444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The Spinal Non-Fusion Technologies market is a rapidly evolving segment within the medical device industry that focuses on providing innovative solutions for spinal disorders without the need for traditional fusion procedures. This market is witnessing significant growth due to the rising prevalence of spinal disorders, advancements in medical technology, and increasing demand for minimally invasive surgical procedures. Spinal non-fusion technologies aim to offer patients improved mobility, reduced post-operative complications, shorter hospital stays, and faster recovery times, making them an attractive option for both patients and healthcare providers.
Meaning
Spinal non-fusion technologies refer to a range of medical devices and treatment approaches designed to address spinal disorders without resorting to fusion procedures. Unlike traditional fusion surgeries that aim to stabilize the spine by fusing two or more vertebrae together, non-fusion technologies employ various techniques such as artificial disc replacement, dynamic stabilization, and interspinous process devices to maintain spinal stability while preserving the natural motion of the spine. These technologies offer a less invasive alternative to fusion procedures and cater to a broader spectrum of patients with different spinal conditions.
Executive Summary
The Spinal Non-Fusion Technologies market has experienced substantial growth in recent years, driven by factors such as an aging population, increasing incidence of spinal disorders, and technological advancements in the field of medical devices. Non-fusion technologies have gained popularity among patients and surgeons alike due to their potential to improve patient outcomes and enhance quality of life. This report provides a comprehensive analysis of the market, including key insights, market drivers, restraints, opportunities, regional analysis, competitive landscape, and future outlook.

Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
The Spinal Non-Fusion Technologies Market is characterized by several critical trends and insights that underscore its dynamic growth:
These insights reflect the market’s potential to transform spinal care by offering solutions that are both clinically effective and aligned with patient expectations for less invasive, motion-preserving treatments.
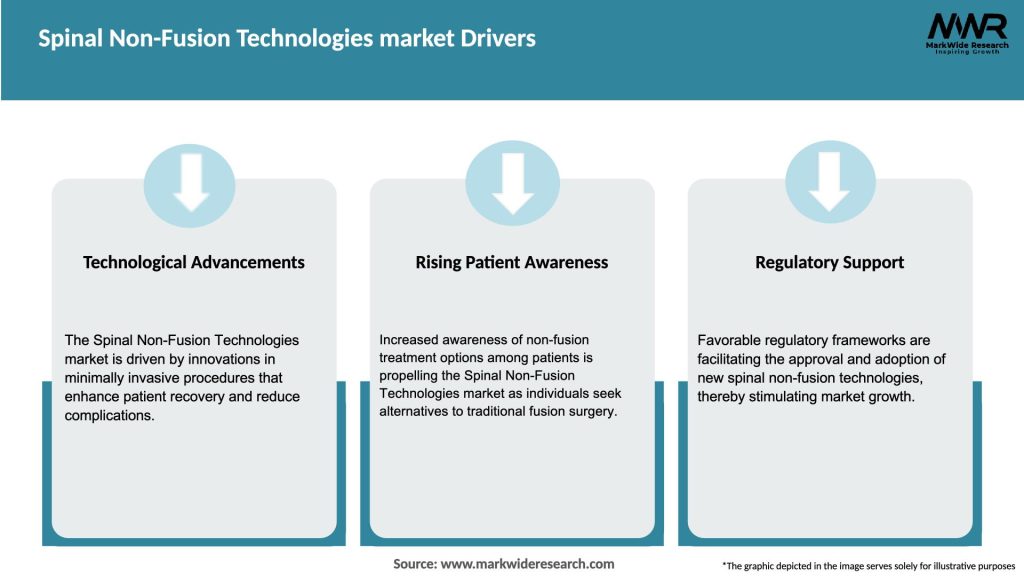
Market Drivers
Several key factors are propelling the growth of the Spinal Non-Fusion Technologies Market:
These drivers are collectively creating a favorable environment for the adoption of spinal non-fusion technologies across diverse healthcare settings.
Market Restraints
Despite the promising outlook, the Spinal Non-Fusion Technologies Market faces several challenges:
These restraints highlight the need for ongoing innovation, enhanced clinical validation, and supportive regulatory and reimbursement frameworks to fully realize the market’s potential.
Market Opportunities
The Spinal Non-Fusion Technologies Market presents several opportunities that can drive future growth and innovation:
Capitalizing on these opportunities will require strategic collaboration between device manufacturers, healthcare providers, and research institutions to drive innovation and adoption.

Market Dynamics
The dynamics of the Spinal Non-Fusion Technologies Market are shaped by a mix of supply-side and demand-side factors, as well as broader economic influences:
Supply Side Factors:
Demand Side Factors:
Economic and Regulatory Factors:
These dynamics underscore the importance of a balanced approach that addresses technological innovation, clinical adoption, and economic viability in driving market success.
Regional Analysis
The Spinal Non-Fusion Technologies Market exhibits varying levels of adoption and growth potential across different regions:
North America:
North America, particularly the United States, remains a leader in the adoption of advanced spinal technologies. High healthcare spending, strong clinical research infrastructure, and favorable reimbursement policies contribute to robust market growth. The region is also a hub for innovation, with many leading companies and research institutions based here.
Europe:
Europe is experiencing steady growth in the spinal non-fusion market due to increasing awareness of minimally invasive procedures, government support for technological innovation, and a well-established healthcare system. Countries such as Germany, the United Kingdom, and France are at the forefront of clinical adoption, driven by both public and private healthcare investments.
