444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The insufflation tubings market is an integral segment of the medical device industry, catering to the needs of various medical procedures requiring insufflation of gases or fluids into the body cavities. These tubings are essential components used in minimally invasive surgeries, endoscopic procedures, laparoscopic surgeries, and gastrointestinal interventions. The market for insufflation tubings is driven by the increasing prevalence of chronic diseases, the growing adoption of minimally invasive surgical techniques, and technological advancements in medical devices.
Meaning
Insufflation tubings are medical-grade tubes designed to deliver gases or fluids into the body during surgical or diagnostic procedures. These tubings are typically made from flexible, biocompatible materials such as silicone or polyurethane and are available in various lengths and diameters to suit different clinical applications. They are used in conjunction with insufflators and other medical devices to maintain a stable pneumoperitoneum or pneumoretroperitoneum, facilitate visualization, and create a working space during minimally invasive surgeries.
Executive Summary
The insufflation tubings market is witnessing steady growth due to the increasing adoption of minimally invasive surgical procedures, technological advancements in medical devices, and the rising prevalence of chronic diseases requiring surgical intervention. Key market players are focusing on product innovation, strategic collaborations, and geographic expansion to gain a competitive edge and capitalize on emerging opportunities in the global healthcare landscape.

Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The insufflation tubings market is characterized by dynamic factors influencing supply and demand dynamics, including technological advancements, regulatory trends, competitive pressures, and evolving healthcare delivery models. Understanding these dynamics is essential for market participants to navigate challenges, capitalize on opportunities, and drive sustainable growth in the global healthcare landscape.
Regional Analysis
The insufflation tubings market exhibits regional variations in terms of market size, growth rates, regulatory frameworks, healthcare infrastructure, and clinical practices. Key regions driving market growth include North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa, each with unique market dynamics and growth drivers.
Competitive Landscape
Leading Companies in Insufflation Tubings Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
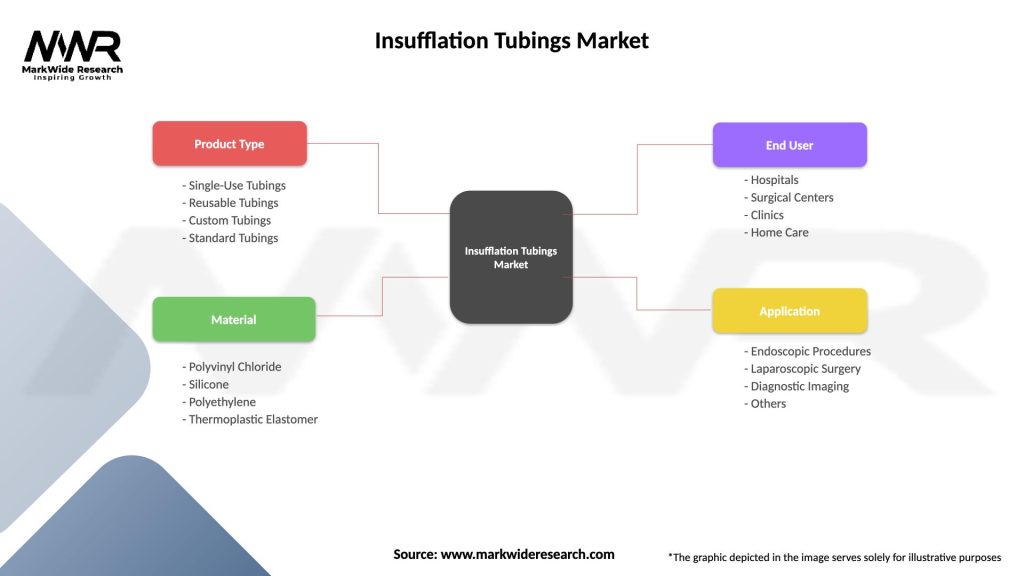
Segmentation
The insufflation tubings market can be segmented based on various parameters, including product type, material composition, application, end-user, and geography. Product types may include disposable and reusable insufflation tubings, while materials may include silicone, polyurethane, or other biocompatible polymers. Applications may encompass laparoscopic surgery, endoscopic procedures, gastrointestinal interventions, and urological surgeries, among others. End-users may include hospitals, ambulatory surgical centers, specialty clinics, and diagnostic laboratories.
Category-wise Insights
Insufflation tubings represent a critical category of medical devices used in minimally invasive surgical procedures, offering benefits such as improved patient outcomes, reduced postoperative complications, and enhanced procedural efficiency. Key insights into this category include the following:
Key Benefits for Industry Participants and Stakeholders
The insufflation tubings market offers several benefits for industry participants and stakeholders:
SWOT Analysis
A SWOT analysis provides insights into the strengths, weaknesses, opportunities, and threats facing the insufflation tubings market:
Market Key Trends
Key trends shaping the insufflation tubings market include:
Covid-19 Impact
The Covid-19 pandemic has had a multifaceted impact on the insufflation tubings market:
Key Industry Developments
Recent developments in the insufflation tubings market include:
Analyst Suggestions
Key recommendations for stakeholders in the insufflation tubings market include:
Future Outlook
The future outlook for the insufflation tubings market is characterized by several trends and projections that are expected to shape the industry landscape in the coming years. These include advancements in technology, evolving healthcare practices, regulatory developments, market dynamics, and the impact of global factors such as demographic shifts, economic trends, and healthcare infrastructure investments. Here’s an overview of the future outlook for the insufflation tubings market:
Technological Advancements and Innovations: Continued advancements in materials science, manufacturing processes, and design engineering are expected to drive innovation in insufflation tubing technology. This includes the development of next-generation materials with enhanced biocompatibility, flexibility, and durability, as well as the integration of smart features such as sensors, connectivity, and data analytics for real-time monitoring and feedback during surgical procedures. Advanced imaging techniques, augmented reality, and artificial intelligence may also play a role in enhancing visualization, navigation, and precision in minimally invasive surgeries, further driving demand for specialized insufflation tubings tailored to specific clinical applications and procedural requirements.
