444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The Antivenom Vaccines market is a rapidly growing sector in the pharmaceutical industry. Antivenom vaccines are specifically developed to counteract the toxic effects of venomous bites or stings from various animals, such as snakes, spiders, scorpions, and other venomous creatures. These vaccines play a crucial role in preventing life-threatening complications associated with envenomation.
Meaning
Antivenom vaccines are biological preparations that contain antibodies derived from the serum of animals immunized with specific venom antigens. These vaccines are designed to neutralize the venom toxins upon administration to individuals who have been bitten or stung by venomous creatures. By providing passive immunity, antivenom vaccines help mitigate the harmful effects of venom, reduce morbidity and mortality rates, and enhance the chances of survival for patients.
Executive Summary
The Antivenom Vaccines market has witnessed significant growth in recent years, driven by the increasing incidence of venomous bites and stings globally. This growth can be attributed to the rising awareness about the importance of antivenom treatment, advancements in biotechnology, and government initiatives aimed at controlling venomous creatures. The market is expected to continue its upward trajectory, fueled by technological advancements, research and development activities, and increasing investment in healthcare infrastructure.
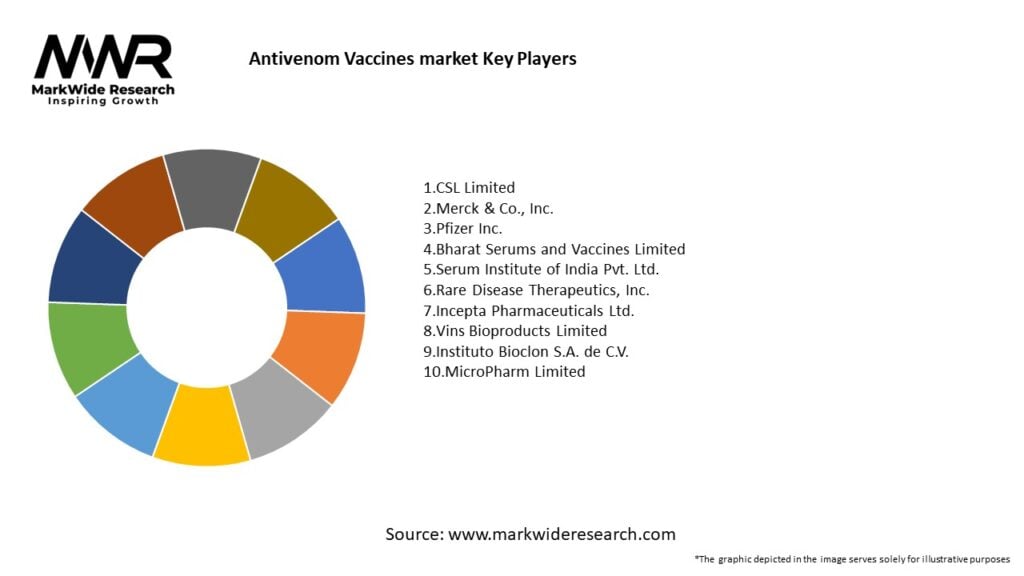
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
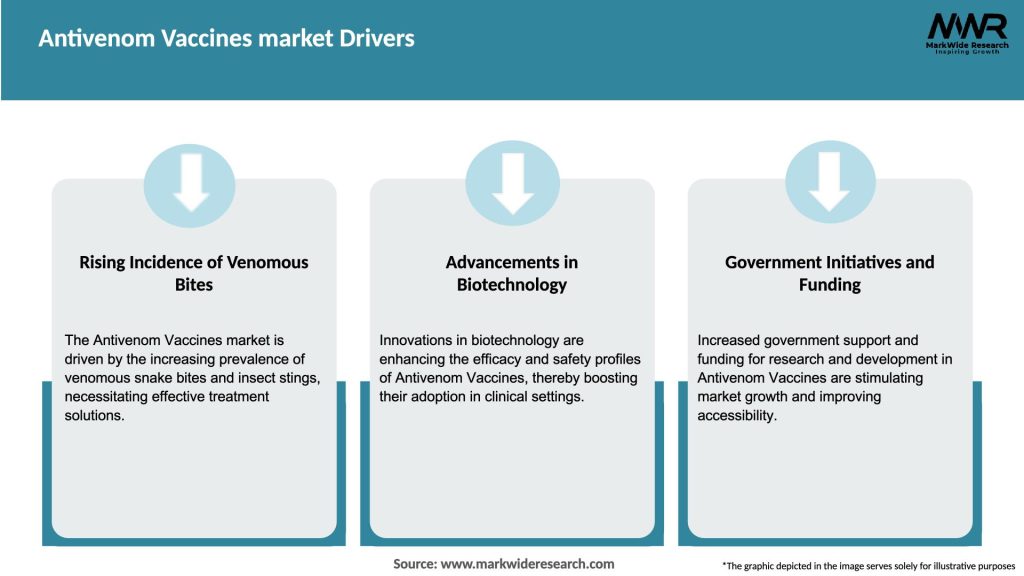
Market Drivers
Market Restraints
Market Opportunities

Market Dynamics
The Antivenom Vaccines market is driven by several factors, including the growing incidence of venomous bites and stings, advancements in biotechnology, government initiatives, and increased healthcare expenditure. However, market growth faces challenges such as the high cost of vaccines, lack of accessibility in remote areas, adverse effects, and regulatory hurdles. Nonetheless, opportunities exist in emerging markets, collaborations and partnerships, technological advancements, and awareness campaigns.
