The PD-1 and PD-L1 inhibitors market represents a burgeoning segment within the field of immuno-oncology, offering novel therapeutic options for the treatment of various cancers. These inhibitors target the programmed cell death protein 1 (PD-1) and its ligand (PD-L1), thereby unleashing the body’s immune system to recognize and attack cancer cells. The market is characterized by extensive research and development efforts, rapid clinical advancements, and a growing portfolio of approved drugs across different cancer indications.
Meaning
PD-1 and PD-L1 inhibitors are a class of immunotherapy drugs designed to modulate the immune response against cancer. By blocking the interaction between PD-1, expressed on the surface of activated T cells, and its ligand PD-L1, expressed on tumor cells and immune cells, these inhibitors restore antitumor immunity and enhance the body’s ability to recognize and eliminate cancer cells. This mechanism of action represents a paradigm shift in cancer treatment, offering durable responses and improved survival outcomes in various malignancies.
Executive Summary
The PD-1 and PD-L1 inhibitors market is witnessing exponential growth, driven by factors such as the increasing prevalence of cancer, the growing understanding of tumor immunology, and the success of immunotherapy in clinical practice. Key market players are focused on expanding the indications for existing drugs, developing novel combination therapies, and exploring predictive biomarkers to optimize patient selection and treatment outcomes.

Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The PD-1 and PD-L1 inhibitors market operates within a dynamic landscape shaped by factors such as scientific advancements, regulatory policies, market competition, and patient advocacy. Market players need to navigate through these dynamics, adapt their strategies, and capitalize on emerging opportunities to maintain a competitive edge and sustain growth in the evolving oncology landscape.
Regional Analysis
Competitive Landscape
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
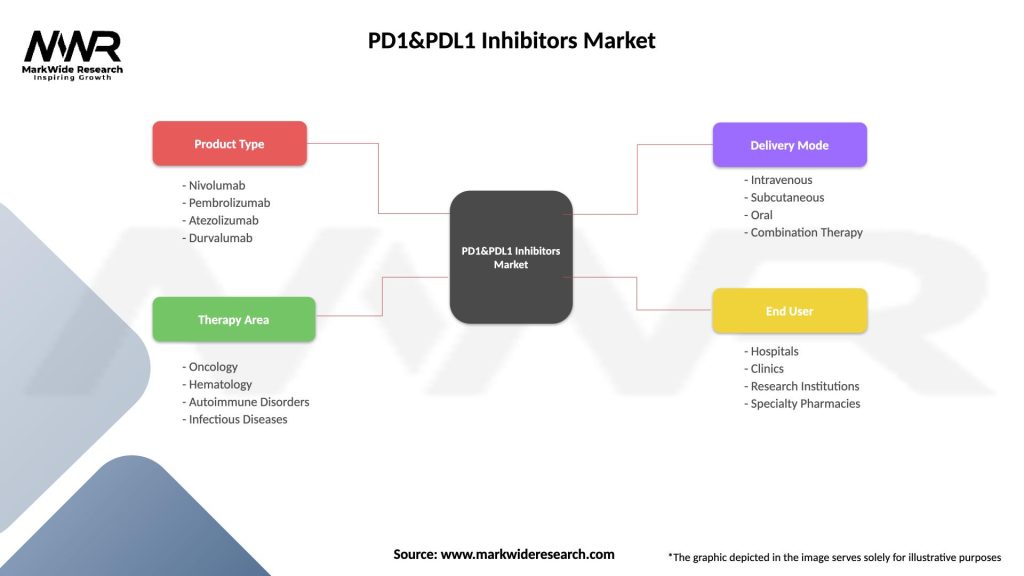
Segmentation
The PD-1 and PD-L1 inhibitors market can be segmented based on various factors, including:
Segmentation provides insights into market dynamics, treatment patterns, patient characteristics, and regional variations, enabling stakeholders to tailor their strategies and interventions accordingly.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
The PD-1 and PD-L1 inhibitors market offer several benefits for industry participants and stakeholders:
SWOT Analysis
Market Key Trends
Covid-19 Impact
The COVID-19 pandemic has had a significant impact on the PD-1 and PD-L1 inhibitors market, influencing trends such as telemedicine adoption, decentralized clinical trials, drug development timelines, and patient access to oncology care. While the pandemic posed challenges in terms of disrupted supply chains, delayed clinical trials, and diverted healthcare resources, it also underscored the importance of innovation, collaboration, and resilience in advancing cancer research and patient care.
