444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$2450
Market Overview
The Germany neonatal and prenatal devices market refers to the segment of the healthcare industry that focuses on medical devices and equipment used for the care and monitoring of newborn infants and pregnant women. These devices play a critical role in ensuring the well-being and health of both mothers and infants during pregnancy, childbirth, and the neonatal period. The market encompasses a wide range of devices, including fetal monitors, incubators, respiratory devices, phototherapy equipment, and neonatal intensive care units (NICUs). The Germany neonatal and prenatal devices market is driven by factors such as the increasing incidence of preterm births, advancements in medical technology, and the growing focus on improving maternal and neonatal healthcare outcomes.
Meaning
Neonatal and prenatal devices are medical devices and equipment specifically designed for the care and monitoring of newborn infants and pregnant women. These devices serve a crucial role in ensuring the safety and well-being of both mothers and infants during pregnancy, labor, and the neonatal period. They are used in various healthcare settings, including hospitals, birthing centers, and neonatal intensive care units (NICUs), to provide specialized care, monitoring, and treatment for high-risk pregnancies, preterm infants, and neonates with medical conditions.
Executive Summary
The Germany neonatal and prenatal devices market is witnessing significant growth, driven by factors such as the rising incidence of preterm births, increasing awareness about the importance of maternal and neonatal health, and advancements in medical technology. The market is characterized by the presence of both domestic and international manufacturers offering a wide range of devices and equipment for neonatal and prenatal care. Key trends include the integration of advanced technologies, such as artificial intelligence and telemedicine, and a focus on developing innovative, user-friendly, and cost-effective solutions to address the unique healthcare needs of mothers and infants.

Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The Germany neonatal and prenatal devices market is driven by several dynamics, including technological advancements, changing demographics, healthcare policies, and patient preferences. Key dynamics shaping the market include:
Regional Analysis
The neonatal and prenatal devices market in Germany exhibits regional variations based on factors such as population demographics, healthcare infrastructure, and economic development. Major cities and regions with advanced healthcare facilities, such as Berlin, Munich, and Hamburg, are likely to have higher market adoption and demand for neonatal and prenatal devices.
Competitive Landscape
Leading Companies in the Germany Neonatal and Prenatal Devices Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.

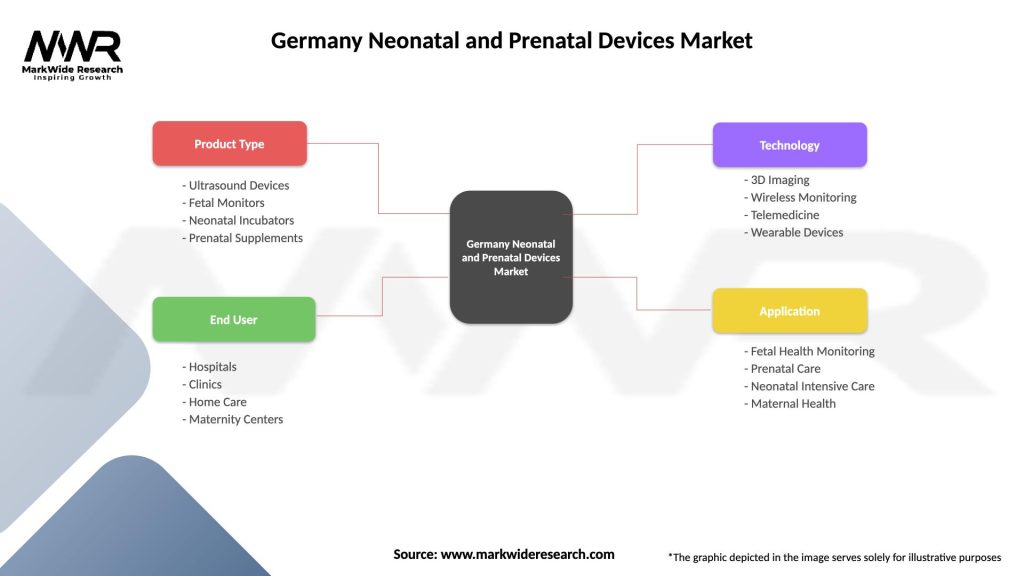
Segmentation
The Germany neonatal and prenatal devices market can be segmented based on various factors, including:
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Covid-19 Impact
The Covid-19 pandemic has had a significant impact on the neonatal and prenatal devices market in Germany. The healthcare system has faced unprecedented challenges, with a focus on managing the pandemic and ensuring the safety of patients and healthcare providers. The pandemic has led to disruptions in supply chains, reduced healthcare visits, and an increased emphasis on remote monitoring and telemedicine solutions. Healthcare facilities have implemented strict infection control measures to protect vulnerable populations, including pregnant women and newborn infants.