Asia-Pacific:
The Asia-Pacific region is emerging as a significant growth market, fueled by rising incomes, expanding healthcare infrastructure, and an increasing prevalence of degenerative spinal disorders. Countries like China, Japan, South Korea, and India are witnessing growing demand for advanced spine care technologies, despite varying reimbursement environments.
Latin America:
In Latin America, the market is in its early stages of development. However, growing investments in healthcare and rising awareness of minimally invasive spine treatments present considerable opportunities for market expansion in countries such as Brazil, Mexico, and Argentina.
Middle East and Africa:
The Middle East and Africa represent untapped markets for spinal non-fusion technologies. Although the adoption rate is currently lower due to limited healthcare infrastructure and budget constraints, ongoing investments in modernizing healthcare facilities and increasing demand for advanced surgical procedures are expected to drive future growth.
Competitive Landscape
Leading Companies in the Spinal Non-Fusion Technologies Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Segmentation
The Spinal Non-Fusion Technologies Market can be segmented based on product type, indication, and end-user, offering a comprehensive view of market structure:
By Product Type:
By Indication:
By End-User:
Category-wise Insights
Each segment within the Spinal Non-Fusion Technologies Market offers unique benefits and challenges:
Key Benefits for Industry Participants and Stakeholders
The adoption of spinal non-fusion technologies offers numerous benefits for manufacturers, healthcare providers, and patients alike:
SWOT Analysis
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Several key trends are shaping the Spinal Non-Fusion Technologies Market:
Covid-19 Impact
The Covid-19 pandemic has exerted a multifaceted impact on the Spinal Non-Fusion Technologies Market:
Key Industry Developments
The Spinal Non-Fusion Technologies Market has seen a number of significant developments that are influencing its evolution:
Analyst Suggestions
Based on current market dynamics and emerging trends, industry analysts offer the following recommendations for stakeholders in the Spinal Non-Fusion Technologies Market:
Future Outlook
The future of the Spinal Non-Fusion Technologies Market appears promising, driven by a confluence of technological, clinical, and demographic factors. As the demand for minimally invasive, motion-preserving spine treatments continues to rise, the market is poised for sustained growth. Key factors that will shape the future include:
While challenges such as reimbursement complexities and high initial costs remain, companies that invest in continuous innovation, robust clinical validation, and strategic market expansion are well-positioned to capture significant market share in the coming years.
Conclusion
The Spinal Non-Fusion Technologies Market is transforming the landscape of spine care by offering innovative, minimally invasive alternatives to traditional fusion surgeries. Driven by technological advancements, changing clinical practices, and the increasing prevalence of degenerative spinal conditions, the market is set for robust growth. These advanced technologies not only preserve natural spinal motion but also provide enhanced patient outcomes, reduced recovery times, and fewer long-term complications.
What is Spinal Non-Fusion Technologies?
Spinal Non-Fusion Technologies refer to innovative medical devices and procedures designed to treat spinal disorders without the need for traditional fusion surgery. These technologies include dynamic stabilization systems, interspinous spacers, and motion-preserving implants that aim to alleviate pain and restore function while maintaining spinal mobility.
What are the key players in the Spinal Non-Fusion Technologies market?
Key players in the Spinal Non-Fusion Technologies market include Medtronic, NuVasive, and Zimmer Biomet, which are known for their advanced spinal solutions. These companies focus on developing innovative products that enhance patient outcomes and reduce recovery times, among others.
What are the growth factors driving the Spinal Non-Fusion Technologies market?
The Spinal Non-Fusion Technologies market is driven by factors such as the increasing prevalence of spinal disorders, the rising demand for minimally invasive surgical procedures, and advancements in technology that improve surgical outcomes. Additionally, the aging population contributes to the growing need for effective spinal treatments.
What challenges does the Spinal Non-Fusion Technologies market face?
Challenges in the Spinal Non-Fusion Technologies market include regulatory hurdles, high costs associated with advanced technologies, and the need for extensive clinical data to support new product approvals. Additionally, competition from traditional fusion methods can hinder market growth.
What opportunities exist in the Spinal Non-Fusion Technologies market?
Opportunities in the Spinal Non-Fusion Technologies market include the development of new materials and designs that enhance device performance, as well as expanding applications in various spinal conditions. Furthermore, increasing awareness and acceptance of non-fusion options among healthcare providers and patients present significant growth potential.
What trends are shaping the Spinal Non-Fusion Technologies market?
Trends in the Spinal Non-Fusion Technologies market include the rise of personalized medicine, where treatments are tailored to individual patient needs, and the integration of robotics and AI in surgical procedures. Additionally, there is a growing focus on patient-centered care and improved recovery protocols.
Spinal Non-Fusion Technologies market
| Segmentation Details | Description |
|---|---|
| Product Type | Interspinous Devices, Dynamic Stabilization Systems, Artificial Discs, Bone Graft Substitutes |
| End User | Hospitals, Ambulatory Surgical Centers, Orthopedic Clinics, Rehabilitation Centers |
| Technology | Minimally Invasive Surgery, Robotic-Assisted Surgery, Biologics, Imaging Techniques |
| Application | Degenerative Disc Disease, Spinal Stenosis, Spondylolisthesis, Trauma |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Spinal Non-Fusion Technologies Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at