Key Industry Developments
Analyst Suggestions
Future Outlook
The PD-1 and PD-L1 inhibitors market are poised for continued growth and innovation, driven by advancements in precision medicine, combination therapies, biomarker-driven strategies, and adjuvant/neoadjuvant approaches. Key trends such as biomarker integration, collaborative research, health economics assessment, and patient-centered care will shape the future landscape of cancer immunotherapy, improving treatment outcomes and quality of life for patients with various malignancies.
Conclusion
PD-1 and PD-L1 inhibitors represent a transformative approach to cancer treatment, harnessing the body’s immune system to recognize and eliminate cancer cells across multiple indications. The market’s rapid growth is fueled by scientific advancements, clinical successes, and patient-centered innovation. Despite challenges such as resistance mechanisms, toxicity profiles, and healthcare disparities, the future outlook for PD-1 and PD-L1 inhibitors remains promising, driven by ongoing research, collaboration, and commitment to improving cancer care globally.
What is PD1&PDL1 Inhibitors?
PD1&PDL1 inhibitors are a class of immunotherapy drugs that block the programmed cell death protein 1 (PD-1) and its ligand (PD-L1), enhancing the immune system’s ability to fight cancer. These inhibitors are primarily used in the treatment of various cancers, including melanoma, lung cancer, and bladder cancer.
What are the key companies in the PD1&PDL1 Inhibitors Market?
Key companies in the PD1&PDL1 inhibitors market include Bristol-Myers Squibb, Merck & Co., and AstraZeneca, which are known for their leading products in this therapeutic area. These companies are actively involved in research and development to expand the applications of PD1&PDL1 inhibitors, among others.
What are the growth factors driving the PD1&PDL1 Inhibitors Market?
The growth of the PD1&PDL1 inhibitors market is driven by the increasing prevalence of cancer, advancements in immunotherapy, and the rising demand for targeted therapies. Additionally, ongoing clinical trials and research are expanding the potential applications of these inhibitors in various cancer types.
What challenges does the PD1&PDL1 Inhibitors Market face?
The PD1&PDL1 inhibitors market faces challenges such as high treatment costs, potential side effects, and competition from alternative therapies. Furthermore, regulatory hurdles and the need for personalized treatment approaches can also impact market growth.
What opportunities exist in the PD1&PDL1 Inhibitors Market?
Opportunities in the PD1&PDL1 inhibitors market include the development of combination therapies, expansion into new cancer indications, and advancements in biomarker research. These factors can enhance treatment efficacy and patient outcomes, driving market growth.
What are the current trends in the PD1&PDL1 Inhibitors Market?
Current trends in the PD1&PDL1 inhibitors market include the increasing focus on personalized medicine, the exploration of novel drug combinations, and the integration of artificial intelligence in drug development. These trends aim to improve treatment strategies and patient care in oncology.
PD1&PDL1 Inhibitors Market
| Segmentation Details | Description |
|---|---|
| Product Type | Nivolumab, Pembrolizumab, Atezolizumab, Durvalumab |
| Therapy Area | Oncology, Hematology, Autoimmune Disorders, Infectious Diseases |
| Delivery Mode | Intravenous, Subcutaneous, Oral, Combination Therapy |
| End User | Hospitals, Clinics, Research Institutions, Specialty Pharmacies |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at