444 Alaska Avenue
Suite #BAA205 Torrance, CA 90503 USA
+1 424 999 9627
24/7 Customer Support
sales@markwideresearch.com
Email us at
Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at
Corporate User License
Unlimited User Access, Post-Sale Support, Free Updates, Reports in English & Major Languages, and more
$3450
Market Overview
The 3D printing drug market represents an innovative approach to pharmaceutical manufacturing, utilizing additive manufacturing technologies to produce personalized dosage forms with precise drug dosages and release profiles. This emerging market is driven by advancements in 3D printing technologies, growing demand for personalized medicine, and the need for novel drug delivery systems to enhance patient compliance and therapeutic outcomes.
Meaning
3D printing drug technology involves the layer-by-layer fabrication of pharmaceutical products using 3D printing equipment and pharmaceutical-grade materials. This transformative approach enables the customization of dosage forms, the incorporation of multiple active ingredients, and the modulation of drug release kinetics, offering new possibilities for personalized medicine and patient-centered care.
Executive Summary
The 3D printing drug market is characterized by rapid technological innovation, increasing investment in research and development, and strategic collaborations between pharmaceutical companies and technology providers. Key market insights reveal a shift towards decentralized manufacturing, on-demand production, and patient-specific formulations, driving market growth and adoption. However, regulatory challenges, intellectual property issues, and scalability concerns pose barriers to market expansion and commercialization.
Important Note: The companies listed in the image above are for reference only. The final study will cover 18–20 key players in this market, and the list can be adjusted based on our client’s requirements.
Key Market Insights
Market Drivers
Market Restraints
Market Opportunities
Market Dynamics
The 3D printing drug market operates within a dynamic ecosystem shaped by technological innovation, regulatory evolution, market competition, and patient demand. These dynamics influence market trends, investment decisions, and strategic partnerships, requiring stakeholders to adapt to changing market conditions and emerging opportunities.
Regional Analysis
The 3D printing drug market exhibits regional variations in adoption rates, regulatory frameworks, and market dynamics. While developed markets like North America and Europe lead in technology innovation and investment, emerging economies in Asia-Pacific and Latin America offer growth opportunities due to expanding healthcare infrastructure and rising demand for personalized medicine.
Competitive Landscape
Leading Companies in the 3D Printing Drug Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
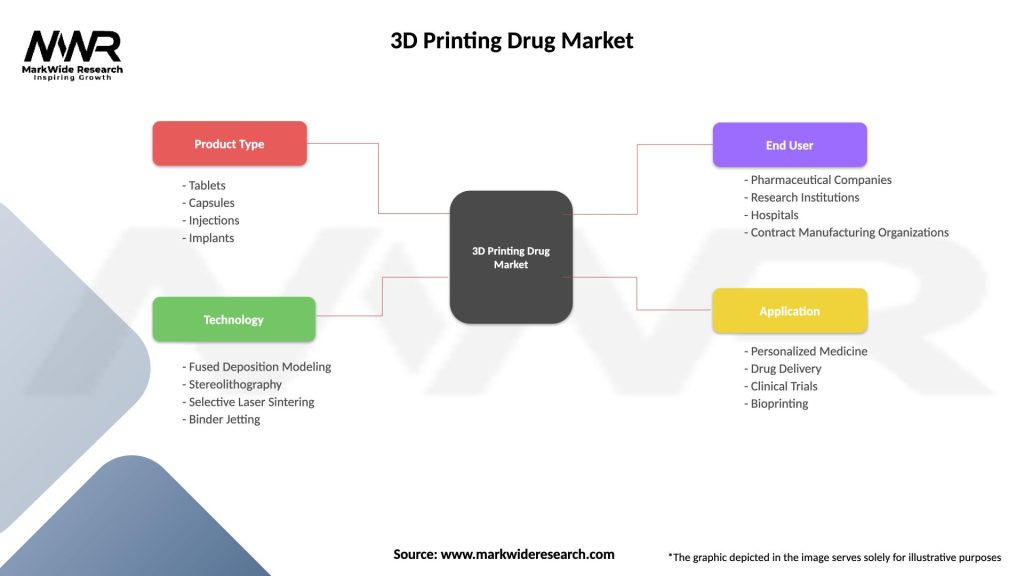
Segmentation
The 3D printing drug market can be segmented based on factors such as technology type, application area, therapeutic indication, and geography. Understanding these segmentation variables enables market players to identify target markets, tailor product offerings, and develop targeted marketing strategies to meet customer needs effectively.
Category-wise Insights
Key Benefits for Industry Participants and Stakeholders
SWOT Analysis
A SWOT analysis provides insights into the strengths, weaknesses, opportunities, and threats facing the 3D printing drug market:
Strengths:
Weaknesses:
Opportunities:
Threats:
Market Key Trends
Covid-19 Impact
The COVID-19 pandemic has influenced the 3D printing drug market in several ways:
Key Industry Developments
Analyst Suggestions
Future Outlook
The future outlook for the 3D printing drug market is promising, driven by technological innovation, regulatory advancements, and growing demand for personalized medicine. Despite challenges such as regulatory uncertainty and competitive pressures, industry stakeholders can capitalize on opportunities presented by digital health integration, biosimilar development, and material innovation to shape the future of pharmaceutical manufacturing and enhance healthcare outcomes. Key trends such as decentralized manufacturing, virtual drug development, and digital health integration will shape the future landscape of the 3D printing drug market, driving innovation and market expansion.