Shift Towards Value-Based Healthcare: The transition towards value-based healthcare models focused on patient outcomes, cost-effectiveness, and quality of care is expected to influence procurement practices, reimbursement policies, and purchasing decisions for medical devices, including insufflation tubings. Healthcare providers and payers will prioritize solutions that demonstrate clinical efficacy, safety, and economic value in terms of reduced hospitalization costs, shorter recovery times, and improved patient satisfaction. This trend may drive demand for innovative insufflation tubing solutions that offer superior performance, reliability, and usability while optimizing resource utilization and healthcare expenditures.
Global Market Expansion and Penetration: The insufflation tubings market is poised for global expansion and penetration into emerging markets, underserved regions, and niche clinical specialties. Rapid urbanization, rising healthcare expenditures, and increasing access to healthcare services in developing countries present opportunities for market players to tap into growing patient populations and unmet medical needs. Strategic partnerships, distribution networks, and market entry strategies tailored to local preferences, regulatory requirements, and healthcare infrastructure will be essential for success in these diverse and dynamic markets.
Regulatory Landscape and Compliance Requirements: Regulatory oversight and compliance requirements for medical devices, including insufflation tubings, are expected to evolve in response to emerging technologies, safety concerns, and market dynamics. Regulatory agencies will continue to emphasize patient safety, product quality, and post-market surveillance through stringent standards, guidelines, and enforcement mechanisms. Market players will need to invest in regulatory compliance initiatives, quality management systems, and risk mitigation strategies to ensure compliance with evolving regulatory requirements and maintain market access across different regions and jurisdictions.
Patient-Centric Care and Personalized Medicine: The growing emphasis on patient-centric care, personalized medicine, and minimally invasive interventions will drive demand for insufflation tubings that offer tailored solutions for individual patient needs, preferences, and clinical conditions. Customized insufflation tubing designs, patient-specific parameters, and procedural protocols may become increasingly prevalent as healthcare providers seek to optimize surgical outcomes, minimize patient discomfort, and enhance overall patient experience. This trend aligns with broader healthcare trends towards precision medicine, digital health, and patient empowerment, driving demand for innovative medical devices that enable personalized and proactive approaches to healthcare delivery.
Conclusion
The insufflation tubings market plays a critical role in supporting minimally invasive surgical procedures, endoscopic interventions, and diagnostic techniques by facilitating the delivery of gases or fluids into the body cavities. Key market drivers include the rising demand for minimally invasive surgeries, technological advancements in medical devices, and the increasing prevalence of chronic diseases requiring surgical intervention. Despite challenges such as pricing pressures, regulatory complexities, and competitive dynamics, the market presents opportunities for innovation, expansion, and collaboration to meet the evolving needs of healthcare providers and patients worldwide. By focusing on product innovation, regulatory compliance, strategic partnerships, and customer education, stakeholders can navigate market complexities, capitalize on emerging trends, and drive sustainable growth in the global insufflation tubings market.
What is Insufflation Tubings?
Insufflation tubings are specialized medical devices used to deliver gas, typically carbon dioxide, into a body cavity during minimally invasive surgical procedures. They play a crucial role in creating space for surgeons to operate effectively.
What are the key players in the Insufflation Tubings Market?
Key players in the Insufflation Tubings Market include companies like Medtronic, Boston Scientific, and Olympus Corporation, which are known for their innovative medical devices and technologies in the surgical field, among others.
What are the growth factors driving the Insufflation Tubings Market?
The Insufflation Tubings Market is driven by the increasing demand for minimally invasive surgeries, advancements in surgical technologies, and a growing aging population requiring surgical interventions. These factors contribute to the rising adoption of insufflation tubings in various medical procedures.
What challenges does the Insufflation Tubings Market face?
Challenges in the Insufflation Tubings Market include stringent regulatory requirements, potential complications associated with insufflation procedures, and the high cost of advanced surgical equipment. These factors can hinder market growth and adoption.
What opportunities exist in the Insufflation Tubings Market?
Opportunities in the Insufflation Tubings Market include the development of innovative materials for tubings, expansion into emerging markets, and increasing investments in healthcare infrastructure. These trends can enhance the market landscape and drive growth.
What trends are shaping the Insufflation Tubings Market?
Current trends in the Insufflation Tubings Market include the integration of smart technologies in surgical devices, a focus on patient safety and comfort, and the rise of robotic-assisted surgeries. These innovations are transforming the way insufflation tubings are utilized in medical procedures.
Insufflation Tubings Market
| Segmentation Details | Description |
|---|---|
| Product Type | Single-Use Tubings, Reusable Tubings, Custom Tubings, Standard Tubings |
| Material | Polyvinyl Chloride, Silicone, Polyethylene, Thermoplastic Elastomer |
| End User | Hospitals, Surgical Centers, Clinics, Home Care |
| Application | Endoscopic Procedures, Laparoscopic Surgery, Diagnostic Imaging, Others |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in Insufflation Tubings Market
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at