444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The 3ml-10ml Pre-filled Flush Syringes Market addresses the demand for convenient and ready-to-use medical devices designed for flushing intravenous (IV) catheters and maintaining catheter patency in healthcare settings. These pre-filled syringes contain sterile saline solution or heparinized saline and are utilized for flushing and clearing IV lines to prevent occlusions, maintain catheter function, and reduce the risk of bloodstream infections. The market serves hospitals, clinics, ambulatory care centers, and other healthcare facilities globally.
Meaning
3ml-10ml pre-filled flush syringes are medical devices prefilled with sterile saline solution or heparinized saline, specifically designed for flushing intravenous (IV) catheters and maintaining catheter patency. These syringes offer convenience, accuracy, and safety in healthcare settings, ensuring efficient IV line maintenance, preventing catheter occlusions, and reducing the risk of bloodstream infections.
Executive Summary
The 3ml-10ml Pre-filled Flush Syringes Market is driven by the increasing prevalence of catheter-related complications, growing emphasis on infection control measures, and rising adoption of standardized flush protocols in healthcare facilities. Market players focus on product differentiation, quality assurance, and regulatory compliance to meet stringent safety standards and address the diverse needs of healthcare providers and patients. The market offers a range of pre-filled flush syringe products tailored to different clinical applications and patient populations.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The 3ml-10ml Pre-filled Flush Syringes Market operates in a dynamic healthcare landscape characterized by evolving clinical practices, regulatory changes, technological advancements, and market competition. Market dynamics such as catheter utilization trends, infection control practices, regulatory mandates, and cost containment pressures influence market growth, product adoption, and industry strategies.
Regional Analysis
The 3ml-10ml Pre-filled Flush Syringes Market exhibits regional variations in demand, adoption rates, regulatory requirements, and market dynamics. Regional factors such as healthcare infrastructure, catheter utilization patterns, infection control practices, and economic conditions shape market performance and growth opportunities across different geographic regions.
Competitive Landscape
Leading Companies in 3ml-10ml Pre-filled Flush Syringes Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
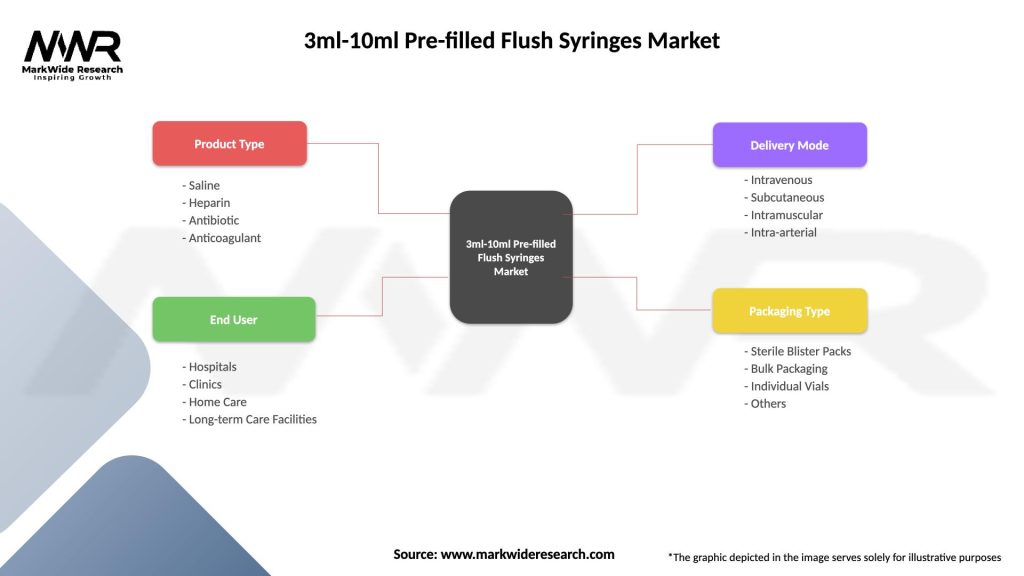
Segmentation
The 3ml-10ml Pre-filled Flush Syringes Market can be segmented based on various factors including:
Segmentation provides insights into market dynamics, customer preferences, and growth opportunities tailored to specific market segments, enabling market players to formulate targeted strategies and optimize product offerings.