Conclusion
In conclusion, the 3D printing drug market represents a transformative approach to pharmaceutical manufacturing, offering unprecedented flexibility, precision, and customization in drug design and formulation. Despite challenges such as regulatory uncertainty and technological limitations, the market holds significant promise for personalized medicine, targeted drug delivery, and improved patient outcomes. By leveraging technological innovation, regulatory collaboration, and strategic partnerships, industry stakeholders can unlock new opportunities, overcome barriers, and drive the adoption of 3D printing drugs, ultimately advancing healthcare delivery and shaping the future of pharmaceutical manufacturing.
What is 3D Printing Drug?
3D printing drug refers to the use of additive manufacturing technologies to create pharmaceutical products, including personalized medications and complex drug delivery systems. This innovative approach allows for precise control over drug composition and dosage forms.
What are the key players in the 3D Printing Drug Market?
Key players in the 3D Printing Drug Market include Aprecia Pharmaceuticals, FabRx, and Merck, among others. These companies are at the forefront of developing and commercializing 3D printed pharmaceuticals and technologies.
What are the growth factors driving the 3D Printing Drug Market?
The growth of the 3D Printing Drug Market is driven by factors such as the increasing demand for personalized medicine, advancements in printing technologies, and the ability to produce complex drug formulations. Additionally, the potential for reduced production costs and improved patient compliance contributes to market expansion.
What challenges does the 3D Printing Drug Market face?
The 3D Printing Drug Market faces challenges including regulatory hurdles, the need for standardization in manufacturing processes, and concerns regarding the scalability of production. These factors can hinder widespread adoption and integration into existing pharmaceutical practices.
What opportunities exist in the 3D Printing Drug Market?
Opportunities in the 3D Printing Drug Market include the development of on-demand drug manufacturing, the potential for creating complex dosage forms, and the ability to address unmet medical needs. As technology advances, new applications in bioprinting and regenerative medicine may also emerge.
What trends are shaping the 3D Printing Drug Market?
Trends shaping the 3D Printing Drug Market include the increasing integration of artificial intelligence in drug design, the rise of bioprinting for tissue engineering, and the growing focus on sustainable manufacturing practices. These trends are expected to enhance the efficiency and effectiveness of drug development processes.
3D Printing Drug Market
| Segmentation Details | Description |
|---|---|
| Product Type | Tablets, Capsules, Injections, Implants |
| Technology | Fused Deposition Modeling, Stereolithography, Selective Laser Sintering, Binder Jetting |
| End User | Pharmaceutical Companies, Research Institutions, Hospitals, Contract Manufacturing Organizations |
| Application | Personalized Medicine, Drug Delivery, Clinical Trials, Bioprinting |
Please note: The segmentation can be entirely customized to align with our client’s needs.
Leading Companies in the 3D Printing Drug Market:
Please note: This is a preliminary list; the final study will feature 18–20 leading companies in this market. The selection of companies in the final report can be customized based on our client’s specific requirements.
North America
o US
o Canada
o Mexico
Europe
o Germany
o Italy
o France
o UK
o Spain
o Denmark
o Sweden
o Austria
o Belgium
o Finland
o Turkey
o Poland
o Russia
o Greece
o Switzerland
o Netherlands
o Norway
o Portugal
o Rest of Europe
Asia Pacific
o China
o Japan
o India
o South Korea
o Indonesia
o Malaysia
o Kazakhstan
o Taiwan
o Vietnam
o Thailand
o Philippines
o Singapore
o Australia
o New Zealand
o Rest of Asia Pacific
South America
o Brazil
o Argentina
o Colombia
o Chile
o Peru
o Rest of South America
The Middle East & Africa
o Saudi Arabia
o UAE
o Qatar
o South Africa
o Israel
o Kuwait
o Oman
o North Africa
o West Africa
o Rest of MEA
Trusted by Global Leaders
Fortune 500 companies, SMEs, and top institutions rely on MWR’s insights to make informed decisions and drive growth.
ISO & IAF Certified
Our certifications reflect a commitment to accuracy, reliability, and high-quality market intelligence trusted worldwide.
Customized Insights
Every report is tailored to your business, offering actionable recommendations to boost growth and competitiveness.
Multi-Language Support
Final reports are delivered in English and major global languages including French, German, Spanish, Italian, Portuguese, Chinese, Japanese, Korean, Arabic, Russian, and more.
Unlimited User Access
Corporate License offers unrestricted access for your entire organization at no extra cost.
Free Company Inclusion
We add 3–4 extra companies of your choice for more relevant competitive analysis — free of charge.
Post-Sale Assistance
Dedicated account managers provide unlimited support, handling queries and customization even after delivery.
GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


GET A FREE SAMPLE REPORT
This free sample study provides a complete overview of the report, including executive summary, market segments, competitive analysis, country level analysis and more.
ISO AND IAF CERTIFIED


Suite #BAA205 Torrance, CA 90503 USA
24/7 Customer Support
Email us at