Regional Analysis
The Antivenom Vaccines market exhibits regional variations based on the prevalence of venomous creatures, healthcare infrastructure, and government initiatives. Regions with a high incidence of venomous bites, such as sub-Saharan Africa, Southeast Asia, and parts of Latin America, present significant opportunities for market growth. These regions are characterized by a rising demand for antivenom vaccines and increasing investments in healthcare infrastructure.
In developed regions like North America and Europe, the market is driven by advancements in biotechnology, research and development activities, and strong regulatory frameworks. These regions also witness collaborations between pharmaceutical companies and research institutions, fostering market growth through innovation.
Competitive Landscape
Leading Companies in Antivenom Vaccines Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
Segmentation
The Antivenom Vaccines market can be segmented based on various factors such as:
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
Market Key Trends
Covid-19 Impact
The Covid-19 pandemic has had an indirect impact on the Antivenom Vaccines market. While the market itself is not directly related to the pandemic, the healthcare infrastructure strain caused by Covid-19 has affected the availability and accessibility of antivenom vaccines in some regions. The redirection of healthcare resources, limited access to healthcare facilities, and disruptions in the supply chain have posed challenges for the market. However, with the gradual recovery of healthcare systems and the resumption of routine healthcare services, the market is expected to regain momentum.
Key Industry Developments
Analyst Suggestions
Future Outlook
The future of the Antivenom Vaccines market looks promising, with opportunities for growth and innovation. Technological advancements in biotechnology, increasing investments in research and development, and government initiatives to control venomous creatures will continue to drive market growth. The expansion into emerging markets, collaborations for innovation, and the focus on affordability and accessibility will shape the future landscape of the market. By addressing the challenges and capitalizing on the opportunities, the Antivenom Vaccines market is poised for significant development and advancements in the coming years.
Conclusion
The Antivenom Vaccines market is witnessing robust growth, driven by the increasing incidence of venomous bites and stings globally. Antivenom vaccines provide a vital treatment option, neutralizing the toxic effects of venom and improving patient outcomes. Despite challenges such as high costs, limited accessibility, adverse effects, and regulatory hurdles, the market offers significant opportunities for pharmaceutical companies, healthcare providers, governments, and research institutions. Collaboration, technological advancements, awareness campaigns, and investments in healthcare infrastructure are key factors that will shape the future of the market. With continued innovation and a focus on affordability and accessibility, the Antivenom Vaccines market is poised for a positive outlook, contributing to public health and saving lives worldwide.
What is Antivenom Vaccines?
Antivenom vaccines are medical products designed to provide immunity against venomous bites or stings from various animals, such as snakes, spiders, and scorpions. They work by introducing a small amount of venom to stimulate the immune system to produce antibodies.
What are the key players in the Antivenom Vaccines market?
Key players in the Antivenom Vaccines market include companies like Sanofi, CSL Behring, and Merck, which are known for their contributions to the development and production of antivenom products. These companies focus on research and innovation to enhance the efficacy and safety of antivenoms, among others.
What are the growth factors driving the Antivenom Vaccines market?
The Antivenom Vaccines market is driven by increasing incidences of venomous bites, rising awareness about snakebite treatment, and advancements in vaccine technology. Additionally, the growing healthcare infrastructure in developing regions contributes to market growth.
What challenges does the Antivenom Vaccines market face?
The Antivenom Vaccines market faces challenges such as high production costs, limited availability of certain antivenoms, and the complexity of developing effective vaccines for diverse venom types. Regulatory hurdles and the need for extensive clinical trials also pose significant challenges.
What opportunities exist in the Antivenom Vaccines market?
Opportunities in the Antivenom Vaccines market include the potential for new product development targeting specific venom types and the expansion of distribution networks in underserved regions. Collaborations between public health organizations and pharmaceutical companies can also enhance access to antivenoms.
What trends are shaping the Antivenom Vaccines market?
Trends in the Antivenom Vaccines market include the increasing use of monoclonal antibodies for antivenom production and the integration of advanced technologies such as recombinant DNA technology. There is also a growing emphasis on sustainable practices in the production of antivenoms.
Antivenom Vaccines market
| Segmentation Details | Description |
|---|---|
| Product Type | Monovalent, Bivalent, Trivalent, Polyvalent |
| Delivery Mode | Intravenous, Subcutaneous, Intramuscular, Oral |
| End User | Hospitals, Clinics, Research Institutions, Veterinary Practices |
| Application | Snake Bites, Scorpion Stings, Insect Bites, Others |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in Antivenom Vaccines Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at