Category-wise Insights
Key Benefits for Healthcare Providers and Patients
SWOT Analysis
Market Key Trends
Covid-19 Impact
The Covid-19 pandemic has underscored the importance of infection control practices, patient safety, and healthcare preparedness, influencing demand for pre-filled flush syringes. Healthcare facilities prioritize catheter maintenance, infection prevention, and patient safety, driving market demand for sterile flush solutions and disposable medical devices. Market players adapt to changing market dynamics, supply chain disruptions, and regulatory challenges to meet healthcare needs and support pandemic response efforts.
Key Industry Developments
Analyst Suggestions
Future Outlook
The 3ml-10ml Pre-filled Flush Syringes Market is poised for growth driven by increasing healthcare awareness, adoption of infection control practices, and demand for standardized catheter maintenance solutions. Market players focus on innovation, quality assurance, and market expansion strategies to capitalize on emerging opportunities, address unmet healthcare needs, and drive market growth. The market’s future will be shaped by technological advancements, regulatory developments, and industry collaborations aimed at enhancing patient safety, clinical outcomes, and healthcare efficiency.
Conclusion
The 3ml-10ml Pre-filled Flush Syringes Market plays a vital role in intravenous catheter maintenance, infection prevention, and patient safety in healthcare settings. Market players offer a range of pre-filled flush syringe products designed to meet diverse clinical needs, enhance workflow efficiency, and optimize patient care. With a focus on innovation, quality, and regulatory compliance, the market continues to evolve to address evolving healthcare challenges and support best practices in catheter care. By prioritizing patient safety, regulatory compliance, and market differentiation, industry stakeholders can drive market growth, foster innovation, and shape the future of catheter maintenance solutions in healthcare.
What is 3ml-10ml Pre-filled Flush Syringes?
3ml-10ml Pre-filled Flush Syringes are medical devices designed to deliver precise doses of medication or saline solutions. They are commonly used in various healthcare settings for flushing catheters and administering medications.
What are the key players in the 3ml-10ml Pre-filled Flush Syringes Market?
Key players in the 3ml-10ml Pre-filled Flush Syringes Market include Becton, Dickinson and Company, Gerresheimer AG, and Medtronic, among others.
What are the growth factors driving the 3ml-10ml Pre-filled Flush Syringes Market?
The growth of the 3ml-10ml Pre-filled Flush Syringes Market is driven by the increasing prevalence of chronic diseases, the rising demand for home healthcare solutions, and advancements in syringe technology.
What challenges does the 3ml-10ml Pre-filled Flush Syringes Market face?
Challenges in the 3ml-10ml Pre-filled Flush Syringes Market include stringent regulatory requirements, potential safety concerns related to device malfunction, and competition from alternative drug delivery systems.
What opportunities exist in the 3ml-10ml Pre-filled Flush Syringes Market?
Opportunities in the 3ml-10ml Pre-filled Flush Syringes Market include the development of innovative syringe designs, increasing investments in healthcare infrastructure, and the growing trend of personalized medicine.
What trends are shaping the 3ml-10ml Pre-filled Flush Syringes Market?
Trends in the 3ml-10ml Pre-filled Flush Syringes Market include the rising adoption of pre-filled syringes for biologics, advancements in materials for better safety and efficacy, and the integration of smart technology for enhanced patient monitoring.
3ml-10ml Pre-filled Flush Syringes Market
| Segmentation Details | Description |
|---|---|
| Product Type | Saline, Heparin, Antibiotic, Anticoagulant |
| End User | Hospitals, Clinics, Home Care, Long-term Care Facilities |
| Delivery Mode | Intravenous, Subcutaneous, Intramuscular, Intra-arterial |
| Packaging Type | Sterile Blister Packs, Bulk Packaging, Individual Vials, Others |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in 3ml-10ml Pre-filled Flush Syringes Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at