Key Industry Developments
Analyst Suggestions
Future Outlook
The future outlook for the Germany neonatal and prenatal devices market is positive, driven by ongoing advancements in medical technology, increasing awareness about the importance of maternal and neonatal health, and the growing emphasis on personalized care. The market is expected to witness continued innovation, with the integration of AI, IoT, and remote monitoring solutions. Collaboration between manufacturers, healthcare providers, and research institutions will play a crucial role in driving advancements and improving patient outcomes.
Conclusion
The Germany neonatal and prenatal devices market is experiencing significant growth, driven by factors such as the rising incidence of preterm births, technological advancements, and a focus on improving maternal and neonatal health outcomes. The market presents opportunities for manufacturers to develop innovative solutions, enhance patient experiences, and improve healthcare efficiency. However, challenges such as the high cost of devices and stringent regulatory requirements need to be addressed. With continued advancements, collaborations, and a patient-centered approach, the neonatal and prenatal devices market in Germany is poised for further advancements and positive developments in the coming years.
What is Neonatal and Prenatal Devices?
Neonatal and Prenatal Devices refer to medical equipment and technologies designed for the care of newborns and expectant mothers. These devices include incubators, fetal monitors, and ultrasound machines, which are essential for monitoring and ensuring the health of both mothers and infants.
What are the key players in the Germany Neonatal and Prenatal Devices Market?
Key players in the Germany Neonatal and Prenatal Devices Market include Siemens Healthineers, GE Healthcare, Philips Healthcare, and Drägerwerk AG, among others. These companies are known for their innovative technologies and comprehensive product offerings in neonatal and prenatal care.
What are the growth factors driving the Germany Neonatal and Prenatal Devices Market?
The growth of the Germany Neonatal and Prenatal Devices Market is driven by increasing birth rates, advancements in medical technology, and a growing focus on maternal and infant health. Additionally, rising awareness about prenatal care and neonatal health is contributing to market expansion.
What challenges does the Germany Neonatal and Prenatal Devices Market face?
The Germany Neonatal and Prenatal Devices Market faces challenges such as stringent regulatory requirements, high costs of advanced medical devices, and the need for continuous innovation. These factors can hinder market entry for new players and affect the overall growth of the sector.
What opportunities exist in the Germany Neonatal and Prenatal Devices Market?
Opportunities in the Germany Neonatal and Prenatal Devices Market include the development of telemedicine solutions, integration of artificial intelligence in monitoring devices, and increasing investments in healthcare infrastructure. These trends can enhance patient care and improve outcomes for mothers and infants.
What trends are shaping the Germany Neonatal and Prenatal Devices Market?
Trends shaping the Germany Neonatal and Prenatal Devices Market include the rise of portable and home-based monitoring devices, advancements in non-invasive diagnostic technologies, and a growing emphasis on personalized medicine. These innovations are transforming how neonatal and prenatal care is delivered.
Germany Neonatal and Prenatal Devices Market
| Segmentation Details | Description |
|---|---|
| Product Type | Ultrasound Devices, Fetal Monitors, Neonatal Incubators, Prenatal Supplements |
| End User | Hospitals, Clinics, Home Care, Maternity Centers |
| Technology | 3D Imaging, Wireless Monitoring, Telemedicine, Wearable Devices |
| Application | Fetal Health Monitoring, Prenatal Care, Neonatal Intensive Care, Maternal Health |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the Germany Neonatal and Prenatal Devices Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at